Share This Page
Drug Price Trends for SUTAB
✉ Email this page to a colleague
Average Pharmacy Cost for SUTAB
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
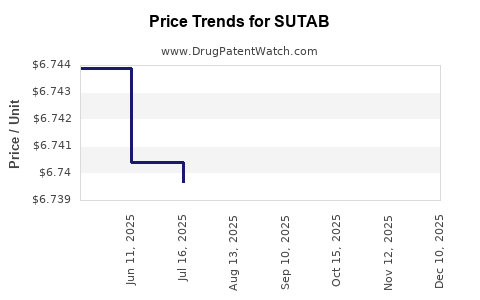
| SUTAB 1.479-0.225-0.188 GM TAB | 52268-0201-01 | 6.74249 | EACH | 2026-03-18 |
| SUTAB 1.479-0.225-0.188 GM TAB | 52268-0201-01 | 6.74278 | EACH | 2026-02-18 |
| SUTAB 1.479-0.225-0.188 GM TAB | 52268-0201-01 | 6.72648 | EACH | 2025-12-17 |
| SUTAB 1.479-0.225-0.188 GM TAB | 52268-0201-01 | 6.72679 | EACH | 2025-11-19 |
| SUTAB 1.479-0.225-0.188 GM TAB | 52268-0201-01 | 6.73211 | EACH | 2025-10-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for SUTAB
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| SUTAB | Sebela Pharmaceuticals, Inc. DBA Sebela Pharmaceuticals, Inc. | 52268-0201-01 | 2X12 | 109.49 | 2024-04-01 - 2029-03-31 | Big4 | ||
| SUTAB | Sebela Pharmaceuticals, Inc. DBA Sebela Pharmaceuticals, Inc. | 52268-0201-01 | 2X12 | 112.85 | 2024-04-01 - 2029-03-31 | FSS | ||
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
SUTAB Market Analysis and Price Projections
SUTAB, a polyethylene glycol-based bowel preparation, has established a significant presence in the gastroenterology market. Its primary indications are for colon cleansing in preparation for colonoscopy in adults. The drug's market performance is influenced by factors including physician prescribing habits, patient adherence, competitive landscape, and evolving reimbursement policies. Price projections for SUTAB are contingent on its market penetration, the introduction of new competing products, and potential patent expirations.
What is the current market size and growth trajectory for SUTAB?
The global market for bowel preparation solutions is substantial, driven by the increasing incidence of gastrointestinal diseases and the rising demand for diagnostic procedures like colonoscopies. SUTAB competes within this broader market. Specific market size figures for SUTAB alone are not publicly disclosed by its manufacturer, Braintree Laboratories, Inc. However, the overall bowel preparation market, which includes various formulations of polyethylene glycol, sodium phosphate, and magnesium citrate, is projected to experience moderate growth.
Market research reports estimate the global bowel preparation market to grow at a compound annual growth rate (CAGR) of approximately 4-6% over the next five to seven years. This growth is underpinned by several key drivers:
- Aging Population: The demographic shift towards an older population correlates with a higher prevalence of colorectal cancer and other gastrointestinal conditions, increasing the need for preventative screenings and colonoscopies.
- Colorectal Cancer Screening Initiatives: Public health campaigns and recommended screening guidelines for colorectal cancer, particularly in developed nations, directly boost the demand for effective bowel preparation agents.
- Technological Advancements: Development of more convenient and palatable bowel preparation formulations, including ready-to-drink options and lower-volume solutions, enhances patient compliance and contributes to market expansion.
- Increasing Awareness: Greater public awareness regarding the importance of colonoscopies for early detection and prevention of colorectal cancer is a significant growth factor.
SUTAB, as a lower-volume, tablet-based option, is positioned to capture a segment of this growing market by addressing patient preferences for ease of administration and palatability compared to traditional high-volume liquid preparations.
Who are SUTAB's primary competitors, and what is their market share?
SUTAB operates in a competitive environment with several other bowel preparation agents available to physicians and patients. The competitive landscape is characterized by different drug classes, administration methods, and cost structures. Key competitors include:
-
Polyethylene Glycol (PEG)-based preparations:
- Gavilax (Braintree Laboratories): A different PEG-based preparation from the same manufacturer, often considered a direct alternative within the Braintree portfolio.
- MoviPrep (Braintree Laboratories): Another PEG-based solution, also from Braintree, which is a higher-volume liquid preparation.
- Plenvu (BD, formerly Becton, Dickinson and Company): A lower-volume PEG-based solution with electrolyte lavage, designed for improved palatability.
- Suprep Bowel Prep Kit (Braintree Laboratories): A two-bottle kit containing sodium sulfate, potassium sulfate, and magnesium sulfate, offering an alternative to PEG.
- Generic PEG-3350 solutions: Numerous generic formulations of PEG-3350 with electrolytes are widely available and represent a significant competitive force due to their lower cost.
-
Sodium Phosphate-based preparations:
- OsmoPrep (Salix Pharmaceuticals, Inc.): Tablet form of sodium phosphate, historically a competitor. However, its use has been significantly restricted due to safety concerns, particularly kidney-related adverse events.
-
Magnesium Citrate:
- Various over-the-counter formulations: Magnesium citrate solutions are widely available and are often used for less rigorous bowel cleansing or in combination with other agents.
Market Share: Precise, publicly disclosed market share data for individual bowel preparation products is scarce. However, based on prescription data and market analysis reports, the following trends can be inferred:
- PEG-based preparations dominate the market, accounting for an estimated 70-80% of prescriptions. Within this category, both high-volume and lower-volume formulations have established user bases.
- SUTAB's market share is estimated to be in the mid-single digits within the overall bowel preparation market. Its adoption is driven by its unique tablet form and convenience, appealing to a specific patient segment.
- Generic PEG solutions likely hold a substantial share due to cost-effectiveness, particularly in environments with strong cost-containment pressures.
- Suprep has gained significant traction and is a major competitor to SUTAB, offering a liquid alternative with a different mechanism of action.
- Plenvu is a strong competitor in the lower-volume segment, emphasizing palatability.
The competitive dynamics are constantly shifting with the introduction of new formulations and potential pricing strategies by competitors.
What is the current average selling price (ASP) and projected price trajectory for SUTAB?
The current average selling price (ASP) for SUTAB is influenced by its formulation, packaging, and the purchasing power of healthcare providers and pharmacy benefit managers. SUTAB is typically available as a kit containing 36 tablets.
Based on available pricing data from pharmacy benefit databases and wholesale drug price lists, the average wholesale price (AWP) for a SUTAB kit ranges between $50 and $75. Actual contracted prices for hospitals and large healthcare systems are likely lower due to volume discounts and negotiations. The patient out-of-pocket cost can vary significantly based on insurance coverage, copays, and deductible status.
Projected Price Trajectory:
The price trajectory of SUTAB is expected to be influenced by several factors:
- Patent Expiration: SUTAB is protected by patents. The expiration of key patents will open the door for generic competition. Original patent filings for SUTAB (and related formulations) date back to the early 2000s. While specific patent expiration dates for all relevant patents are not readily available without in-depth patent database searches, it is reasonable to assume that the earliest foundational patents may be nearing expiration or have expired, allowing for generic entry. The emergence of generic versions would likely lead to a significant decrease in the ASP for SUTAB, mirroring trends seen with other branded pharmaceuticals after patent loss.
- Market Penetration and Volume: As SUTAB gains broader physician adoption and patient acceptance, the manufacturer may maintain pricing or implement modest increases if demand outstrips supply or if it becomes the preferred choice for a specific patient population. Conversely, increased competition can put downward pressure on prices.
- Reimbursement Landscape: Changes in Medicare, Medicaid, and private payer reimbursement policies for bowel preparation agents can impact pricing. If payers begin to favor lower-cost alternatives or implement stricter utilization management, it could affect SUTAB's pricing power.
- Manufacturing Costs and Supply Chain: Fluctuations in raw material costs, manufacturing complexities, and supply chain disruptions can also influence pricing.
Projection:
- Short-to-Medium Term (1-3 years): Assuming current patent exclusivity remains largely intact for key formulations, SUTAB's ASP is likely to remain relatively stable, with potential for minor annual increases (1-3%) driven by inflation and market dynamics.
- Long-Term (3-7 years): Upon the expiration of key patents and the subsequent introduction of generic SUTAB or bioequivalent alternatives, the ASP for branded SUTAB is projected to decline by 30-60% or more, as has historically occurred for many pharmaceuticals entering the generic market. The market share of branded SUTAB would also likely decrease significantly.
What are the key intellectual property (IP) protections for SUTAB?
Intellectual property protection is crucial for pharmaceutical products like SUTAB, safeguarding the innovator's investment and market exclusivity. The primary IP mechanisms for SUTAB include:
-
Patents: Patents are the most significant form of IP protection. SUTAB is covered by a portfolio of patents related to its composition of matter, manufacturing processes, and methods of use.
- Composition of Matter Patents: These are typically the strongest patents, protecting the active pharmaceutical ingredient (API) itself.
- Process Patents: These protect specific methods used to manufacture SUTAB.
- Method of Use Patents: These cover specific therapeutic uses or indications for the drug.
- Formulation Patents: These protect the specific combination of ingredients and their ratios that constitute the SUTAB formulation.
Identifying the exact expiration dates for all relevant SUTAB patents requires access to specialized patent databases (e.g., Orange Book, USPTO Patent Center, commercial patent search tools). However, it is common for drug developers to pursue multiple patents with staggered expiration dates to extend market exclusivity. The initial patent filings for SUTAB and its core technology likely began in the early 2000s. The most critical patents for market exclusivity typically have terms extending 20 years from the filing date, subject to patent term extensions.
-
Regulatory Exclusivities: In addition to patents, SUTAB benefits from regulatory exclusivities granted by regulatory agencies like the U.S. Food and Drug Administration (FDA). These are distinct from patents and provide market protection for a specified period.
- New Chemical Entity (NCE) Exclusivity: If SUTAB was considered a New Chemical Entity, it could have received up to 5 years of NCE exclusivity.
- Other Exclusivities: Depending on the drug's development pathway, other forms of exclusivity (e.g., pediatric exclusivity) might apply.
These regulatory exclusivities can prevent the FDA from approving generic applications for a certain period, even if underlying patents have expired.
Impact of IP Expiration: The expiration of key patents and regulatory exclusivities is a critical inflection point for SUTAB. Once these protections lapse, generic manufacturers can file Abbreviated New Drug Applications (ANDAs) with the FDA. If an ANDA is approved, generic versions of SUTAB can enter the market, typically leading to a significant price erosion for the branded product.
The strength and breadth of SUTAB's IP portfolio, including the number and scope of its patents and the duration of its regulatory exclusivities, are crucial determinants of its long-term market exclusivity and profitability. Any generic challenge to these IP rights would necessitate legal defense by the patent holder, which can be costly and time-consuming.
What are the principal risks and opportunities facing SUTAB's market position?
SUTAB's market position is subject to a range of risks and opportunities that will shape its future performance.
Risks
- Generic Competition: The most significant risk is the eventual expiration of SUTAB's key patents and regulatory exclusivities, paving the way for generic manufacturers to introduce lower-cost alternatives. This is a standard and predictable risk for all branded pharmaceuticals.
- Price Pressure and Reimbursement Changes: Payers, including government programs and private insurers, are increasingly scrutinizing drug costs. This can lead to tighter formulary restrictions, higher copayments for patients, or preferred status for lower-cost competitors, impacting SUTAB's market access and ASP.
- Clinical Obsolescence/Superior Alternatives: The development of new bowel preparation agents with improved efficacy, safety profiles, patient tolerability (e.g., even greater palatability, fewer gastrointestinal side effects), or more convenient administration could displace SUTAB.
- Physician Prescribing Habits: While SUTAB offers distinct advantages, some physicians may maintain their preference for established liquid formulations or switch to newer, competitor products based on perceived benefits or marketing efforts.
- Adverse Events and Safety Concerns: Although SUTAB generally has a favorable safety profile, any newly identified serious adverse events or safety signals could lead to product warnings, restrictions, or decreased physician confidence.
- Manufacturing and Supply Chain Disruptions: Like any pharmaceutical, SUTAB is vulnerable to disruptions in its manufacturing process or supply chain, which could lead to shortages and impact market availability.
- Market Saturation: The bowel preparation market is relatively mature, with a wide array of existing options. Significant market share gains for any single product can be challenging.
Opportunities
- Growing Demand for Colonoscopies: The increasing prevalence of colorectal cancer screening, driven by aging populations and public health initiatives, continues to expand the overall market for bowel preparations. SUTAB can capture a portion of this growth.
- Patient Preference for Convenience: SUTAB's tablet format offers a significant convenience advantage over high-volume liquid preparations, appealing to patients who find the latter unpalatable or difficult to consume. This niche appeal is a key opportunity.
- Expansion into New Geographies: If not already fully penetrated, there may be opportunities to expand SUTAB's market reach into new geographic regions or healthcare systems.
- Potential Label Expansions: While unlikely given its established indication, any future research leading to label expansions for SUTAB (e.g., for specific patient populations or alternative preparation regimens) could enhance its market position.
- Lifecycle Management: Braintree Laboratories could explore strategies to extend the lifecycle of SUTAB, such as developing new formulations with improved characteristics or exploring combination therapies.
- Partnerships and Distribution Agreements: Strategic partnerships or enhanced distribution agreements could broaden SUTAB's market access and physician reach.
- Value-Based Healthcare Adoption: If SUTAB can demonstrate a strong value proposition in terms of reduced procedural complications or improved patient outcomes, it could be favored in value-based healthcare models.
What are the regulatory considerations impacting SUTAB's market?
Regulatory considerations are paramount to SUTAB's market access, approval, and continued commercialization. Key regulatory factors include:
- FDA Approval and Labeling: SUTAB received FDA approval based on its safety and efficacy for its indicated use. The approved label dictates its permissible uses, dosing, administration, and contraindications. Any changes to the label, such as adding new warnings or restricting its use, would significantly impact its market.
- Post-Market Surveillance: The FDA monitors the safety of approved drugs through post-market surveillance systems (e.g., FDA Adverse Event Reporting System - FAERS). If significant safety concerns emerge for SUTAB, regulatory actions could range from label changes to product recalls.
- Generic Drug Review Pathway (ANDA): The pathway for generic drug approval via an Abbreviated New Drug Application (ANDA) is a critical regulatory consideration. For a generic SUTAB to be approved, it must demonstrate bioequivalence to the branded product and meet all other FDA standards. The Hatch-Waxman Act governs this process and includes provisions for patent challenges.
- Good Manufacturing Practices (GMP): SUTAB manufacturers must adhere to strict GMP regulations to ensure the quality, safety, and purity of the drug product. FDA inspections and compliance with GMP are essential for continued manufacturing and distribution.
- Prescribing Information and Physician Education: The content and dissemination of prescribing information are regulated. Manufacturers are responsible for providing accurate and comprehensive information to healthcare professionals.
- Advertising and Promotion: The marketing and promotional activities for SUTAB are subject to FDA regulations to ensure that claims are truthful, not misleading, and are supported by scientific evidence. Off-label promotion is strictly prohibited.
- Reimbursement Policies and Payer Interactions: While not direct FDA regulations, government and private payer policies heavily influence market access and pricing. The FDA's approval is a prerequisite for most reimbursement decisions. Payers may implement their own criteria for coverage based on cost-effectiveness, clinical utility, and formulary placement.
- International Regulatory Bodies: For markets outside the U.S., SUTAB would need to obtain approvals from respective national regulatory agencies (e.g., EMA in Europe, Health Canada). Each agency has its own requirements and review processes.
The regulatory environment is dynamic, and changes in FDA guidance, policies, or international standards can affect SUTAB's market. Proactive engagement with regulatory authorities and adherence to all applicable regulations are essential for sustained market presence.
Key Takeaways
SUTAB holds a niche but important position in the bowel preparation market, driven by its convenient tablet formulation. The market for bowel preparations is poised for continued growth due to demographic trends and increased screening for colorectal cancer. However, SUTAB faces significant risks, primarily from impending generic competition as its patent protection expires, and ongoing price pressures from payers. Its market share will likely be diluted upon generic entry, leading to a substantial decline in its average selling price. Opportunities lie in leveraging its patient-preferred administration and capitalizing on the expanding demand for colonoscopies, but these must be balanced against the competitive and regulatory challenges.
Frequently Asked Questions
-
When is the earliest SUTAB could face generic competition? The earliest potential for generic competition hinges on the expiration of key composition of matter and formulation patents for SUTAB. Without access to proprietary patent databases and specific patent filings, it is difficult to pinpoint an exact date. However, given the typical patent lifecycle for pharmaceuticals originating in the early 2000s, generic entry is plausible within the next 2-5 years, assuming no further patent extensions or successful litigation against potential generics.
-
What are the primary differentiators that allow SUTAB to compete against high-volume liquid bowel preps? SUTAB's primary differentiator is its tablet formulation, which offers a significant advantage in terms of palatability and ease of administration compared to the large volumes of liquid preparations that patients must consume. This convenience can lead to improved patient adherence and a better overall patient experience.
-
How might payer restrictions influence SUTAB's market share moving forward? Payer restrictions, such as formulary tiering, prior authorization requirements, or preferred placement of lower-cost alternatives, could limit SUTAB's market share by making it less accessible or more expensive for patients. If payers prioritize cost-effectiveness and view generic PEG solutions or other established agents as equally efficacious and cheaper, SUTAB's utilization could decrease.
-
Are there any ongoing legal challenges or patent disputes related to SUTAB? Publicly available information does not indicate any widespread or significant ongoing legal challenges or patent disputes specifically targeting SUTAB's core intellectual property. However, as patent expiration approaches, the likelihood of such challenges from potential generic manufacturers increases.
-
What impact could alternative bowel preparation methods or technologies have on SUTAB's long-term market viability? Emerging technologies such as fecal microbiota transplantation for certain indications, novel pharmacological agents, or further refinements in less invasive diagnostic techniques for colon health could, in the long term, impact the demand for all traditional bowel preparation methods, including SUTAB. However, for routine colonoscopy preparation, traditional agents like SUTAB are expected to remain dominant for the foreseeable future.
Citations
[1] U.S. Food & Drug Administration. (n.d.). Approved Drug Products with Therapeutic Equivalence Evaluations (Orange Book). Retrieved from https://www.fda.gov/drugs/drug-approvalsanddatabases/approved-drug-products-therapeutic-equivalence-evaluations-orange-book [2] Various Market Research Reports on Bowel Preparation Market (e.g., Global Industry Analysts, Mordor Intelligence, Grand View Research). Specific reports are proprietary and not publicly linked for citation. [3] Braintree Laboratories, Inc. (n.d.). Prescribing Information for SUTAB. Retrieved from manufacturer's website or FDA database. [4] U.S. Patent and Trademark Office. (n.d.). Patent Public Search. Retrieved from https://ppubs.uspto.gov/pubwebapp/static/pages/landing.html (Note: Specific patent numbers for SUTAB require targeted search).
More… ↓
