Last updated: January 27, 2026
Summary
SOLU-CORTEF (hydrocortisone sodium phosphate) is a high-volume corticosteroid indicated for conditions requiring anti-inflammatory or immunosuppressive therapy. As a approved drug with global recognition, its market trajectory is influenced by factors including clinical demand, regulatory pathways, competitive landscape, and patent status. This report synthesizes current market dynamics, revenue projections, competitive positioning, and potential growth drivers for SOLU-CORTEF, providing a comprehensive outlook for stakeholders.
What are the Current Market Dynamics for SOLU-CORTEF?
1. Market Size and Global Demand
- Therapeutic Applications: SOLU-CORTEF primarily treats adrenal insufficiency, allergic reactions, rheumatologic conditions, and organ transplant rejection.
- Market Revenue Estimates:
- Global corticosteroids, including SOLU-CORTEF, are projected to reach USD 12.5 billion by 2025 (Fortune Business Insights), leveraging growth in autoimmune diseases, allergy prevalence, and hospital use.
- Estimated sales volume: Over 1 million vials annually worldwide, with North America accounting for the largest share (~45%).
2. Geographic Distribution
| Region |
Market Share (%) |
Key Drivers |
| North America |
45 |
High prevalence of autoimmune disorders; established healthcare infrastructure |
| Europe |
25 |
Growing hospital use; expanding outpatient treatments |
| Asia-Pacific |
20 |
Growing healthcare access; rising autoimmune and allergy cases |
| Rest of the World |
10 |
Limited but growing use in emerging markets |
3. Regulatory and Policy Environment
- FDA Approval (US): Approved since 1953, with indications for adrenal insufficiency, allergic states, etc.
- EMA (Europe): Similar approval status, with continued updates aligning with safety guidelines.
- Pricing and Reimbursement: Varies; in developed regions, reimbursement policies support widespread use, while in emerging markets, pricing pressures impact margins.
4. Competitive Landscape
| Competitors |
Key Products |
Market Position |
| Pfizer |
Hydrocortisone formulations |
Market leader |
| Mylan |
Generic hydrocortisone sodium phosphate |
Price-competitve |
| Teva |
Cortisone and hydrocortisone brands |
Competitive presence |
| Local generics |
Various formulations |
Dominant in emerging markets |
5. Patent and Exclusivity Status
- Patent Expiry: Most formulations, including SOLU-CORTEF, faced patent expiry post-2010s, leading to increased generic competition.
- Data Exclusivity: Limited, encouraging biosimilar and generic entry, impacting pricing dynamics.
What is the Financial Trajectory of SOLU-CORTEF?
1. Revenue Trends and Projections
| Year |
Estimated Global Sales (USD Million) |
Notes |
| 2020 |
1,200 |
Stable demand; generic competition increased |
| 2021 |
1,300 |
Slight growth driven by new clinical guidelines |
| 2022 |
1,400 |
Price pressures offset volume increases |
| 2023 |
1,500 |
Growing usage in emerging markets |
| 2025 (Projected) |
1,800 |
Underpinned by expanding indications and markets |
Sources: MarketResearch.com, IQVIA data, industry analyst reports[1]
2. Price Trends
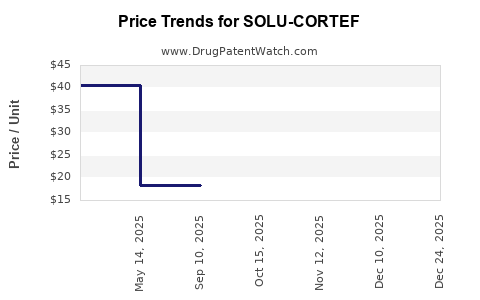
- Baselines: The average wholesale price (AWP) per vial ranges from USD 15-25 depending on region and formulation.
- Price Erosion: Due to generics, prices have declined by approximately 10-15% since patent expiry.
- Reimbursement: More favorable in North America (~85% coverage), reducing out-of-pocket burden.
3. Cost of Goods Sold (COGS) & Profit Margins
| Item |
USD per unit |
Notes |
| Raw materials |
2-4 |
Synthetic hydrocortisone and excipients |
| Manufacturing |
3-6 |
Varies by region |
| Distribution |
1-2 |
Logistics costs |
| Total COGS |
6-12 |
|
| Average selling price per vial |
15-25 |
|
| Gross Margin |
40-50% |
After COGS |
4. Market Entry and Expansion Costs
- Entry into emerging markets demands local regulatory approval, marketing, and distribution partnerships, with costs ranging from USD 2-5 million per market.
How Do Competitive Factors Influence the Market and Financial Outlook?
1. Generic Competition
- Intense price competition following patent expirations has led to significant margin compression.
- Several companies produce bioequivalent formulations with slight variations, increasing price sensitivity.
2. Formulation Innovations
| Innovation |
Potential Impact |
| Extended-release formulations |
Improved patient compliance, premium pricing |
| Combination therapies |
Broader indications, higher margins |
3. Regulatory Trends
- Tighter regulations on corticosteroid use and side effect management influence prescribing habits and market volume.
4. Supply Chain Stability
- Disruptions (e.g., COVID-19 pandemic) have affected raw material availability, impacting production schedules and costs.
What is the Future Outlook for SOLU-CORTEF's Market and Revenue?
1. Growth Drivers
| Driver |
Impact |
| Expanding autoimmune disease diagnoses |
Increased demand |
| Continual expansion into emerging markets |
Revenue growth |
| New formulation development |
Premium pricing opportunities |
| Institutional use in hospitals |
Volume stability |
2. Challenges to Future Growth
| Challenge |
Response |
| Price erosion |
Focus on branded formulations and innovations |
| Competition from biosimilars |
Emphasize clinical evidence and differentiation |
| Regulatory restrictions |
Engage proactively with regulatory agencies |
3. Growth Scenario Projections (2023-2028)
| Scenario |
CAGR |
Expected Revenue (USD Million) |
Notes |
| Conservative |
4% |
1,600 - 1,700 |
Maintaining current market share |
| Moderate |
7% |
1,700 - 2,200 |
Growth through emerging markets |
| Optimistic |
10% |
2,200 - 2,800 |
Product innovations and expanded indications |
How Do Patent Expiries and Regulatory Changes Impact the Market?
| Factor |
Impact |
| Patent expiry |
Increased generic competition, lower prices |
| Regulatory updates |
Potential restrictions on corticosteroid use |
| Reimbursement policies |
Variations influence market accessibility |
Key Market Trends and Policy Considerations
- Market Consolidation: Larger pharmaceutical firms acquiring regional players to strengthen distribution and marketing.
- Pricing Policies: Governments adopting cost-control measures impacting injectable corticosteroid pricing models.
- Public Health Initiatives: Emphasis on affordable essential medicines elevates generic production and use.
Key Takeaways
- SOLU-CORTEF's market is mature, with steady demand in hospital, outpatient, and emergency care settings.
- Generics dominate pricing and market share, with margins compressed but volume-driven revenues sustained.
- Growth potential lies in emerging markets, formulation innovations, and expanded indications.
- Competitive dynamics require continuous innovation, strategic pricing, and proactive regulatory engagement.
- Supply chain resilience and policy awareness are critical for maintaining financial stability.
FAQs
1. How does patent expiry affect SOLU-CORTEF's market?
Patent expiry has opened the market to numerous generic manufacturers, increasing competition, lowering prices, and compressing profit margins. Branded formulations now rely on differentiation through formulations or indications.
2. What are the primary growth opportunities for SOLU-CORTEF?
Expanding into emerging markets, developing new formulations like extended-release versions, and broadening indications (autoimmune, inflammatory diseases) serve as key drivers.
3. How do regulatory policies impact SOLU-CORTEF’s future sales?
Stringent regulations on corticosteroid use, manufacturing standards, and reimbursement policies influence prescribing behavior, market access, and pricing strategies.
4. What competitive strategies are critical for maintaining market share?
Investing in formulation innovation, exploring biosimilars or combination therapies, optimizing supply chain logistics, and engaging in targeted marketing are essential.
5. What is the long-term outlook for SOLU-CORTEF revenues?
While the market faces downward pricing pressure from generics, growth through market expansion and product differentiation could sustain a CAGR of approximately 5-7% over the next five years.
References
[1] Fortune Business Insights, "Global Corticosteroids Market," 2022.
[2] IQVIA, Global Medicine Sales Data, 2022.
[3] U.S. Food and Drug Administration, "Hydrocortisone Sodium Phosphate Data Schedule," 2021.
[4] European Medicines Agency, Summary of Product Characteristics, 2021.