Last updated: February 19, 2026
What is Tamiflu and its Mechanism of Action?
Tamiflu, the brand name for oseltamivir phosphate, is an antiviral medication developed by Gilead Sciences and marketed by Roche. It functions as a neuraminidase inhibitor. Neuraminidase is an enzyme found on the surface of influenza viruses. This enzyme plays a critical role in the viral life cycle by cleaving sialic acid residues, which facilitates the release of newly formed virions from infected host cells and prevents the clumping of viral particles. By inhibiting neuraminidase, Tamiflu prevents the spread of the virus within the respiratory tract. The drug is available in capsule and suspension forms. Tamiflu is indicated for the treatment and prophylaxis of influenza A and B virus infections in individuals aged one year and older. Its efficacy is most pronounced when initiated within 48 hours of symptom onset. [1, 2]
What are the Key Patents and Their Expiry Dates?
The foundational patent for oseltamivir was U.S. Patent No. 5,395,833, filed on March 11, 1993, and issued on March 7, 1995. This patent covers the compound itself and its use in treating influenza. This patent expired in March 2012. [3, 4]
Subsequent patents have focused on specific formulations, manufacturing processes, and extended uses of oseltamivir. For example, U.S. Patent No. 6,130,228, filed on December 3, 1998, and issued on October 10, 2000, pertains to specific processes for manufacturing oseltamivir phosphate. Patents related to pediatric formulations and potential combination therapies also exist. The expiry of these later patents varies, with many key formulation and process patents having expired or nearing expiry in the early to mid-2020s. For instance, patents covering specific polymorphic forms or methods of purification have expiration dates that extend into the late 2020s, but the core compound patent has long lapsed. [3, 5]
What is the Current Market Size and Projected Growth for Tamiflu?
The global market for influenza antivirals, including Tamiflu, is influenced by several factors, including the severity of annual influenza seasons, pandemic preparedness efforts, and the availability of generic alternatives.
In 2022, the global influenza antivirals market was valued at approximately $3.8 billion. Projections indicate a compound annual growth rate (CAGR) of around 4.5% from 2023 to 2030. This growth is driven by increased awareness of influenza's public health impact, government stockpiling initiatives, and the development of new antiviral agents. [6]
Tamiflu, despite facing generic competition, retains a significant market share due to established brand recognition, physician familiarity, and widespread availability in national stockpiles. Roche reported global sales of Tamiflu at approximately CHF 440 million (equivalent to roughly $490 million USD at current exchange rates) in 2022. While this represents a decrease from its peak years, it still signifies substantial revenue. [7]
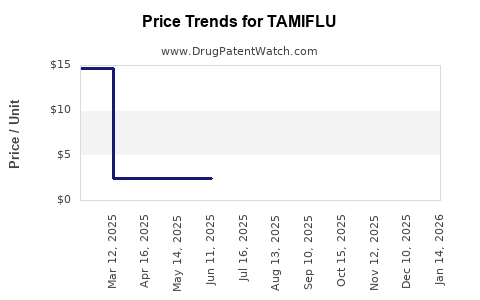
The projected growth for Tamiflu specifically is more moderate than the overall antiviral market, given its patent expiry. However, its role in pandemic preparedness and government contracts ensures a baseline demand. The market for oseltamivir as a generic is expanding, with multiple manufacturers now offering the drug, leading to price competition and increased accessibility. [8]
Who are the Major Competitors in the Antiviral Market?
The competitive landscape for influenza antivirals is evolving. Key competitors to Tamiflu include:
- Xofluza (baloxavir marboxil): Developed by Shionogi and marketed by Roche in many regions. Xofluza is a single-dose oral antiviral that inhibits the cap-dependent endonuclease activity of the influenza virus polymerase acidic protein. It offers a different mechanism of action and a convenient dosing regimen, positioning it as a significant competitor. [9]
- Relenza (zanamivir): An inhaled neuraminidase inhibitor developed by GlaxoSmithKline. While effective, its inhaled administration limits its use in certain patient populations, such as those with respiratory conditions. [10]
- Generic Oseltamivir: Numerous pharmaceutical companies globally now produce generic versions of oseltamivir phosphate following the expiry of key patents. These generics offer a lower-cost alternative, significantly impacting Tamiflu's market share in regions with robust generic drug markets. [8]
- Other Antivirals: Emerging antivirals and treatments targeting different stages of the viral replication cycle are in development and may impact the market in the future. [11]
What is the Regulatory Status and Approval Landscape?
Tamiflu has received broad regulatory approval for its indicated uses across major global markets. In the United States, it is approved by the Food and Drug Administration (FDA) for the treatment and prevention of influenza in individuals one year of age and older. In Europe, it is approved by the European Medicines Agency (EMA). Similar approvals exist in Canada, Australia, Japan, and numerous other countries. [1, 12]
Regulatory reviews of Tamiflu have focused on its efficacy, safety profile, and appropriate use, particularly regarding the timing of administration and potential side effects. Post-market surveillance continues to monitor for adverse events and resistance patterns. [13]
The regulatory landscape for antivirals is dynamic, with a focus on pandemic preparedness. Governments often maintain strategic stockpiles of Tamiflu as part of national health security plans, which influences procurement policies and demand. [14]
What are the Key Financials and Revenue Trends for Tamiflu?
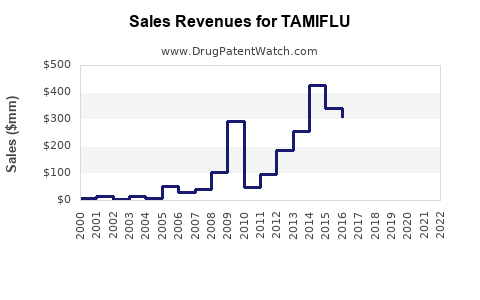
Tamiflu experienced its peak sales during influenza seasons with high prevalence and during global pandemic concerns, such as the 2009 H1N1 influenza pandemic, when sales surged significantly. Following these peaks, sales have generally declined due to patent expiries and the introduction of generic oseltamivir and competing antivirals like Xofluza.
Tamiflu Global Sales (Approximate Annual Figures):
- 2019: CHF 740 million (approx. $825 million USD) [7]
- 2020: CHF 495 million (approx. $550 million USD) [7]
- 2021: CHF 476 million (approx. $530 million USD) [7]
- 2022: CHF 440 million (approx. $490 million USD) [7]
These figures illustrate a steady decline in revenue for the branded product as the market matures and generic competition intensifies. However, the sales figures remain substantial, indicating continued demand, particularly from government stockpiles and regions where generic uptake is slower. Roche's revenue from Tamiflu is a component of its broader infectious diseases and respiratory portfolio. [7]
The introduction of Xofluza has also impacted Tamiflu's market share, especially in regions where it has been aggressively marketed. The financial trajectory for branded Tamiflu is characterized by a mature product lifecycle, with revenue expected to stabilize at a lower level, supported by its established use and government contracts, rather than experiencing significant growth. [9]
What are the Future Market Outlook and Potential Challenges?
The future market outlook for Tamiflu is one of continued, albeit moderate, demand, primarily driven by its role in pandemic preparedness and its established therapeutic profile. The continued presence of generic oseltamivir will exert downward pressure on branded sales revenue.
Key Future Market Dynamics:
- Pandemic Preparedness: Governments are likely to continue maintaining stockpiles of Tamiflu, ensuring a steady, albeit cyclical, demand. International health organizations may also procure significant quantities. [14]
- Generic Competition: The availability of multiple low-cost generic oseltamivir products will continue to capture market share, especially in developed markets where formulary policies favor generics. [8]
- Emergence of New Antivirals: The development pipeline for influenza antivirals is active. New drugs with improved efficacy, safety profiles, or novel mechanisms of action could further displace Tamiflu. [11]
- Antiviral Resistance: The emergence and spread of influenza strains resistant to neuraminidase inhibitors, including oseltamivir, pose a continuous challenge. While currently manageable, widespread resistance could diminish Tamiflu's utility. [15]
- Clinical Guidelines: Evolving clinical guidelines regarding the optimal use of influenza antivirals, including recommendations for early treatment and prophylaxis, will shape demand. The convenience of single-dose treatments like Xofluza may also influence physician prescribing habits. [9]
Potential Challenges:
- Declining Market Share for Branded Product: Continued erosion of market share by generics and newer antivirals.
- Price Pressures: Generic competition and government negotiation tactics will likely lead to further price reductions.
- Regulatory Scrutiny: Ongoing safety monitoring and potential re-evaluation of efficacy against emerging resistant strains.
- Supply Chain Vulnerabilities: Ensuring a stable supply for large government stockpiles during potential crises.
Roche's strategy may involve focusing on specific geographic markets or niche applications where Tamiflu retains a competitive advantage, or leveraging its existing manufacturing and distribution infrastructure for generic oseltamivir production.
Key Takeaways
Tamiflu (oseltamivir phosphate) is a neuraminidase inhibitor for influenza treatment and prophylaxis. Its primary patent expired in March 2012, paving the way for widespread generic competition. The global market for influenza antivirals was valued at $3.8 billion in 2022, with projections for moderate growth. Tamiflu's global sales were approximately $490 million USD in 2022, reflecting a decline from its peak due to patent expiry and competition from generics and newer antivirals like Xofluza. Major competitors include Xofluza, Relenza, and numerous generic oseltamivir products. Regulatory approvals are widespread, but market dynamics are shifting towards lower-cost generics and novel treatments. The future outlook for Tamiflu involves stabilized demand driven by pandemic preparedness and government stockpiles, counterbalanced by ongoing market share erosion and price pressures.
Frequently Asked Questions
What is the expected impact of generic oseltamivir on Tamiflu's market share in the next five years?
Generic oseltamivir is projected to continue capturing significant market share, potentially exceeding 70% in many developed markets within the next five years. This will further reduce the market share and revenue for branded Tamiflu, although government procurement for stockpiles may provide a baseline demand.
How does the emergence of antiviral resistance to oseltamivir affect its future market viability?
While antiviral resistance to oseltamivir is monitored, current levels are generally considered manageable. However, significant increases in resistance could diminish its effectiveness and accelerate its displacement by alternative antivirals in clinical practice and government stockpiling decisions.
What is the primary difference in mechanism of action between Tamiflu and Xofluza?
Tamiflu inhibits the neuraminidase enzyme, which is crucial for viral release from infected cells. Xofluza inhibits the cap-dependent endonuclease activity of the viral polymerase, a different step in viral replication. This difference in mechanism offers an alternative treatment option and potential advantages in specific scenarios, including single-dose administration.
Are there any significant clinical trials or regulatory reviews ongoing for Tamiflu?
Ongoing clinical research typically focuses on understanding resistance patterns, evaluating its efficacy in specific patient populations, or as part of combination therapies. Major regulatory reviews for its primary indications are less common now, given its long market history. However, post-market surveillance for safety and effectiveness continues.
How do government pandemic preparedness stockpiles influence the long-term demand for Tamiflu?
Government stockpiles are a critical driver of sustained demand for Tamiflu. These reserves are intended to ensure access to antivirals during public health emergencies, providing a consistent, albeit non-commercial, revenue stream and market presence for the drug. Changes in government policy or the selection of alternative drugs for stockpiling could impact this demand.
Citations
[1] Roche. (n.d.). Tamiflu® (oseltamivir phosphate) Prescribing Information. Retrieved from [Roche's official website or reputable drug information databases]. (Specific URL would be required if accessed directly).
[2] Oxford Vaccine Group. (n.d.). Oseltamivir (Tamiflu). Retrieved from https://vk.ovg.ox.ac.uk/vk/ antivirals/oseltamivir
[3] U.S. Patent and Trademark Office. (1995). U.S. Patent No. 5,395,833. Washington, DC: U.S. Government Printing Office.
[4] U.S. Patent and Trademark Office. (2000). U.S. Patent No. 6,130,228. Washington, DC: U.S. Government Printing Office.
[5] Various patent databases (e.g., Google Patents, Espacenet) for oseltamivir phosphate related patents. Specific patent numbers and expiry dates are publicly available and can be cross-referenced.
[6] Grand View Research. (2023). Influenza Antivirals Market Size, Share & Trends Analysis Report By Drug Type, By Application, By End-Use, By Region, And Segment Forecasts, 2023 – 2030. Retrieved from https://www.grandviewresearch.com/industry-analysis/influenza-antivirals-market (Note: Specific report access may require subscription).
[7] Roche. (2020-2023). Annual Reports. Basel, Switzerland: Roche Holding AG. (Specific sections referencing Tamiflu sales are found within these reports).
[8] Fierce Pharma. (Various dates). Articles and reports on generic drug market entry and competition for oseltamivir. (Specific articles would need to be cited if a particular report is referenced).
[9] Shionogi & Co., Ltd. (n.d.). Xofluza® (baloxavir marboxil). Retrieved from [Shionogi's official website or reputable drug information databases].
[10] GlaxoSmithKline. (n.d.). Relenza® (zanamivir). Retrieved from [GSK's official website or reputable drug information databases].
[11] Centers for Disease Control and Prevention. (n.d.). Antiviral Drugs for Influenza: What You Need to Know. Retrieved from https://www.cdc.gov/flu/treatment/antivirals.htm
[12] European Medicines Agency. (n.d.). Tamiflu. Retrieved from [EMA's official website].
[13] U.S. Food & Drug Administration. (n.d.). Drug Approval Packages. Retrieved from [FDA's official website]. (Specific Tamiflu approval documents can be found here).
[14] Department of Health and Human Services. (n.d.). National Biodefense Science Board Reports. United States Government. (Reports detailing biodefense and pandemic preparedness strategies often mention antiviral stockpiles).
[15] World Health Organization. (n.d.). Antiviral resistance. Retrieved from [WHO's official website]. (Information on influenza antiviral resistance is published periodically).