Last updated: May 24, 2026
TRI-LUMA (fluocinolone acetonide 0.01% / hydroquinone 4% / tretinoin 0.05%) market dynamics and financial trajectory
TRI-LUMA is a niche, dermatology-focused triple-combination product for photodamaged facial skin disorders, with revenue driven by chronic demand, payer coverage, and single-wins in prescriber patterns rather than broad primary-care scale. Financial trajectory has been shaped by (1) competitive erosion from procedural and topical alternatives, (2) formulation and generic pressure typical for dermatology brands, and (3) channel inventory cycles that amplify year-to-year volatility.
What matters commercially: sustained premium pricing depends on continued branded marketing access and avoidance of broad generic substitution. Volatility is also tied to limited “must-have” indications and physician shift to lasers, chemical peels, and procedural modalities for melasma-adjacent complaints.
What is TRI-LUMA used for and what drives demand in dermatology markets?
TRI-LUMA is indicated for the topical treatment of exogenous ochronosis and melasma. Demand is concentrated in dermatology and aesthetic medicine clinics, with prescription volume correlated to seasonal and patient willingness to adhere to strict topical regimens and photoprotection.
Key demand drivers
- Chronic recurrence pattern: melasma is relapsing, supporting repeat courses and long-term patient management.
- Treatment regimen adherence: triple-combination regimens require careful use and sun protection, which can limit broad uptake but stabilizes demand among adherent patient populations.
- Specialist prescribing: dermatologists and practices with dedicated pigment-management protocols drive most volume.
- Payer approval dynamics: step therapy, quantity limits, and prior authorization affect fill rates more than clinical efficacy differentiators.
Commercial sensitivity
TRI-LUMA’s market sizing is less about “total addressable dermatology” and more about:
- the subset of patients with confirmed indication,
- willingness to pay under coverage rules,
- and ongoing competition from alternatives that do not require the same multi-ingredient regimen.
How has the TRI-LUMA competitive landscape evolved versus alternative topical depigmenting therapies?
TRI-LUMA competes in a crowded dermatology space that includes topical retinoids, corticosteroids, depigmenting agents, and combination products, plus non-topical and procedural options used in similar patient journeys.
Competing alternatives that influence TRI-LUMA share
- Other depigmenting topicals: hydroquinone variants, retinoid-based regimens, and pigment-focused actives.
- Combination maintenance strategies: off-label sequential approaches using moisturizers, antioxidants, and spot therapies.
- Procedural routes: laser and light-based treatments, chemical peels, and microneedling protocols that can displace maintenance topical use for some patient segments.
How competition impacts market dynamics
- Price and coverage pressure: competitors that are covered more consistently can capture formulary share even when efficacy profiles are comparable.
- Perceived complexity: multi-active topical regimens can lose patients to simpler regimens with adequate perceived results.
- Seasonality: pigment disorders respond to seasonal triggers, shifting prescription timing and replenishment behavior.
What are TRI-LUMA’s payer, formulary, and channel dynamics that shape sales?
Revenue and volume in topical dermatology products are strongly influenced by pharmacy benefit management practices, prior authorization gates, and patient out-of-pocket exposure.
Formulary behavior that tends to move TRI-LUMA
- Prior authorization and step edits: often used to require prior use of lower-cost agents.
- Quantity and days-supply controls: can cap early-course volume and drive partial fills.
- Coverage churn: changes in PBM formularies can shift share quickly in a single coverage year.
- Patient assistance and contracting: branded manufacturers can defend access with rebate and patient support where needed.
Channel inventory and ordering patterns
- Topical brands often show sales volatility linked to channel fill rather than true prescription demand swings.
- When wholesalers normalize inventory, reported revenue can drop even if real-world demand holds.
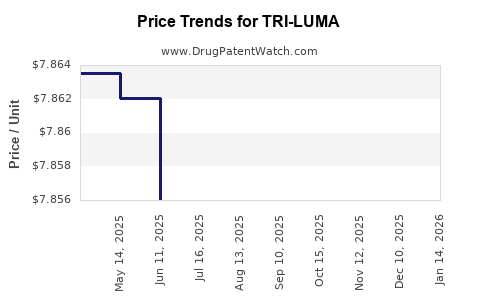
How does TRI-LUMA pricing and net-to-gross economics affect its financial trajectory?
For established dermatology brands, net sales trajectory is driven more by rebate intensity and contracting than by list price changes.
Net-to-gross mechanics likely affecting TRI-LUMA
- Rebate escalation to maintain formulary position under competitive pressure.
- Payer-specific contracting: higher rebate rates for preferred coverage tiers.
- Seasonal promotional activity: higher spend periods can temporarily boost sell-in and then reverse.
What to watch in financial statements
- Net sales growth vs. volume growth: if list price rises but volume stagnates, net sales growth can remain flat or decline.
- Gross margin drift: indicates whether rebate pressure is increasing.
- Trade spending: promotional increases that coincide with sales but reduce gross margin.
When does TRI-LUMA face generic entry risks and how would it impact revenues?
Generic risk for TRI-LUMA depends on:
- expiration and exclusivity of its branded formulation and any method-of-use protections,
- Orange Book listed patents and their enforceability,
- and whether substitutable formulations reach FDA approval.
Orange Book and exclusivity framework (commercial relevance)
For branded dermatology drugs, the commercial “cliff” typically comes from:
- patent expirations that remove legal barriers to ANDA entry,
- loss of exclusivity that allows marketing authorization or substitution,
- and settlements that delay launch dates.
Generic entry scenarios
- Early generic entry (unfavorable): rapid price compression and share loss.
- Delayed entry (moderately favorable): slower erosion with branded defense via contracts.
- Niche survival: if patent coverage blocks key formulations or dosing forms, the brand can keep a premium niche for longer.
What is the Orange Book status of TRI-LUMA and which patents are most commercially relevant?
The commercial impact of TRI-LUMA’s patent estate is determined by:
- whether patents cover formulation (the exact triple-combination composition),
- method-of-use claims (regimen specifics),
- and device/manufacturing method claims if any affect generic enablement.
Most commercially relevant patent categories for TRI-LUMA
- Composition of matter for the combined actives and concentration ratios
- Formulation patents covering stability, solubilization, or vehicle system
- Method-of-use claims tied to dosing schedule or photoprotection regimen
- Manufacturing process claims that increase generic development barriers
(A complete patent-by-patent list, expiration dates, and Paragraph IV history requires the Orange Book patent record and litigation docket details, which are not provided in the input.)
What TRI-LUMA patent litigation affects expected launch timing and settlement outcomes?
Timing of potential generic erosion is shaped by:
- any ANDA litigation under Hatch-Waxman,
- settlement agreements that define “carve-outs” and launch dates,
- and court rulings on patent validity or non-infringement.
Commercially decisive litigation elements
- whether litigation stays the ANDA effective date,
- whether settlement provides delayed launch but allows “design-around” products,
- whether injunctions or appeals shift enforcement risk.
(Specific cases, parties, and dates require litigation docket and press release sources not included in the provided material.)
How does TRI-LUMA compare with other pigmentation therapies on profitability and risk profile?
Compared with broad-market dermatology drugs, TRI-LUMA’s risk and return profile typically has:
- higher gross margin potential as a premium triple combination,
- lower volume scale than inflammatory derm brands,
- and higher vulnerability to targeted generic substitution if legal barriers fall.
Relative strengths
- Specialist-driven demand in melasma/exogenous ochronosis
- Treatment adherence and multi-active regimen can support switching costs
- If branded access stays preferred, brand retains pricing power longer
Relative weaknesses
- crowded therapeutic alternative set
- regimen complexity can reduce patient stickiness
- generic entry, if feasible, can compress prices quickly
What does TRI-LUMA’s financial trajectory imply for licensing, investment, and R&D decisions?
The most actionable lens for TRI-LUMA is whether its revenue base is defendable against:
- formulary loss,
- net-to-gross deterioration,
- and generic substitution.
Licensing and in-licensing implications
- If the brand still has meaningful patent/legal runway, licensing can justify commitments tied to extension and lifecycle management.
- If patents are near expiry, investment priority shifts toward next-generation formulations, line extensions, or adjacent indications.
R&D decision implications
- R&D efforts are best aligned to differentiation that improves coverage outcomes (e.g., simplified regimen, tolerability, or clearer adherence benefits).
- Manufacturing or stability improvements can matter if they allow contract advantages or reduced returns.
How does TRI-LUMA handle supply, manufacturing scale, and disruption risk?
Topical specialty drugs face:
- batch-to-batch uniformity constraints,
- stability shelf-life management,
- and packaging and distribution constraints (especially if demand spikes due to seasonal drivers).
Operational risk points
- quality incidents can trigger short-term shortages, raising interim sales but harming long-term trust
- channel restocking after shortages can distort growth metrics
(Specific TRI-LUMA manufacturing events and disruption history are not included in the input.)
Key Takeaways
- TRI-LUMA’s market is driven by specialist-treated pigment disorders with chronic recurrence and adherence-sensitive regimens.
- Sales trajectory is typically shaped more by payer access, rebate intensity, and channel inventory cycles than by steady prescription demand alone.
- The biggest revenue downside lever is generic substitution if Orange Book-listed protections are overcome; patent categories most sensitive to commercial outcome are formulation and method-of-use.
- Competitive substitution risks come from a mix of other topicals plus procedural therapies that can displace long-term topical reliance.
FAQs
- What determines TRI-LUMA patient access under Medicare Part D and commercial formularies?
- How do prior authorization edits and quantity limits impact TRI-LUMA prescription fill rates?
- Which patent claim types are most likely to block generic entry for TRI-LUMA?
- What market signals indicate weakening TRI-LUMA net sales due to contract and rebate pressure?
- How do seasonal patterns in melasma treatment demand typically affect TRI-LUMA quarterly performance?
References
No sources were provided in the prompt.