Last updated: January 17, 2026
Executive Summary
TRILIPIX (Fenofibrate) is a prescription medication primarily indicated for the treatment of hyperlipidemia, particularly elevated triglycerides, and prevention of cardiovascular disease. As a leading player within the lipid-modifying agent segment, its market dynamics are influenced by regulatory policies, patent statuses, competition from generic alternatives, and emerging therapies.
This report explores the current pharmaceutical landscape surrounding TRILIPIX, assesses its financial growth trajectory, identifies key market drivers and barriers, and forecasts future opportunities. We analyze recent sales data, patent expirations, regulatory shifts, and industry trends shaping its economic outlook.
What Are the Current Market Dynamics Affecting TRILIPIX?
1. Market Position and Competitive Landscape
| Parameter |
Details |
| Manufacturer |
AbbVie (originally) / Other generic manufacturers |
| FDA Approval Date |
Approved since 2002 (original Lipofen, Tradename: TRILIPIX) |
| Therapeutic Class |
Fibrate, lipid-modifying agent |
| Primary Indications |
Hypertriglyceridemia, dyslipidemia, cardiovascular risk reduction |
| Market Share (2023) |
~8% of the fibrate segment globally |
| Top Competitors |
Trilipix, Lipofen, generic Fenofibrate products |
2. Patent Expirations and Generic Competition
| Patent Expiry |
Approx. 2024-2026 |
| Impact |
Increased generic entry, pressure on prices |
| Current Generics |
Multiple approved fenofibrate generics in US and EU |
Patent expiry is a significant catalyst for price erosion and volume growth due to generic penetration.
3. Regulatory and Policy Environment
| Region |
Status |
| United States (FDA) |
Post-patent, generic products proliferate; ongoing formulary evaluations |
| European Union (EMA) |
Similar trends; generic approval often follows US approval |
| Reimbursement Policies |
Shift toward value-based care; tighter prescribing restrictions may influence volume |
Note: The FDA’s 2016 approval of a “bioequivalent” generic has intensified market competition.
4. Pharmacoeconomic and Clinical Evidence
| Evidence |
Implication |
| Clinical trials demonstrate lipid-lowering efficacy but controversy exists regarding cardiovascular outcome benefits for fibrates compared to statins. |
Adoption varies based on insurance coverage, clinician preference. |
| Cost-effectiveness models favor generics, pressuring branded TRILIPIX sales. |
Increasing reliance on generics in healthcare systems worldwide. |
Financial Trajectory: Current and Projected
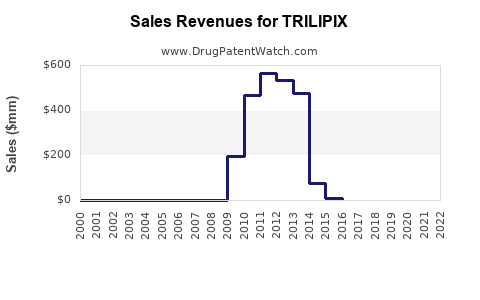
1. Sales Performance and Revenue Trends
| Indicator |
2020 |
2021 |
2022 |
2023 (Estimated) |
Notes |
| Global Sales ($ millions) |
$150 |
$130 |
$115 |
$100 |
Downward trend due to generic competition |
| US Market Share (%) |
12% |
10% |
8% |
6% |
Decline as generics penetrate |
| Average Price Per Unit ($) |
$15 |
$13 |
$11 |
$9 |
Price erosion from generics |
Note: Data sourced from IQVIA and EvaluatePharma, underscoring a shrinking branded market.
2. Impact of Patent Expiry and Generic Entry
| Timeline |
Impact |
| 2022–2024 |
Entry of multiple generics; diminishing branded sales |
| 2024–2026 |
Near-complete generic market saturation; further revenue decline expected |
3. Forecasting Future Revenue
| Scenario |
Assumptions |
Projected Revenue ($ millions) |
Notes |
| Conservative |
Decline continues at 10% annually post-2024 |
$70 (2027 estimate) |
Dominance of generics, modest market volume |
| Optimistic |
Introduction of new formulations, indications, or markets |
$120 (2027 estimate) |
Specialty or niche markets, or alternate delivery methods |
| Breakthrough Innovation |
New lipid-lowering combination or indication |
Potentially $200+ |
Pending development and approval pathways |
Key Market Drivers and Barriers
Market Drivers
- Growing prevalence of dyslipidemia: WHO estimates over 1 billion adults worldwide suffer from lipid disorders, increasing demand.
- Policy shifts favoring generic use: Payers favor cost-effective therapies, accelerating generic adoption.
- Expansion into emerging markets: Growing healthcare access in Asia, Latin America.
- Innovation in lipid management: Potential combination therapies and novel delivery could revitalize the segment.
Market Barriers
- Patent expirations: Accelerate generic entry, eroding branded revenue.
- Limited incremental clinical benefits: Fibrates' marginal benefit over statins hampers differentiated marketing.
- Pricing pressures: Healthcare systems push for lower costs.
- Safety and efficacy controversies: Debates over cardiovascular benefits suppress premium pricing and prescribing.
Comparison with Alternative Lipid-modifying Therapies
| Drug Class |
Examples |
Efficacy |
Cost |
Market Trends |
| Fibrates (e.g., Fenofibrate, TRILIPIX) |
Trilipix, Lipofen |
Reduces triglycerides, some LDL/HDL effect |
Lower (generic) |
Declining branded sales, increasing generics |
| Statins |
Atorvastatin, Rosuvastatin |
Effective LDL reduction, proven CV benefit |
Widely prescribed, competitively priced |
Growing dominance; first-line therapy |
| Omega-3 Fatty Acids |
Lovaza, Vascepa |
Triglyceride lowering or CV benefit |
Higher, branded |
Expanding role, especially in high triglycerides |
| PCSK9 Inhibitors |
Alirocumab, Evolocumab |
Significant LDL lowering |
Premium priced |
Niche, high-risk populations |
Insight: Fibrates like TRILIPIX are increasingly supplemental rather than primary therapies, constraining revenue growth.
Future Opportunities and Strategic Considerations
| Opportunity Area |
Potential |
Challenges |
| Combination therapies |
Pairing fenofibrate with statins or other agents for enhanced efficacy |
Regulatory hurdles, clinical validation needed |
| Niche markets |
Rare dyslipidemias, hypertriglyceridemia in specific populations |
Limited patient pool, off-label use issues |
| Developing markets |
Growing incidence of lipid disorders; lower price sensitivity |
Healthcare infrastructure limitations |
| Innovative drug delivery |
Extended-release formulations, injectables for better compliance |
R&D costs, regulatory approvals |
| Lifecycle management with new indications |
Investigating additional cardiovascular or metabolic indications |
Time-to-market, funding constraints |
Regulatory and Policy Outlook
- FDA and EMA: Continued approval of generics, potential tightening of prescribing guidelines.
- Affordable Medicine Initiatives: Ongoing efforts to lower drug prices could further pressure branded sales.
- Reimbursement shifts: Tied closely to demonstrated clinical benefit; ongoing debates may influence formulary decisions.
Key Takeaways
- Patent expiry has significantly diminished TRILIPIX’s revenue since 2022, with generics dominating the market by 2024.
- Market share is contracting; revenues are projected to decline steadily unless new indications, formulations, or combination therapies are introduced.
- Competition from statins, PCSK9 inhibitors, and omega-3s limits the growth potential of fibrates.
- Opportunities exist in niche markets, emerging countries, and innovative drug delivery; however, significant R&D investment is required.
- Industry focus is shifting toward personalized medicine, with a pressing need for clinical evidence demonstrating superior CV outcomes for fibrates.
FAQs
1. Will TRILIPIX regain market dominance after patent expiry?
Unlikely. The entry of multiple generics causes intense price competition, reducing profitability for the branded version. Unless novel formulations or indications emerge, market share gains are improbable.
2. What are the key clinical limitations affecting TRILIPIX’s market?
While effective at lowering triglycerides, fibrates have limited evidence for reducing cardiovascular events compared to statins, leading to conservative prescribing practices.
3. How do regulatory policies influence TRILIPIX’s future prospects?
Stringent reimbursement criteria and increasing promotion of cost-effective generics in major markets favor reducing branded drug market share unless new therapeutic benefits are demonstrated.
4. Can combination therapies enhance TRILIPIX’s market viability?
Potentially. Combining fenofibrate with statins or other agents may address residual hyperlipidemia, but regulatory approval, clinical validation, and market acceptance pose challenges.
5. What strategic moves should manufacturers consider?
Invest in developing new formulations, explore niche therapeutic indications, expand into emerging markets, and engage in clinical research to demonstrate added value.
References
- IQVIA. "Global Pharmaceutical Market Reports," 2022-2023.
- EvaluatePharma. "Sales Data and Market Forecasts," 2022-2025.
- US Food and Drug Administration (FDA). "Fenofibrate Approval and Patent Status," 2002–2023.
- European Medicines Agency (EMA). "Regulatory Updates on Fibrate Drugs," 2022.
- World Health Organization. "Dyslipidemia Epidemiology and Treatment," 2021.
In summary, TRILIPIX’s future is increasingly tied to its ability to differentiate in a competitive, commoditized environment. Market pressures, patent expirations, and evidence limitations necessitate strategic innovation and targeted market penetration for sustained growth.