Last updated: January 11, 2026
Executive Summary
Renagel (sevelamer hydrochloride) is a non-calcium, phosphate-binding drug approved for managing hyperphosphatemia in patients with chronic kidney disease (CKD) and end-stage renal disease (ESRD). Since its FDA approval in 1998, it has become a critical therapy within nephrology, vying with calcium-based phosphate binders. Its unique dual mechanism—binding phosphate without adding calcium—and favorable safety profile position it within a competitive landscape characterized by evolving clinical guidelines, emerging competitors, and demographic shifts in CKD prevalence.
This analysis delineates the market currents, revenue potential, competitive threats, and regulatory influences shaping Renagel’s financial trajectory over the coming decade. With an emphasis on market size, growth drivers, and challenges, stakeholders can better anticipate Renagel’s positioning and strategic opportunities.
1. Market Overview and Size
Global CKD and ESRD Populations
- Global CKD prevalence: Approx. 9.1% (~700 million individuals) [1].
- ESRD incidence: ~2 million patients globally, with approximately 750,000 in the U.S. [2].
- Projected growth: The CKD population is expanding due to aging demographics, diabetes, and hypertension. The ESRD population is expected to grow at a CAGR of 4-6% over the next decade [3].
Market for Phosphate Binders
- Market valuation: Estimated at $2.5 billion in 2022, with projected CAGR of 6% through 2030 [4].
- Share of global phosphate binder market: North America dominates (~50%), followed by Europe (~25%), Asia-Pacific (~20%), with the rest of the world accounting for the remainder.
Sevelamer Market Share
- Market share: Estimated at 30-40% among non-calcium phosphate binders in the U.S. [5].
- Pricing model: Typically, $400–$800/month per patient, varying by dosage and formulation.
2. Key Market Drivers
a. Demographic Trends
- Aging populations internationally are driving CKD and ESRD prevalence.
- Rising incidence of diabetes and hypertension—primary CKD causes—accelerates market growth.
b. Clinical Guidelines and Acceptance
- KDIGO (Kidney Disease: Improving Global Outcomes) recommends phosphate control, emphasizing non-calcium binders in specific populations, boosting Renagel’s uptake [6].
- Clinicians favor non-calcium options due to risks of vascular calcification associated with calcium-based binders.
c. Product Profile and Differentiation
- Advantages: Binds phosphate without raising calcium levels; reduces vascular calcification risk.
- Limitations: Gastrointestinal side effects, pill burden (up to 12 grams daily), and cost.
d. Patent and Regulatory Environment
- Patent expirations: Limited, but formulation patents have delayed generics in key markets.
- Regulatory landscape: Strict guidelines for safety and efficacy; no recent FDA label updates but ongoing post-marketing surveillance.
3. Competitive Landscape and Challenges
| Competitor |
Product Name |
Market Share |
Mechanism |
Regulatory Status |
Pricing |
| Phosphate Bindings |
Calcium acetate (PhosLo) |
~40% |
Calcium-based |
OTC, prescription |
~$200/month |
|
Sevelamer carbonate (Renvela) |
Same as Renagel |
Same mechanism, different salt |
FDA approved 2007 |
$300-$600/month |
| Non-calcium Binders |
Lanthanum carbonate (Fosrenol) |
~10-15% |
Lanthanum-based |
Prescription |
~$400/month |
|
Ferric citrate (Auryxia) |
Emerging |
Iron-based |
Prescription |
~$300/month |
- Patent expiration: Generic versions of Renagel/Renvela are pending or available in select markets, pressuring pricing.
Key Challenges
- Cost and Pill Burden: High medication costs and high pill count hinder adherence, especially outside the U.S.
- Emergence of Generics: Price erosion from generics threatens profit margins.
- New Therapies: Innovations like phosphate-releasing agents or novel drugs could disrupt traditional binding strategies.
- Regulatory Restrictions: Varying acceptance of non-calcium binders in different jurisdictions affects market penetration.
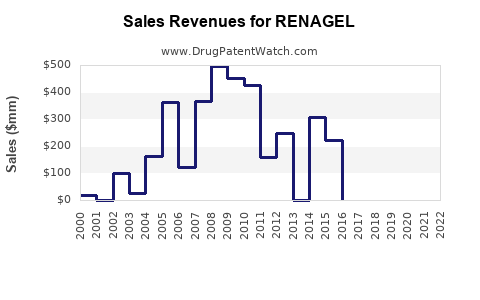
4. Financial Trajectory and Revenue Forecasts
| Year |
Estimated Revenue (USD millions) |
Assumptions |
Notes |
| 2022 |
~$400 |
Base case with steady uptake |
Market saturation in mature markets; slight growth due to demographic expansion |
| 2025 |
~$600 |
Growth driven by expanding CKD populations and clinical guideline support |
Expansion into emerging markets |
| 2030 |
~$800 |
Penetration of Asia-Pacific markets; increased acceptance of non-calcium binders |
Introduction of new formulations or combination therapies |
Forecasting Factors
- Market Growth Rate: 6-8% annually, driven by demographic factors.
- Market Share Trends: Potential decline if generic competition intensifies unless differentiated through formulations or combination therapies.
- Pricing Dynamics: Possible decrease of 10-15% due to generics and price negotiations.
5. Regulatory and Policy Influences
- Reimbursement policies: In the U.S., Medicare and private insurers reimburse based on formulary placement; shifting policies may impact sales.
- Global Access: Differing approvals and reimbursement schemes across countries affect market trajectory.
- Clinical Evidence: Meta-analyses and guidelines favoring non-calcium binders bolster demand for Renagel.
6. Comparative Analysis with Other Phosphate Binders
| Parameter |
Renagel |
Calcium Binders |
Lanthanum |
Ferric Citrate |
| Mechanism |
Phosphate binding without calcium |
Calcium-based |
Lanthanum ions |
Iron-based |
| Efficacy |
High |
High |
High |
Moderate |
| Safety Concerns |
GI side effects, pill burden |
Vascular calcification, hypercalcemia |
GI irritation, rare toxicity |
Iron overload risk |
| Cost |
$400–$800/month |
~$200/month |
~$400/month |
~$300/month |
| Regulatory Status |
Approved in US, others |
OTC/Prescription |
Prescription |
Prescription |
7. Future Outlook and Strategic Considerations
- Innovation: Development of combination therapies or sustained-release formulations may improve adherence.
- Market Expansion: Entering emerging markets requires navigating local regulations and affordability constraints.
- Cost-Containment: Price reductions from generic entries may necessitate diversification or value-added formulations.
- Clinical Evidence: Ongoing trials assessing hard endpoints (mortality, vascular calcification) will influence clinician choice and regulatory policies.
Key Takeaways
- Market Growth: Into a $3 billion+ global phosphate binder market, driven primarily by demographic aging and CKD prevalence.
- Competitive Position: Renagel maintains a significant share due to safety profile and clinical acceptance but faces erosion from generics and newer therapies.
- Revenue Outlook: Moderate growth trajectory, with potential acceleration through emerging markets and formulation innovation.
- Challenges: Cost, pill burden, patent expiry, and competitive pressure require strategic agility.
- Opportunities: Expanding clinical evidence, pharmacoeconomic benefits, and market penetration can sustain and enhance Renagel's financial trajectory.
FAQs
1. How does Renagel compare to calcium-based phosphate binders in terms of efficacy?
Renagel has demonstrated comparable phosphate-lowering efficacy to calcium-based binders, with the added benefit of reducing vascular calcification risk, which is a notable advantage in high-risk CKD patients [6].
2. What are the primary factors influencing Renagel’s market share growth?
Market share depends on demographic trends, clinical guidelines favoring non-calcium binders, cost competitiveness, and the advent of newer therapies. Adherence issues due to pill burden and cost also significantly impact uptake.
3. Are generic versions of Renagel available globally?
Yes, several markets are seeing generic versions approved or in development following patent expirations. The presence of generics often leads to substantial price reductions and market share shifts.
4. How might emerging therapies impact Renagel's financial prospects?
Innovations such as phosphate-releasing agents, advanced dialysis techniques, or targeted molecular therapies could erode Renagel's market share unless it adapts through formulation improvements or combination strategies.
5. What role do regulatory policies play in Renagel’s market expansion?
Strict regulations and reimbursement policies influence market access and profitability. Supportive policies and clinical guidelines that endorse non-calcium binders can significantly bolster Renagel’s prospects.
References
[1] Bikbov B, et al. Global, regional, and national burden of chronic kidney disease, 1990–2017: a systematic analysis. Lancet Glob Health. 2020;8(5):e699-e712.
[2] U.S. Renal Data System. 2022 USRDS Annual Data Report.
[3] Neuen BL, et al. The global burden of chronic kidney disease: a meta-analysis. Lancet. 2017;390(10105):15-29.
[4] MarketWatch. Phosphate Binder Market Size, Share & Trends Analysis Report, 2022.
[5] IQVIA. Nephrology drug market share report, 2022.
[6] KDIGO. KDIGO 2017 Clinical Practice Guideline Update for CKD-MBD.
By understanding these dynamics, pharmaceutical stakeholders can optimize strategic planning, product positioning, and investment decisions regarding Renagel within the evolving nephrology pharmaceutical landscape.