TRUVADA Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Truvada, and when can generic versions of Truvada launch?
Truvada is a drug marketed by Gilead and is included in one NDA. There are four patents protecting this drug and two Paragraph IV challenges.
This drug has sixty-two patent family members in twenty-nine countries.
The generic ingredient in TRUVADA is emtricitabine; tenofovir disoproxil fumarate. There are eighteen drug master file entries for this compound. Twenty-nine suppliers are listed for this compound. Additional details are available on the emtricitabine; tenofovir disoproxil fumarate profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Truvada
A generic version of TRUVADA was approved as emtricitabine; tenofovir disoproxil fumarate by TEVA PHARMS USA on June 8th, 2017.
Summary for TRUVADA
| International Patents: | 62 |
| US Patents: | 4 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 114 |
| Clinical Trials: | 227 |
| Formulation / Manufacturing: | see details |
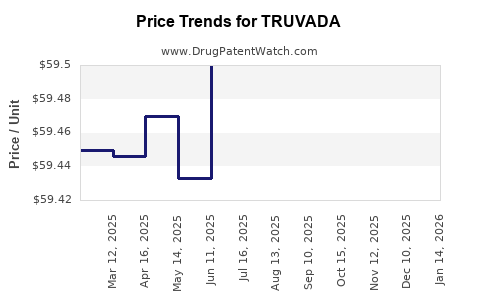
| Drug Prices: | Drug price information for TRUVADA |
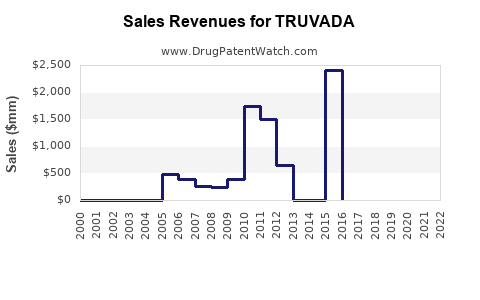
| Drug Sales Revenues: | Drug sales revenues for TRUVADA |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for TRUVADA |
| What excipients (inactive ingredients) are in TRUVADA? | TRUVADA excipients list |
| DailyMed Link: | TRUVADA at DailyMed |
Recent Clinical Trials for TRUVADA
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Assistance Publique - Hôpitaux de Paris, FRANCE | Phase 3 |
| Ministry of Health, Thailand | Phase 3 |
| Chiang Mai University, Thailand | Phase 3 |
Pharmacology for TRUVADA
Anatomical Therapeutic Chemical (ATC) Classes for TRUVADA
Paragraph IV (Patent) Challenges for TRUVADA
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| TRUVADA | Tablets | emtricitabine; tenofovir disoproxil fumarate | 100 mg/150 mg 133 mg/200 mg 167 mg/250 mg | 021752 | 1 | 2017-05-19 |
| TRUVADA | Tablets | emtricitabine; tenofovir disoproxil fumarate | 200 mg/300 mg | 021752 | 1 | 2008-09-26 |
US Patents and Regulatory Information for TRUVADA
TRUVADA is protected by six US patents.
Patents protecting TRUVADA
Compositions and methods for combination antiviral therapy
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: TREATMENT OF HIV
Compositions and methods for combination antiviral therapy
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: METHOD OF TREATMENT OF ADULTS INFECTED WITH HIV-1
Compositions and methods for combination antiviral therapy
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: TREATMENT OF HIV-1 INFECTION IN PEDIATRIC PATIENTS 12 YEARS OF AGE AND OLDER
Compositions and methods for combination antiviral therapy
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: TREATMENT OF HIV INFECTION
Compositions and methods for combination antiviral therapy
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: TREATMENT OF HIV INFECTION
Compositions and methods for combination antiviral therapy
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: TREATMENT OF HIV INFECTION
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Gilead | TRUVADA | emtricitabine; tenofovir disoproxil fumarate | TABLET;ORAL | 021752-002 | Mar 10, 2016 | AB | RX | Yes | No | ⤷ Sign Up | ⤷ Sign Up | ⤷ Sign Up | |||
| Gilead | TRUVADA | emtricitabine; tenofovir disoproxil fumarate | TABLET;ORAL | 021752-001 | Aug 2, 2004 | AB | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | Y | ⤷ Sign Up | ||
| Gilead | TRUVADA | emtricitabine; tenofovir disoproxil fumarate | TABLET;ORAL | 021752-003 | Mar 10, 2016 | AB | RX | Yes | No | ⤷ Sign Up | ⤷ Sign Up | ⤷ Sign Up | |||
| Gilead | TRUVADA | emtricitabine; tenofovir disoproxil fumarate | TABLET;ORAL | 021752-004 | Mar 10, 2016 | AB | RX | Yes | No | ⤷ Sign Up | ⤷ Sign Up | ⤷ Sign Up | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for TRUVADA
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Gilead | TRUVADA | emtricitabine; tenofovir disoproxil fumarate | TABLET;ORAL | 021752-001 | Aug 2, 2004 | ⤷ Sign Up | ⤷ Sign Up |
| Gilead | TRUVADA | emtricitabine; tenofovir disoproxil fumarate | TABLET;ORAL | 021752-004 | Mar 10, 2016 | ⤷ Sign Up | ⤷ Sign Up |
| Gilead | TRUVADA | emtricitabine; tenofovir disoproxil fumarate | TABLET;ORAL | 021752-003 | Mar 10, 2016 | ⤷ Sign Up | ⤷ Sign Up |
| Gilead | TRUVADA | emtricitabine; tenofovir disoproxil fumarate | TABLET;ORAL | 021752-003 | Mar 10, 2016 | ⤷ Sign Up | ⤷ Sign Up |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for TRUVADA
See the table below for patents covering TRUVADA around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Norway | 970385 | ⤷ Sign Up | |
| Ireland | 20060130 | ⤷ Sign Up | |
| Luxembourg | 91073 | ⤷ Sign Up | |
| Norway | 337917 | ⤷ Sign Up | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for TRUVADA
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 0582455 | 08C0021 | France | ⤷ Sign Up | PRODUCT NAME: EFAVIRENZ; EMTRICITABINE; TENOFOVIR DISOPROXIL FUMARATE; REGISTRATION NO/DATE: EU/1/07/430/001 20071213 |
| 3808743 | LUC00275 | Luxembourg | ⤷ Sign Up | PRODUCT NAME: COMBINAISON DE RILPIVIRINE OU D'UNE FORME THERAPEUTIQUEMENT EQUIVALENTE DE CELLE-CI PROTEGEE PAR LE BREVET DE BASE, TELLE QU'UN SEL D'ADDITION PHARMACEUTIQUEMENT ACCEPTABLE DE RILPIVIRINE, Y COMPRIS LE SEL D'ACIDE CHLORHYDRIQUE DE RILPIVIRINE, ET D'EMTRICITABINE; AUTHORISATION NUMBER AND DATE: EU/1/11/737/001-002 20111128 |
| 0915894 | C00915894/02 | Switzerland | ⤷ Sign Up | PRODUCT NAME: TENOFOVIRDISOPROXIL + EFAVIRENZ + EMTRICITABIN; REGISTRATION NUMBER/DATE: SWISSMEDIC 60011 20.11.2009 |
| 1663240 | 300851 | Netherlands | ⤷ Sign Up | PRODUCT NAME: COMBINATIE VAN: - RILPIVIRINE OF EEN THERAPEUTISCH EQUIVALENTE VORM DAARVAN ZOALS BESCHERMD DOOR HET BASISOCTROOI, ZOALS EEN FARMACEUTISCH AANVAARDBAAR ZOUT VAN RILPIVIRINE, WAARONDER HET HYDROCHLORIDEZOUT VAN RILPIVIRINE; - EMTRICITABINE; EN - TENOFOVIRALAFENAMIDE OF EEN FARMACEUTISCH AANVAARDBAAR ZOUT DAARVAN, IN HET BIJZONDER TENOFOVIRALAFENAMIDEFUMARAAT; REGISTRATION NO/DATE: EU/1/16/1112 20160623 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |


