QINLOCK Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Qinlock, and what generic alternatives are available?
Qinlock is a drug marketed by Deciphera Pharms and is included in one NDA. There are forty-one patents protecting this drug.
This drug has one hundred and twenty-four patent family members in twenty-five countries.
The generic ingredient in QINLOCK is ripretinib. One supplier is listed for this compound. Additional details are available on the ripretinib profile page.
DrugPatentWatch® Generic Entry Outlook for Qinlock
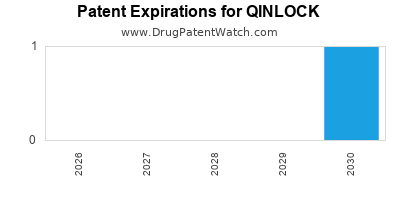
Qinlock was eligible for patent challenges on May 15, 2024.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be December 30, 2040. This may change due to patent challenges or generic licensing.
There have been three patent litigation cases involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
Indicators of Generic Entry
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for QINLOCK?
- What are the global sales for QINLOCK?
- What is Average Wholesale Price for QINLOCK?
Summary for QINLOCK
| International Patents: | 124 |
| US Patents: | 41 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 33 |
| Clinical Trials: | 1 |
| Patent Applications: | 344 |
| Drug Prices: | Drug price information for QINLOCK |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for QINLOCK |
| What excipients (inactive ingredients) are in QINLOCK? | QINLOCK excipients list |
| DailyMed Link: | QINLOCK at DailyMed |
DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for QINLOCK
Generic Entry Date for QINLOCK*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
TABLET;ORAL |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for QINLOCK
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Deciphera Pharmaceuticals LLC | Phase 1 |
Pharmacology for QINLOCK
US Patents and Regulatory Information for QINLOCK
QINLOCK is protected by forty-one US patents and one FDA Regulatory Exclusivity.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of QINLOCK is ⤷ Start Trial.
This potential generic entry date is based on patent 11,185,535.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Deciphera Pharms | QINLOCK | ripretinib | TABLET;ORAL | 213973-001 | May 15, 2020 | RX | Yes | Yes | 11,911,370 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Deciphera Pharms | QINLOCK | ripretinib | TABLET;ORAL | 213973-001 | May 15, 2020 | RX | Yes | Yes | 8,188,113 | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| Deciphera Pharms | QINLOCK | ripretinib | TABLET;ORAL | 213973-001 | May 15, 2020 | RX | Yes | Yes | 11,576,903 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Deciphera Pharms | QINLOCK | ripretinib | TABLET;ORAL | 213973-001 | May 15, 2020 | RX | Yes | Yes | 8,461,179 | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
EU/EMA Drug Approvals for QINLOCK
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Deciphera Pharmaceuticals (Netherlands) B.V. | Qinlock | ripretinib | EMEA/H/C/005614Qinlock is indicated for the treatment of adult patients with advanced gastrointestinal stromal tumour (GIST) who have received prior treatment with three or more kinase inhibitors, including imatinib. | Authorised | no | no | yes | 2021-11-18 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for QINLOCK
When does loss-of-exclusivity occur for QINLOCK?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Argentina
Patent: 2354
Estimated Expiration: ⤷ Start Trial
Patent: 2355
Estimated Expiration: ⤷ Start Trial
Australia
Patent: 20417282
Estimated Expiration: ⤷ Start Trial
Patent: 20419197
Estimated Expiration: ⤷ Start Trial
Patent: 23241368
Estimated Expiration: ⤷ Start Trial
Patent: 23248048
Estimated Expiration: ⤷ Start Trial
Patent: 24227597
Estimated Expiration: ⤷ Start Trial
Patent: 24259742
Estimated Expiration: ⤷ Start Trial
Patent: 25279824
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 2022013109
Estimated Expiration: ⤷ Start Trial
Patent: 2022013169
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 63051
Estimated Expiration: ⤷ Start Trial
Patent: 63053
Estimated Expiration: ⤷ Start Trial
China
Patent: 5135308
Estimated Expiration: ⤷ Start Trial
Patent: 5243681
Estimated Expiration: ⤷ Start Trial
Patent: 8948772
Estimated Expiration: ⤷ Start Trial
Patent: 8948773
Estimated Expiration: ⤷ Start Trial
Patent: 8948774
Estimated Expiration: ⤷ Start Trial
Patent: 9950433
Estimated Expiration: ⤷ Start Trial
Patent: 9970649
Estimated Expiration: ⤷ Start Trial
Patent: 0827525
Estimated Expiration: ⤷ Start Trial
Croatia
Patent: 0231699
Estimated Expiration: ⤷ Start Trial
Patent: 0241699
Estimated Expiration: ⤷ Start Trial
Denmark
Patent: 84778
Estimated Expiration: ⤷ Start Trial
Patent: 84779
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 84778
Estimated Expiration: ⤷ Start Trial
Patent: 84779
Estimated Expiration: ⤷ Start Trial
Patent: 27827
Estimated Expiration: ⤷ Start Trial
Patent: 01931
Estimated Expiration: ⤷ Start Trial
Finland
Patent: 84778
Estimated Expiration: ⤷ Start Trial
Patent: 84779
Estimated Expiration: ⤷ Start Trial
Hungary
Patent: 65493
Estimated Expiration: ⤷ Start Trial
Patent: 70151
Estimated Expiration: ⤷ Start Trial
Israel
Patent: 3864
Patent: תכשירים של 1-(4-bromo-5-(1-ethyl-7-(methylamino)-2-oxo-1,2-dihydro-1,6-naphthyridin-3-yl)-2-fluorophenyl)-3-phenylurea (Compositions of 1-(4-bromo-5-(1-ethyl-7-(methylamino)-2-oxo-1,2-dihydro-1,6-naphthyridin-3-yl)-2-fluorophenyl)-3-phenylurea)
Estimated Expiration: ⤷ Start Trial
Patent: 3866
Patent: הרכבים של מעכבי קינאז אמורפיים ושיטות לשימוש בהם (Amorphous kinase inhibitor formulations and methods of use thereof)
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 34416
Estimated Expiration: ⤷ Start Trial
Patent: 95672
Estimated Expiration: ⤷ Start Trial
Patent: 23509628
Patent: 1-(4-ブロモ-5-(1-エチル-7-(メチルアミノ)-2-オキソ-1,2-ジヒドロ-1,6-ナフチリジン-3-イル)-2-フルオロフェニル)-3-フェニル尿素の組成物
Estimated Expiration: ⤷ Start Trial
Patent: 23509629
Patent: 非晶質キナーゼ阻害剤の製剤およびその使用方法
Estimated Expiration: ⤷ Start Trial
Patent: 24097009
Patent: 1-(4-ブロモ-5-(1-エチル-7-(メチルアミノ)-2-オキソ-1,2-ジヒドロ-1,6-ナフチリジン-3-イル)-2-フルオロフェニル)-3-フェニル尿素の組成物 (COMPOSITIONS OF 1-(4-BROMO-5-(1-ETHYL-7-(METHYLAMINO)-2-OXO-1,2-DIHYDRO-1,6-NAPHTHYRIDIN-3-YL)-2-FLUOROPHENYL)-3-PHENYLUREA)
Estimated Expiration: ⤷ Start Trial
Patent: 25028954
Patent: 非晶質キナーゼ阻害剤の製剤およびその使用方法 (AMORPHOUS KINASE INHIBITOR FORMULATIONS AND METHODS OF USE THEREOF)
Estimated Expiration: ⤷ Start Trial
Lithuania
Patent: 84778
Estimated Expiration: ⤷ Start Trial
Patent: 84779
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 22008097
Patent: COMPOSICIONES DE 1-(4-BROMO-5-(1-ETIL-7-(METILAMINO)-2-OXO-1,2-DIH IDRO-1,6-NAFTIRIDIN-3-YL)-2-FLUOROFEIL)-3-FENILUREA. (COMPOSITIONS OF 1-(4-BROMO-5-(1-ETHYL-7-(METHYLAMINO)-2-OXO-1,2-D IHYDRO-1,6-NAPHTHYRIDIN-3-YL)-2-FLUOROPHENYL)-3-PHENYLUREA.)
Estimated Expiration: ⤷ Start Trial
Patent: 22008103
Patent: FORMULACIONES DE INHIBIDORES DE LA CINASA AMORFA Y METODOS DE ESTAS. (AMORPHOUS KINASE INHIBITOR FORMULATIONS AND METHODS OF USE THEREOF.)
Estimated Expiration: ⤷ Start Trial
New Zealand
Patent: 9199
Patent: Compositions of 1-(4-bromo-5-(1-ethyl-7-(methylamino)-2-oxo-1,2-dihydro-1,6-naphthyridin-3-yl)-2-fluorophenyl)-3-phenylurea
Estimated Expiration: ⤷ Start Trial
Poland
Patent: 84778
Estimated Expiration: ⤷ Start Trial
Patent: 84779
Estimated Expiration: ⤷ Start Trial
Portugal
Patent: 84778
Estimated Expiration: ⤷ Start Trial
Patent: 84779
Estimated Expiration: ⤷ Start Trial
San Marino
Patent: 02300467
Estimated Expiration: ⤷ Start Trial
Patent: 02400484
Estimated Expiration: ⤷ Start Trial
Serbia
Patent: 058
Patent: FORMULACIJE INHIBITORA AMORFNE KINAZE I POSTUPCI NJIHOVE PRIMENE (AMORPHOUS KINASE INHIBITOR FORMULATIONS AND METHODS OF USE THEREOF)
Estimated Expiration: ⤷ Start Trial
Patent: 335
Patent: KOMPOZICIJE 1-(4-BROMO-5-(1-ETIL-7-(METILAMINO)-2-OKSO-1,2-DIHIDRO-1,6-NAFTIRIDIN-3-IL)-2-FLUOROFENIL)-3-FENILUREA (COMPOSITIONS OF 1-(4-BROMO-5-(1-ETHYL-7-(METHYLAMINO)-2-OXO-1,2-DIHYDRO-1,6-NAPHTHYRIDIN-3-YL)-2-FLUOROPHENYL)-3-PHENYLUREA)
Estimated Expiration: ⤷ Start Trial
Slovenia
Patent: 84778
Estimated Expiration: ⤷ Start Trial
Patent: 84779
Estimated Expiration: ⤷ Start Trial
South Korea
Patent: 2800493
Estimated Expiration: ⤷ Start Trial
Patent: 220123057
Patent: 비정질 키나아제 억제제 제형 및 이의 사용 방법
Estimated Expiration: ⤷ Start Trial
Patent: 220123058
Patent: 1--3-페닐우레아의 조성물
Estimated Expiration: ⤷ Start Trial
Patent: 250057151
Patent: 1--3-페닐우레아의조성물 (1-4--5-1--7--2--12--16--3--2--3- COMPOSITIONS OF 1-4-BROMO-5-1-ETHYL-7-METHYLAMINO-2-OXO-12-DIHYDRO-16-NAPHTHYRIDIN-3-YL-2-FLUOROPHENYL-3-PHENYLUREA)
Estimated Expiration: ⤷ Start Trial
Patent: 250060322
Patent: 비정질 키나아제 억제제 제형 및 이의 사용 방법 (AMORPHOUS KINASE INHIBITOR FORMULATIONS AND METHODS OF USE THEREOF)
Estimated Expiration: ⤷ Start Trial
Spain
Patent: 66336
Estimated Expiration: ⤷ Start Trial
Patent: 91414
Estimated Expiration: ⤷ Start Trial
Taiwan
Patent: 2136257
Patent: Amorphous kinase inhibitor formulations and methods of use thereof
Estimated Expiration: ⤷ Start Trial
Patent: 2136258
Patent: Compositions of 1-(4-bromo-5-(1-ethyl-7-(methylamino)-2-oxo-1,2-dihydro-1,6-naphthyridin-3-yl)-2-fluoropheyl)-3-phenylurea
Estimated Expiration: ⤷ Start Trial
Patent: 99133
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering QINLOCK around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Serbia | 65058 | FORMULACIJE INHIBITORA AMORFNE KINAZE I POSTUPCI NJIHOVE PRIMENE (AMORPHOUS KINASE INHIBITOR FORMULATIONS AND METHODS OF USE THEREOF) | ⤷ Start Trial |
| South Korea | 101110530 | ⤷ Start Trial | |
| Japan | 2024097009 | ⤷ Start Trial | |
| South Korea | 20220123058 | 1--3-페닐우레아의 조성물 | ⤷ Start Trial |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Qinlock: Market Dynamics and Financial Trajectory
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.