Last updated: February 8, 2026
What Are the Market Dynamics for Terconazole?
Terconazole, an azole antifungal agent, is primarily used topically to treat vulvovaginal candidiasis. Its market is shaped by several factors:
Market Size and Segmentation:
- The global antifungal drugs market was valued at approximately $14 billion in 2021 and is projected to reach $23 billion by 2028, growing at a CAGR of 7.8% (source: Grand View Research).
- Terconazole accounts for a niche portion within vaginal antifungals, with dominant competitors including fluconazole, miconazole, and clotrimazole.
Key Drivers:
- Rising prevalence of vulvovaginal candidiasis, notably in women aged 20-40, driven by factors like antibiotic use, pregnancy, diabetes, and immunosuppression.
- Increasing awareness and diagnosis of fungal infections.
- Local biosimilar and generic versions expanding availability and reducing costs.
Constraints:
- Competition from oral antifungals with broader spectrum and easier administration—fluconazole prescriptions for candida infections run higher.
- Limited expandsion potential due to localized application, reducing scope for volume growth.
- Regulatory hurdles and patent expirations influence market entry timing and competitive landscape.
Regional Variations:
- North America and Europe dominate sales due to healthcare infrastructure, high diagnosis rates, and insurance coverage.
- Emerging markets in Asia-Pacific and Latin America experience growth through increased healthcare access and disease awareness.
What Is the Current Financial Trajectory of Terconazole?
Market Share and Revenue Estimates:
- Exact global sales figures for terconazole are limited; estimates suggest sales are in the low hundreds of millions USD annually.
- For example, in the U.S., branded Terazol (containing terconazole) sales are decreasing but remain significant within topical antifungal prescriptions.
Pricing Trends:
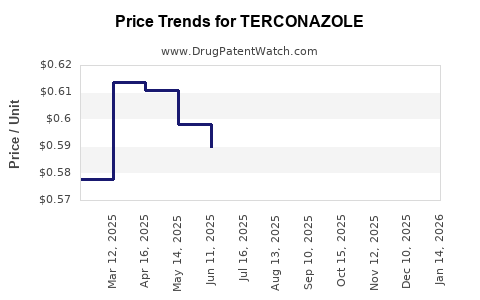
- Generic versions have driven prices down; average retail price per 0.4% vaginal cream (typical dosage) ranges from $20 to $50 per tube, varying by region.
- Brand-name Terazol historically commanded higher prices but has seen declines post-generic entry.
Pipeline and Patent Status:
- No recent patent protections are active on terconazole formulations, encouraging generic competition.
- No new formulations or delivery methods are under clinical development, limiting pipeline growth.
Profitability Outlook:
- Profit margins are constrained due to intense generic competition and price pressures.
- Manufacturers derive revenue mainly from volume rather than premium pricing.
Forecasts:
- Market analysts project flat to modest growth in terconazole sales, with potential declines in mature markets as oral alternatives capture more prescriptions.
- Emerging markets may offer opportunities for incremental growth, contingent on local healthcare policies.
How Do Competition and Regulatory Factors Influence the Market?
Competitive Landscape:
- Fluconazole, an oral agent, dominates with similar efficacy but benefits from oral dosing convenience.
- Clotrimazole and miconazole gels compete in the topical space with broader consumer recognition.
- Some generic manufacturers produce terconazole cream, exerting downward pressure on prices and margins.
Regulatory Environment:
- Regulatory bodies, such as the FDA and EMA, classify terconazole as a restricted use medication for specific indications, affecting prescribing practices.
- Healthcare guidelines favor simplicity and broad-spectrum agents, further marginalizing niche drugs like terconazole.
Patent Cessation and Generic Entry:
- Generic versions entered markets circa 2010-2015, eroding brand dominance.
- No new patents or exclusive rights are in effect, leading to pricing competition and sales plateau.
What Are the Key Takeaways?
- The market for terconazole is limited to niche topical antifungal applications for vulvovaginal candidiasis.
- Growth is moderate at best; the market faces substantial price competition from generics and oral alternatives.
- Sales are stable but declining in mature markets; emerging markets present incremental opportunities.
- Revenue primarily depends on volume, with profit margins suppressed due to intense competition.
- Future prospects for terconazole as a standalone product are limited absent new formulations or indications.
FAQs
1. Can terconazole expand into new indications?
No significant pipelines or clinical trials are ongoing for additional indications; current use is confined to vulvovaginal candidiasis.
2. How does the market share of terconazole compare to other antifungals?
It holds a small market share compared to oral agents like fluconazole but maintains niche relevance in topical formulations.
3. What are the primary challenges facing terconazole manufacturers?
Patent expiry, generic competition, price erosion, and competition from broader-spectrum or more convenient oral therapies.
4. Are there any emerging delivery methods for terconazole?
There are no significant developments in alternative delivery methods; current formulations include creams, suppositories, and foams.
5. What factors could alter the market trajectory of terconazole?
Introduction of new topical formulations, approval for additional indications, or changes in prescribing guidelines could influence future sales.
References
- Grand View Research. "Antifungal Drugs Market Size, Share & Trends Analysis Report." 2022.
- U.S. Food and Drug Administration. "Approved Drugs: Terazol."
- IQVIA. "Prescription Drug Trends and Market Data." 2022.
- European Medicines Agency. "Market authorization status for antifungal agents." 2023.