TUDORZA PRESSAIR Drug Patent Profile
✉ Email this page to a colleague
When do Tudorza Pressair patents expire, and what generic alternatives are available?
Tudorza Pressair is a drug marketed by Covis and is included in one NDA. There are four patents protecting this drug.
This drug has one hundred and fifty-six patent family members in forty-six countries.
The generic ingredient in TUDORZA PRESSAIR is aclidinium bromide. There is one drug master file entry for this compound. Two suppliers are listed for this compound. Additional details are available on the aclidinium bromide profile page.
DrugPatentWatch® Generic Entry Outlook for Tudorza Pressair
Tudorza Pressair was eligible for patent challenges on July 23, 2016.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be February 10, 2025. This may change due to patent challenges or generic licensing.
Indicators of Generic Entry
Summary for TUDORZA PRESSAIR
| International Patents: | 156 |
| US Patents: | 4 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 2 |
| Raw Ingredient (Bulk) Api Vendors: | 56 |
| Patent Applications: | 377 |
| Drug Prices: | Drug price information for TUDORZA PRESSAIR |
| What excipients (inactive ingredients) are in TUDORZA PRESSAIR? | TUDORZA PRESSAIR excipients list |
| DailyMed Link: | TUDORZA PRESSAIR at DailyMed |
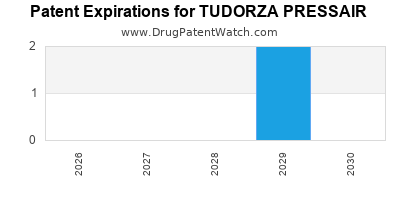

DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for TUDORZA PRESSAIR
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Pharmacology for TUDORZA PRESSAIR
| Drug Class | Anticholinergic |
| Mechanism of Action | Cholinergic Antagonists |
Anatomical Therapeutic Chemical (ATC) Classes for TUDORZA PRESSAIR
US Patents and Regulatory Information for TUDORZA PRESSAIR
TUDORZA PRESSAIR is protected by four US patents.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of TUDORZA PRESSAIR is ⤷ Sign Up.
This potential generic entry date is based on patent ⤷ Sign Up.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
Patents protecting TUDORZA PRESSAIR
Dosage and formulation
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: MAINTENANCE TREATMENT OF CHRONIC OBSTRUCTIVE PULMONARY DISEASE (COPD)
Dosage and formulation
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: MAINTENANCE TREATMENT OF CHRONIC OBSTRUCTIVE PULMONARY DISEASE (COPD)
Inhaler for the administration of powdered pharmaceuticals, and a powder cartridge system for use with this inhaler
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Quinuclidine derivatives and their use as muscarinic M3 receptor ligands
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: MAINTENANCE TREATMENT OF CHRONIC OBSTRUCTIVE PULMONARY DISEASE (COPD)
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Covis | TUDORZA PRESSAIR | aclidinium bromide | POWDER, METERED;INHALATION | 202450-001 | Jul 23, 2012 | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | Y | ⤷ Sign Up | |||
| Covis | TUDORZA PRESSAIR | aclidinium bromide | POWDER, METERED;INHALATION | 202450-001 | Jul 23, 2012 | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | Y | ⤷ Sign Up | |||
| Covis | TUDORZA PRESSAIR | aclidinium bromide | POWDER, METERED;INHALATION | 202450-001 | Jul 23, 2012 | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | Y | Y | ⤷ Sign Up | ||
| Covis | TUDORZA PRESSAIR | aclidinium bromide | POWDER, METERED;INHALATION | 202450-001 | Jul 23, 2012 | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | Y | ⤷ Sign Up | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for TUDORZA PRESSAIR
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Covis | TUDORZA PRESSAIR | aclidinium bromide | POWDER, METERED;INHALATION | 202450-001 | Jul 23, 2012 | ⤷ Sign Up | ⤷ Sign Up |
| Covis | TUDORZA PRESSAIR | aclidinium bromide | POWDER, METERED;INHALATION | 202450-001 | Jul 23, 2012 | ⤷ Sign Up | ⤷ Sign Up |
| Covis | TUDORZA PRESSAIR | aclidinium bromide | POWDER, METERED;INHALATION | 202450-001 | Jul 23, 2012 | ⤷ Sign Up | ⤷ Sign Up |
| Covis | TUDORZA PRESSAIR | aclidinium bromide | POWDER, METERED;INHALATION | 202450-001 | Jul 23, 2012 | ⤷ Sign Up | ⤷ Sign Up |
| Covis | TUDORZA PRESSAIR | aclidinium bromide | POWDER, METERED;INHALATION | 202450-001 | Jul 23, 2012 | ⤷ Sign Up | ⤷ Sign Up |
| Covis | TUDORZA PRESSAIR | aclidinium bromide | POWDER, METERED;INHALATION | 202450-001 | Jul 23, 2012 | ⤷ Sign Up | ⤷ Sign Up |
| Covis | TUDORZA PRESSAIR | aclidinium bromide | POWDER, METERED;INHALATION | 202450-001 | Jul 23, 2012 | ⤷ Sign Up | ⤷ Sign Up |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
EU/EMA Drug Approvals for TUDORZA PRESSAIR
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Covis Pharma Europe B.V. | Bretaris Genuair | aclidinium bromide | EMEA/H/C/002706 Bretaris Genuair is indicated as a maintenance bronchodilator treatment to relieve symptoms in adult patients with chronic obstructive pulmonary disease (COPD). |
Authorised | no | no | no | 2012-07-20 | |
| Covis Pharma Europe B.V. | Eklira Genuair | aclidinium bromide | EMEA/H/C/002211 Eklira Genuair is indicated as a maintenance bronchodilator treatment to relieve symptoms in adult patients with chronic obstructive pulmonary disease (COPD). |
Authorised | no | no | no | 2012-07-20 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for TUDORZA PRESSAIR
See the table below for patents covering TUDORZA PRESSAIR around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Taiwan | 200938232 | Novel dosage and formulation | ⤷ Sign Up |
| Croatia | P20220929 | ⤷ Sign Up | |
| Australia | 719449 | ⤷ Sign Up | |
| Israel | 180378 | משאף למתן תרופות אבקתיות, ומערכת לאחסון האבקה לשימוש עם משאף זה (Inhaler for the administration of powdered pharmaceuticals and a powder cartridge system for use with said inhaler) | ⤷ Sign Up |
| Denmark | 1200431 | ⤷ Sign Up | |
| Mexico | 2007000592 | INHALADOR PARA LA ADMINISTRACION DE FARMACEUTICOS EN POLVO, Y UN SISTEMA DE CARTUCHOS DE POLVO PARA USO CON ESTE INHALADOR. (INHALER FOR THE ADMINISTRATION OF POWDERED PHARMACEUTICALS, AND A POWDER CARTRIDGE SYSTEM FOR USE WITH THIS INHALER.) | ⤷ Sign Up |
| Peru | 03972001 | ⤷ Sign Up | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for TUDORZA PRESSAIR
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1200431 | PA2013001,C1200431 | Lithuania | ⤷ Sign Up | PRODUCT NAME: ACLIDINII BROMIDUM; REGISTRATION NO/DATE: EU/1/12/778/001 - EU/1/12/778/003, 2012 07 20 EU/1/12/781/001 - EU/1/12/781/003 20120720 |
| 1200431 | PA2013001 | Lithuania | ⤷ Sign Up | PRODUCT NAME: ACLIDINII BROMIDUM; REGISTRATION NO/DATE: EU/1/12/778/001 - EU/1/12/778/003, 2012 07 20 EU/1/12/781/001 - EU/1/12/781/003 20120720 |
| 1200431 | C300573 | Netherlands | ⤷ Sign Up | PRODUCT NAME: ACLIDINIUMZOUT MET EEN FARMACEUTISCH AANVAARDBAAR ANION VAN EEN EEN- OF MEERWAARDIG ZUUR, IN HET BIJZONDER ACLIDINIUMBROMIDE; REGISTRATION NO/DATE: EU/1/12/778/001-003EU/1/12/781/001-003 2012200720 |
| 1200431 | C01200431/01 | Switzerland | ⤷ Sign Up | PRODUCT NAME: ACLIDINIUM; REGISTRATION NO/DATE: SWISSMEDIC 62590 25.04.2013 |
| 1200431 | 2013C/001 | Belgium | ⤷ Sign Up | PRODUCT NAME: SEL D'ACLIDINIUM AVEC UN ANION PHARMACEUTIQUEMENT ACCEPTABLE D'UN ACIDE MONO OU POLYVALENT EN PARTICULIER LE BROMURE D'ACLIDINIUM; AUTHORISATION NUMBER AND DATE: EU/1/12/781/001 20120725 |
| 1200431 | SPC/GB13/006 | United Kingdom | ⤷ Sign Up | PRODUCT NAME: ACLIDINIUM SALT WITH PHARMACEUTICALLY ACCEPTABLE ANION OF A MONO OR POLYVALENT ACID ESPECIALLY AS ACLIDINIUM BROMIDE; REGISTERED: UK EU/1/12/778/001 20120720; UK EU/1/12/778/002 20120720; UK EU/1/12/778/003 20120720; UK EU/1/12/781/001 20120720; UK EU/1/12/781/002 20120720; UK EU/1/12/781/003 20120720 |
| 1200431 | 13C0001 | France | ⤷ Sign Up | PRODUCT NAME: SEL D'ACLIDINIUM AVEC UN ANION PHARMACEUTIQUEMENT ACCEPTABLE D'UN ACIDE MONO OU POLYVALENT, EN PARTICULIER LE BROMURE D'ACLIDINIUM; REGISTRATION NO/DATE: EU/1/12/778/001-003 20120720 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |


