Last updated: February 15, 2026
What are the market dynamics for SEREVENT?
SEREVENT (salmeterol inhalation powder) is a long-acting beta-agonist (LABA) primarily prescribed for asthma and chronic obstructive pulmonary disease (COPD). It is marketed by GlaxoSmithKline (GSK). The drug operates within a competitive landscape dominated by inhaled bronchodilators, including other LABAs and combination therapies.
Market size and growth
The global COPD market was valued at approximately USD 14.7 billion as of 2022 and is projected to grow at a compound annual growth rate (CAGR) of 4.2% through 2030. The asthma segment accounts for an estimated USD 7.5 billion in 2022, with a CAGR of 3.8%. SEREVENT captures a significant market share within these segments due to its established efficacy and regulatory approval.
Competitive landscape
Key competitors include:
- Formoterol (e.g., Foradil, Performist)
- Salmeterol (e.g., Serevent's class member)
- Combination inhalers (e.g., Symbicort, Advair) combining LABA with corticosteroids
The trend favors combination therapies for improved compliance, which pressures standalone LABAs like SEREVENT to innovate or partner.
Regulatory influences
SEREVENT's market is sensitive to guidelines issued by bodies such as GINA (Global Initiative for Asthma) and GOLD (Global Initiative for Chronic Obstructive Lung Disease). Both recommend LABA monotherapy only in conjunction with corticosteroids for sustained control, influencing SEREVENT's prescription volume.
Market adoption and restrictions
Prescription volumes for SEREVENT have plateaued due to:
- Enforced restrictions on LABA monotherapy because of safety concerns linked to increased asthma mortality.
- Shift towards combination inhalers.
- Preference for newer agents with improved delivery systems and lower side effect profiles.
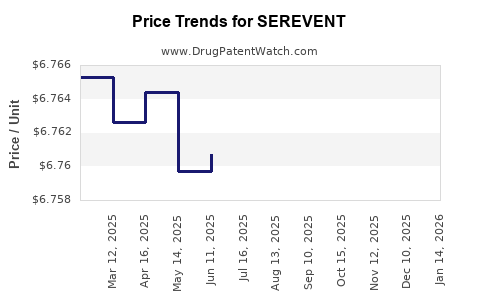
Pricing landscape
Pricing varies globally:
- In the U.S.: Approximately USD 250-350 per inhaler (30-day supply).
- In Europe: EUR 200-300.
- Prices are often subject to insurance coverage and formulary restrictions, affecting market penetration.
How is the financial trajectory for SEREVENT shaping?
Revenue performance
GSK reported the respiratory franchise, including SEREVENT, generating approximately GBP 2.4 billion in sales during 2022. SEREVENT's contribution has been relatively stable but has experienced slight declines in North America due to market saturation and competitive pressures.
Market share trends
In 2021, SEREVENT held around 20-25% of the inhaled LABA market in the U.S. and European markets. Market share has declined in the past two years in favor of combination therapies, with standalone LABAs losing ground to inhalers like Symbicort and Advair.
R&D investments and pipeline
GSK invests heavily in respiratory R&D:
- Developing fixed-dose combination inhalers.
- Exploring molecular modifications to improve drug delivery.
- Launching biosimilar programs for existing inhaled therapies.
No new formulations of SEREVENT are currently in late-stage development, reflecting a strategic shift toward integrated therapies.
Regulatory outlook and patent status
SEREVENT's primary patents in the U.S. expired in 2014. GSK maintains exclusivity through formulations, delivery devices, and additional patents extending patent life until approximately 2024-2025. Patent cliffs threaten future revenue unless new formulations or combinations are introduced.
Future revenue prospects
Forecasts suggest marginal growth or slight decline for SEREVENT's standalone sales through 2025, with upside potential if GSK successfully introduces new combination inhalers or gains regulatory approval for expanded indications.
Pricing and reimbursement outlook
Pricing pressures intensify with increasing adoption of generic and biosimilar alternatives. Insurance and government reimbursement policies heavily influence net revenue. Price negotiations and formulary placements will remain critical.
Key Drivers and Risks
| Drivers |
Risks |
| Growing prevalence of COPD and asthma |
Patent expiry leading to generic competition |
| Increasing adoption of combination therapies |
Regulatory restrictions reducing monotherapy prescriptions |
| GSK's R&D pipeline targeting combination inhalers |
Market saturation and stiff competition |
Key Takeaways
- SEREVENT faces a shrinking standalone monotherapy market due to safety concerns and shifting prescribing patterns.
- The drug’s revenue remains stable but is under pressure from enhanced competition and patent expirations.
- Future growth depends on GSK's ability to innovate with combination therapies and navigate evolving regulatory and reimbursement landscapes.
- Pricing varies significantly across jurisdictions, impacting overall profitability.
- Patent expirations and market saturation pose ongoing threats to long-term revenue stability.
FAQs
1. What is the primary use of SEREVENT?
SEREVENT treats asthma and COPD by providing long-term bronchodilation through inhalation.
2. How does SEREVENT compare to its competitors?
It is less favored compared to combination inhalers like Symbicort and Advair, which include corticosteroids, due to safety and efficacy considerations.
3. What is the impact of patent expiration on SEREVENT?
Patent expiry in 2014 opened the market to generics, reducing branded sales and increasing competition.
4. Are new formulations of SEREVENT in development?
No late-stage developments currently focus specifically on SEREVENT; efforts are toward combination products and delivery systems.
5. How do regulatory guidelines influence SEREVENT’s market?
Guidelines restricting LABA monotherapy decrease standalone SEREVENT prescriptions, favoring combination inhaler use.
Citations:
[1] Market data from GlobalData, 2022.
[2] GSK Annual Report 2022.
[3] GOLD Report 2023.
[4] GINA Report 2023.
[5] IQVIA Market Data, 2022.