Last updated: February 19, 2026
SUTAB, a polyethylene glycol-3350-based bowel preparation, exhibits a focused market presence driven by its defined clinical utility and reimbursement landscape. Its financial trajectory is intrinsically linked to prescription volume, generic competition, and formulary access within the U.S. gastrointestinal preparation market.
What is SUTAB's Primary Market Segment?
SUTAB is positioned within the U.S. prescription bowel preparation market. This segment caters to individuals requiring intestinal cleansing prior to medical procedures such as colonoscopies. The total market for bowel preparations is characterized by a mix of prescription and over-the-counter (OTC) products, with SUTAB occupying a specific prescription niche. The market size for bowel preparations in the U.S. is estimated to be in the hundreds of millions of dollars annually, with prescription agents representing a significant portion. Factors influencing this segment include the prevalence of gastrointestinal conditions, screening recommendations for colorectal cancer, and physician prescribing habits. SUTAB's market share is contingent on its ability to secure and maintain favorable positioning against competing prescription agents.
What is the Regulatory Status of SUTAB?
SUTAB received U.S. Food and Drug Administration (FDA) approval in October 2020 [1]. It is classified as a prescription drug. Its approval was based on demonstrated efficacy and safety for bowel preparation. The regulatory pathway for bowel preparations involves rigorous review of clinical data, manufacturing processes, and labeling. Post-market surveillance is also a component of its regulatory oversight. Generic competition is a primary concern for branded prescription drugs once patents expire or are challenged. The Hatch-Waxman Act facilitates generic drug entry after patent protection. The longevity of SUTAB's market exclusivity is directly tied to its patent portfolio.
What are the Key Differentiating Features of SUTAB?
SUTAB's formulation is a key differentiator. It comprises two distinct drug products: SUTAB Tablet 1 (sodium picosulfate, magnesium oxide, citric acid) and SUTAB Tablet 2 (magnesium citrate) [2]. This tablet-based delivery system contrasts with liquid-based bowel preparations. The tablet format offers advantages in terms of palatability and ease of administration for some patients, potentially reducing compliance challenges associated with large-volume liquid intake. The efficacy of SUTAB is measured by the quality of bowel cleansing achieved, typically assessed using standardized scales such as the Boston Bowel Preparation Scale (BBPS). Its primary indication is for colon cleansing in adults scheduled for colonoscopy.
Who are SUTAB's Primary Competitors?
The competitive landscape for prescription bowel preparations includes established brands and emerging agents. Key competitors include:
- MoviPrep (PEG 3350, electrolytes, ascorbic acid and sodium ascorbate): A widely prescribed liquid preparation.
- Suprep (Sodium sulfate, potassium sulfate, magnesium sulfate): Another significant competitor in the prescription market.
- Plenvu (Macrogol/electrolyte solution with ascorbic acid and sweeteners): A newer entrant offering a lower-volume liquid option.
- Generic PEG-based solutions: Various generic versions of polyethylene glycol-based liquid preparations are available, offering cost advantages.
These competitors vary in their administration protocols, taste profiles, and electrolyte content, influencing physician and patient preferences. SUTAB competes on factors such as efficacy, tolerability, and ease of use.
What is the Reimbursement Landscape for SUTAB?
SUTAB is covered by most major U.S. commercial health insurance plans and Medicare Part D. Reimbursement rates and patient co-pays are determined by individual formularies, which are lists of prescription drugs covered by a health plan. Factors influencing formulary placement include clinical efficacy, safety profile, cost-effectiveness compared to alternatives, and established clinical guidelines. Prior authorization may be required by some insurers, adding a layer of administrative complexity. The financial success of SUTAB is contingent on its favorable placement on formularies and reasonable co-insurance/co-payment levels for patients, as high out-of-pocket costs can deter prescriptions.
What is the Pricing and Sales Performance of SUTAB?
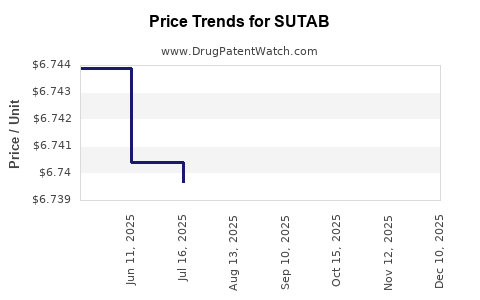
SUTAB's pricing is set by its manufacturer, Bausch Health Companies Inc. The list price for a SUTAB prescription is a primary factor, though actual sales revenue is influenced by negotiated discounts, rebates, and net price realization. Sales performance is tracked through prescription data, which includes new prescriptions (NRx) and total prescriptions (TRx). Market analytics firms report on these prescription trends. For instance, prescription volume data from IQVIA or Symphony Health Services would be the primary source for detailed sales performance analysis. Specific revenue figures for SUTAB are typically reported by Bausch Health in their quarterly and annual financial statements, often segmented within their Gastroenterology division. Without direct access to proprietary sales data, an analysis of publicly available financial reports from Bausch Health would reveal trends in their Gastroenterology segment, where SUTAB contributes.
What are the Key Patent Expirations for SUTAB?
The patent landscape for SUTAB is critical for its long-term market exclusivity. Patents protecting drug formulations, methods of use, and manufacturing processes grant market protection. Key patents for SUTAB would include those filed by the originators and potentially those filed by Bausch Health. Information on patent expiry dates can be found in the FDA's Orange Book (Approved Drug Products with Therapeutic Equivalence Evaluations) or through specialized patent databases. A typical patent for a new drug formulation can last up to 20 years from the filing date, subject to extensions. The expiration of these patents opens the door for generic manufacturers to seek FDA approval for their own versions of SUTAB, leading to price erosion and a potential decline in SUTAB's market share. Analyzing the remaining lifespan of core patents is essential for forecasting future revenue streams.
What is the Future Market Outlook for SUTAB?
The future market outlook for SUTAB is influenced by several factors. Continued growth in the colonoscopy market, driven by aging populations and colorectal cancer screening guidelines, will underpin demand for bowel preparations. However, increasing competition from both branded and generic alternatives, alongside potential formulary restrictions favoring lower-cost options, poses a challenge. Bausch Health's marketing and sales strategies, including physician education and patient access programs, will be crucial in maintaining SUTAB's market position. Innovation in bowel preparation technology, such as lower-volume formulations or improved palatability, could also shift market dynamics. The success of SUTAB will depend on its ability to demonstrate clinical and economic advantages over competing products in a competitive therapeutic area.
Key Takeaways
- SUTAB operates within the U.S. prescription bowel preparation market, primarily for colon cleansing prior to colonoscopies.
- The drug is a two-tablet system containing polyethylene glycol-3350, magnesium sulfate, sodium picosulfate, magnesium oxide, and citric acid.
- Key competitors include MoviPrep, Suprep, and Plenvu, alongside generic PEG-based preparations.
- Reimbursement is primarily through commercial health plans and Medicare Part D, with formulary placement and patient cost-sharing being critical factors.
- Patent expiry dates are the most significant factor influencing long-term market exclusivity and the potential for generic competition.
- Market growth drivers include an aging population and colorectal cancer screening recommendations, while competitive pressures and pricing dynamics will shape its financial trajectory.
Frequently Asked Questions
-
What is the typical patient co-pay for SUTAB?
Patient co-pays vary significantly based on individual insurance plans, deductible status, and formulary tier. It can range from a few dollars to over \$100 per prescription.
-
Are there any specific contraindications for SUTAB?
Yes, SUTAB is contraindicated in patients with gastrointestinal obstruction, gastric retention, ileus, toxic megacolon, gastric emptying disorders, or known hypersensitivity to any of its ingredients.
-
How does SUTAB's tablet format compare to liquid bowel preparations in terms of patient adherence?
Studies suggest that tablet-based preparations like SUTAB may improve patient adherence due to perceived ease of administration and improved palatability compared to large-volume liquid preparations. However, adherence is multifactorial and can still be influenced by dosage timing and potential side effects.
-
What is the predicted timeline for generic SUTAB entry into the market?
The timeline for generic entry depends on the specific patent litigation and expiration dates. Without direct knowledge of the detailed patent portfolio and any ongoing legal challenges, a precise prediction is not possible. However, generic entry typically occurs shortly after the expiration of key composition of matter or method of use patents.
-
Does SUTAB have any clinical advantages over its main competitors?
SUTAB's primary clinical advantage is its tablet-based delivery, which can enhance patient tolerability and adherence. Its efficacy in achieving adequate bowel preparation is comparable to other leading prescription agents when used as directed.
Citations
[1] U.S. Food and Drug Administration. (2020, October 7). FDA approves SUTAB (Sodium Picosulfate, Magnesium Oxide, and Citric Acid) Tablets for oral solution for colonoscopy preparation. [Press Release]. Retrieved from https://www.fda.gov/drugs/postmarket-drug-safety-information-for-patients-and-providers/fda-approves-sutab-sodium-picosulfate-magnesium-oxide-and-citric-acid-tablets-oral-solution-colonoscopy
[2] Bausch Health Companies Inc. (n.d.). SUTAB® (sodium picosulfate, magnesium oxide, and citric acid) Tablets. Retrieved from https://www.bauschhealth.com/our-products/patient-solutions/gastroenterology/sutab