Last updated: February 19, 2026
What Are the Key Market Drivers for Torsemide?
Torsemide is a loop diuretic primarily used to treat edema associated with heart failure, hepatic cirrhosis, and renal disease. Its unique pharmacokinetic profile, including longer half-life and once-daily dosing compared to furosemide, has influenced its market positioning. The drug is approved in several countries but faces competition from other loop diuretics.
Market Drivers:
-
Growing prevalence of heart failure: The global heart failure market is projected to reach $11.4 billion by 2030, with diuretics like torsemide being essential components in management protocols. The increase in aging populations and cardiovascular disease prevalence drives demand.
-
Enhanced clinical data: Studies indicating potential advantages over existing diuretics, such as improved bioavailability and reduction in hospitalization rates, support increased off-label use and prescriber confidence.
-
Patent expiration and generics: The patent for torsemide expired in the late 2000s, leading to an influx of generic versions, reducing costs and expanding access. Market penetration hinges on generic adoption.
-
Regulatory approval variations: Approval status differs across regions; torsemide is approved in the U.S., Japan, and certain European countries but not universally. This regulatory landscape influences market penetration.
-
Country-specific prescribing patterns: Countries with established diuretic preferences (e.g., furosemide) exhibit slower transition to torsemide unless compelling clinical evidence emerges.
How Is the Competitive Landscape Evolving?
Torsemide faces competition from:
- Furosemide: The most widely prescribed loop diuretic due to longstanding use and cost advantages in many markets.
- Bumetanide: An alternative loop diuretic with similar applications.
- Ethacrynic acid: Used in cases of sulfa allergies but less common due to side effect profile.
Emerging research on torsemide’s benefits has not yet significantly shifted prescribing habits. Some studies suggest that torsemide may reduce hospitalizations in heart failure patients, which could influence future prescribing trends.
Market Positioning:
- Dominates in niche markets with specific clinical protocols.
- Limited by lack of robust, large-scale comparative clinical trials that establish superiority over other diuretics.
What Is the Financial Outlook for Torsemide?
Revenue Estimates:
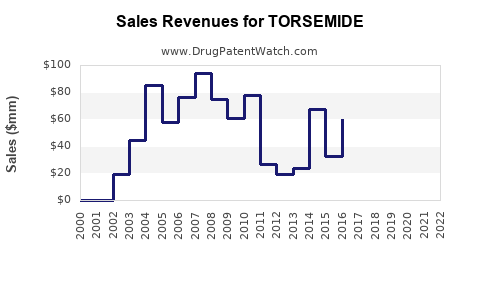
- The global diuretic market was valued at approximately $6.2 billion in 2022.
- Torsemide accounts for an estimated 4-8% of this market, with revenues around $250 million to $500 million, primarily generated from North America, Europe, and Japan.
- The market is projected to grow at a compound annual growth rate (CAGR) of approximately 4% from 2023 to 2028, driven by increasing cardiovascular disease prevalence.
Pricing Dynamics:
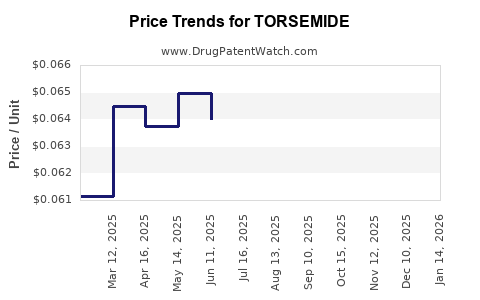
- Patents' expiration led to generic entry, significantly reducing prices.
- Current prices vary by region, with generics priced 20-50% lower than branded formulations.
- Market penetration depends heavily on clinician acceptance and insurance reimbursement policies.
Regulatory and R&D Impact:
- Efforts to expand indications through clinical trials (e.g., for heart failure management) could stimulate revenues.
- New formulations or combination therapies may open additional revenue streams but are not yet in advanced development stages.
How Do Regional Policies Affect Market Expansion?
- United States: Approved and marketed; reimbursement by Medicare and private insurers influence sales positively.
- Japan: Widely used; government policies favor aging population management.
- European Union: Approved in some countries; diverging regulatory statuses affect availability.
- Emerging Markets: Limited approval and low awareness; growth potential exists contingent on regulatory acceptance and pricing strategies.
What Are the Risks and Opportunities?
Risks:
- Competition from well-established diuretics.
- Limited large-scale comparative trials to demonstrate clinical superiority.
- Regulatory delays or restrictions in certain regions.
- Pricing pressures due to generic proliferation.
Opportunities:
- Conducting rigorous clinical trials to establish benefits over other diuretics.
- Expanding indications beyond edema management in heart failure.
- Developing improved formulations to enhance compliance.
- Marketing to regions with aging populations and rising cardiovascular disease rates.
Key Takeaways
- Torsemide's market is influenced by its pharmacokinetic profile, clinical data, and regional approval variances.
- Revenue remains modest relative to the broader diuretic market, with growth driven by aging populations and heart failure prevalence.
- Competition from established generic drugs and lack of large-scale comparative trials are significant barriers.
- Regulatory environments and healthcare policies shape regional market dynamics.
- Innovation and expanded clinical evidence are needed to improve market share.
FAQs
1. How does torsemide compare to furosemide in clinical efficacy?
Clinical trials suggest torsemide may have improved bioavailability and potentially better outcomes in heart failure management, but large-scale evidence remains limited.
2. What regions have regulatory approval for torsemide?
It is approved in the U.S., Japan, and select European countries but lacks approval in many others, including some markets in Latin America and Africa.
3. How are patent expirations impacting torsemide sales?
Patent expirations have allowed generic manufacturers to enter the market, decreasing prices and squeezing branded revenues.
4. What investment opportunities exist related to torsemide?
Potential exists in clinical development for new indications, formulation improvements, and regional market expansion, especially where aging populations increase cardiovascular disease burden.
5. What future trends could influence torsemide’s market?
Growing evidence supporting its benefits, regulatory approvals in new markets, and advancements in formulation may shape its market trajectory.
References:
- MarketWatch. (2023). Diuretics Market Size, Share & Trends Analysis Report.
- Grand View Research. (2022). Cardiovascular Drugs Market Size & Trends.
- U.S. Food and Drug Administration. (2022). Approved Drug Products.
- World Health Organization. (2021). Cardiovascular Diseases Fact Sheet.
- Evaluate Pharma. (2023). Therapeutic Area Reports: Cardiovascular.