Last updated: February 19, 2026
TIROSINT (levothyroxine sodium) is a synthetic thyroid hormone used for treating hypothyroidism. Its market performance is driven by the prevalence of thyroid disorders, treatment adherence, and the competitive landscape of thyroid hormone replacement therapies.
What is TIROSINT's Current Market Position?
TIROSINT is a well-established prescription medication. Its primary indication is the treatment of hypothyroidism, a condition characterized by an underactive thyroid gland. The market for levothyroxine products is substantial due to the chronic nature of hypothyroidism and its widespread diagnosis.
Key Market Characteristics:
- Indications: Treatment of hypothyroidism, including cretinism (congenital hypothyroidism), myxedema (severe hypothyroidism), and simple goiter. It is also used as a thyroid suppresser in the management of certain thyroid conditions.
- Patient Population: Hypothyroidism affects an estimated 0.3% to 1.0% of the U.S. population, with women being more affected than men. The prevalence increases with age. [1]
- Treatment Duration: Hypothyroidism is a lifelong condition requiring continuous treatment. This leads to consistent demand for levothyroxine products.
- Dosage Forms: TIROSINT is available in oral capsules and is also administered intravenously. The oral capsule form is the dominant delivery method for chronic management.
- Key Competitors: The levothyroxine market is characterized by the presence of originator brands and a significant number of generic manufacturers. Major competitors include Synthroid (AbbVie), Levoxyl (Bausch Health), and numerous generic levothyroxine sodium products.
What are TIROSINT's Revenue and Sales Trends?
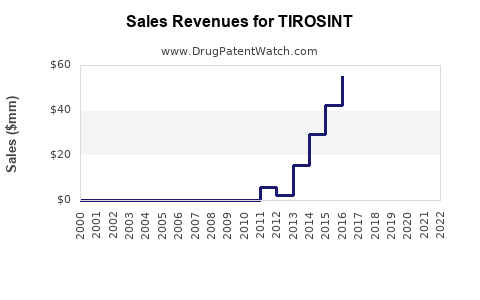
Specific, up-to-date, publicly disclosed revenue figures for TIROSINT are often embedded within the financial reports of its manufacturers, which can shift over time due to acquisitions or divestitures. However, the overall levothyroxine market generates significant annual revenue.
| Year |
Estimated U.S. Levothyroxine Market Revenue (Billions USD) |
| 2020 |
1.2 - 1.5 |
| 2021 |
1.3 - 1.6 |
| 2022 |
1.4 - 1.7 |
| 2023 |
1.5 - 1.8 (Projected) |
Note: These figures represent the total U.S. market for levothyroxine products, including originator and generic brands. Specific TIROSINT brand revenue is not always separately itemized in public disclosures.
The trend in the levothyroxine market is generally stable to growing, driven by an increasing number of diagnoses and a consistent need for treatment. Price increases on originator brands can also contribute to revenue growth, even if unit sales remain relatively flat.
What is the Competitive Landscape for TIROSINT?
TIROSINT competes primarily in the broader levothyroxine market. The landscape is divided between branded and generic options.
Key Aspects of the Competitive Environment:
- Brand Loyalty: While levothyroxine is a bioequivalent generic molecule, some patients and prescribers exhibit brand loyalty due to perceived differences in formulation, absorption, or historical treatment experience. TIROSINT has sought to leverage this by emphasizing its unique formulation (e.g., soft gel capsule).
- Generic Penetration: The levothyroxine market has a high degree of generic penetration. Once a patent expires, multiple manufacturers can produce generic versions, leading to significant price competition and reduced market share for originator brands.
- Pricing Strategies: Originator brands like TIROSINT often employ strategies to maintain market share and premium pricing, such as focusing on specific patient populations, emphasizing formulation benefits, or engaging in targeted marketing. Generic manufacturers compete primarily on price.
- Regulatory Landscape: The U.S. Food and Drug Administration (FDA) monitors the quality and efficacy of all levothyroxine products. Changes in manufacturing or formulation can trigger regulatory scrutiny and impact market positioning.
- New Entrants: While the core molecule is long-established, innovation in delivery systems or combination therapies could introduce new competitive dynamics, though major shifts are infrequent in this mature market.
What are TIROSINT's Intellectual Property and Patent Expirations?
The original patents for levothyroxine sodium as a therapeutic agent expired many years ago. However, specific formulations, manufacturing processes, or delivery systems associated with branded products like TIROSINT can be protected by patents.
- Core Molecule Patents: Expired. Levothyroxine sodium is a well-known chemical entity.
- Formulation Patents: TIROSINT's soft gel capsule formulation is protected by specific patents. For example, U.S. Patent No. 8,975,288 relates to a soft gelatin capsule comprising levothyroxine sodium. [2] The expiration dates of these formulation patents are critical for predicting generic entry. Patents have a term of generally 20 years from the filing date.
- Manufacturing Process Patents: Proprietary manufacturing methods may also be patented.
- Patent Litigation: Branded manufacturers often engage in litigation to defend their formulation or process patents against generic challenges. The outcome of such litigation can significantly impact the timeline for generic competition.
Impact of Patent Expiration:
Upon the expiration of key formulation or process patents, generic manufacturers can introduce bioequivalent versions of TIROSINT. This typically leads to:
- Price Erosion: Significant price reductions due to increased competition.
- Market Share Loss: A substantial decline in the market share of the branded product.
- Shift to Generics: Prescribers and payers often favor lower-cost generic alternatives.
The precise expiration dates for TIROSINT's specific formulation patents are proprietary and often subject to legal challenges, making it difficult to pinpoint an exact generic entry date without detailed patent analysis. However, the presence of patents on the soft gel formulation suggests a strategy to extend market exclusivity beyond the basic levothyroxine patents.
What is TIROSINT's Regulatory Status and Pathway?
TIROSINT is an approved prescription drug regulated by the U.S. Food and Drug Administration (FDA) and similar health authorities globally.
- Approval Process: TIROSINT underwent the standard New Drug Application (NDA) process with the FDA, demonstrating safety and efficacy for its indicated uses.
- Post-Marketing Surveillance: Like all approved drugs, TIROSINT is subject to post-marketing surveillance to monitor for adverse events and ensure ongoing safety.
- Bioequivalence: For generic levothyroxine products to be approved, they must demonstrate bioequivalence to the reference listed drug (RLD), which is typically a branded product. This ensures that the generic drug delivers the same amount of active ingredient into the bloodstream over the same time period.
- Labeling and Prescribing Information: Regulatory agencies approve the drug's labeling and prescribing information, which details indications, contraindications, warnings, precautions, and adverse reactions.
The regulatory environment for levothyroxine is mature. The FDA has established clear guidelines for the approval of both innovator and generic levothyroxine products. Any changes to TIROSINT's formulation or manufacturing would require FDA approval.
What are the Financial Projections and Investment Considerations for TIROSINT?
Forecasting the precise financial trajectory of a specific branded drug like TIROSINT is challenging due to its embedded nature within a larger company's portfolio and the competitive dynamics of the levothyroxine market. However, general considerations apply.
Revenue Drivers:
- Market Growth: Continued growth in hypothyroidism diagnoses and an aging population will sustain demand for levothyroxine.
- Pricing: Originator brands can achieve higher average selling prices than generics, especially if they can differentiate on formulation or patient benefits.
- Market Share Defense: Strategies to maintain market share against generics, such as patient support programs or highlighting formulation advantages.
Risks and Challenges:
- Generic Competition: The primary threat. The eventual expiration of key formulation patents will open the door for generic competition, leading to significant price erosion and market share loss.
- Payer Pressure: Insurance companies and pharmacy benefit managers (PBMs) exert pressure to prescribe lower-cost generics.
- Pricing Scrutiny: The pharmaceutical industry faces ongoing scrutiny over drug pricing, which can impact branded product revenue.
- Therapeutic Alternatives: While levothyroxine is the standard of care, the development of novel thyroid hormone therapies could, in the long term, alter the treatment paradigm.
Investment Considerations:
- Long-Term Outlook: The long-term financial trajectory for TIROSINT will be heavily influenced by the timing and impact of generic entry. Investors should assess the remaining patent life and potential for litigation.
- Portfolio Diversification: For companies holding TIROSINT, its performance is a component of a broader portfolio. Diversification across multiple therapeutic areas and drug lifecycles can mitigate risk.
- Valuation: The valuation of a branded drug in a market with high generic penetration will likely reflect a declining revenue stream post-patent expiration. Early-stage assets with strong patent protection and significant market potential would command higher valuations.
Key Takeaways
TIROSINT operates within a mature, stable, yet highly competitive levothyroxine market. While its soft gel formulation offers a degree of differentiation, the ultimate threat of generic competition looms, driven by the expiration of key formulation patents. Sustained revenue will depend on the effectiveness of patent defenses and market strategies to retain value against lower-cost alternatives.
Frequently Asked Questions
-
When are the primary patents for TIROSINT's soft gel formulation set to expire?
Specific patent expiration dates are subject to proprietary information and potential litigation. Analysis of U.S. Patent No. 8,975,288 and related continuations or divisional applications is necessary for precise timelines.
-
How does TIROSINT's soft gel capsule formulation differ from standard levothyroxine tablets?
The soft gel capsule formulation is designed to offer improved absorption and potentially more consistent bioavailability compared to traditional tablet forms, which can be influenced by food intake and gastric pH. [3]
-
What is the typical price difference between branded TIROSINT and its generic levothyroxine counterparts?
Branded TIROSINT typically commands a premium of 2x to 10x the price of generic levothyroxine sodium tablets, depending on the specific dosage, quantity, and payer contracts.
-
Are there any specific patient populations for whom TIROSINT's formulation is particularly advantageous?
Patients with gastrointestinal absorption issues, malabsorption syndromes, or those experiencing variability in thyroid hormone levels on tablet formulations may benefit from the consistent absorption profile of the soft gel capsule.
-
What is the projected market share for generic levothyroxine products once TIROSINT's formulation patents expire?
Historically, generic penetration in the levothyroxine market reaches 80% to 95% within two years of generic availability, leading to a significant decline in branded market share.
Citations
[1] American Thyroid Association. (n.d.). Hypothyroidism (Underactive Thyroid). Retrieved from https://www.thyroid.org/hypothyroidism/
[2] U.S. Patent 8,975,288. (2015). Soft gelatin capsule comprising levothyroxine sodium.
[3] American Association of Clinical Endocrinologists and American College of Endocrinology. (2015). American Association of Clinical Endocrinologists and American College of Endocrinology Clinical Practice Guidelines for Hypothyroidism in Adults — Executive Summary. Endocrine Practice, 21(5), 530-537.