Last updated: February 19, 2026
ProAir RespiClick, a dry powder inhaler (DPI) formulation of albuterol sulfate, is a key product in the respiratory drug market. Its market performance is influenced by patent exclusivity, generic competition, and evolving treatment guidelines for asthma and chronic obstructive pulmonary disease (COPD).
What is the Market Size and Growth Projection for Respiratory Inhalers?
The global respiratory inhaler market was valued at approximately $26.5 billion in 2022 and is projected to reach $37.6 billion by 2030, exhibiting a compound annual growth rate (CAGR) of 4.5% from 2023 to 2030. This growth is driven by the increasing prevalence of respiratory diseases, rising air pollution levels, and advancements in inhaler device technology. The DPI segment, in particular, is expected to experience sustained growth due to advantages such as dose accuracy and ease of use compared to metered-dose inhalers (MDIs).
What is ProAir RespiClick's Competitive Landscape?
ProAir RespiClick faces competition from several fronts:
- Other Albuterol Formulations: This includes both generic and branded MDIs and nebulized solutions. While ProAir RespiClick offers a DPI advantage, cost-effective generic MDIs remain a significant competitor.
- Other Bronchodilators: Short-acting beta-agonists (SABAs) like levalbuterol are alternatives. The choice often depends on physician preference, patient convenience, and cost-effectiveness.
- Combination Therapies: For patients with more severe or persistent asthma/COPD, long-acting bronchodilators (LABAs) or long-acting muscarinic antagonists (LAMAs) used in combination with corticosteroids are the standard of care. ProAir RespiClick, as a rescue inhaler, plays a complementary role rather than a primary treatment role in these severe cases.
- Newer Inhalation Technologies: Advancements in smart inhalers and breath-actuated devices are also influencing the market.
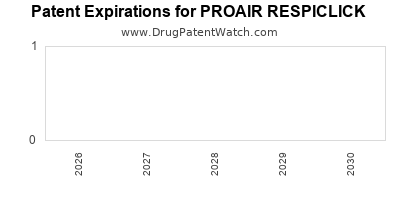
What are the Key Patents and Exclusivity Periods for ProAir RespiClick?
ProAir RespiClick's intellectual property protection is crucial to its market position. Key patents have covered the drug formulation and the unique RespiClick device.
| Patent Number |
Filing Date |
Expiration Date (Approximate) |
Description |
| US 7,708,005 |
2007 |
2028 |
Formulation patents related to albuterol sulfate DPI |
| US 8,505,471 |
2011 |
2027 |
Device patents for the RespiClick mechanism |
Note: Patent expiration dates can be subject to extensions due to patent term adjustments (PTA) or regulatory exclusivities. These dates represent general projections and should be verified through official patent databases.
Regulatory Exclusivity:
As a New Drug Application (NDA) product, ProAir RespiClick benefited from various regulatory exclusivities granted by the U.S. Food and Drug Administration (FDA) prior to the expiration of its core patents. These exclusivities, such as 5-year or 7-year exclusivities, delayed the approval of generic versions.
When did Generic Competition Emerge?
The emergence of generic competition for ProAir RespiClick is a critical factor in its financial trajectory. Teva Pharmaceuticals, for instance, launched a generic version of ProAir RespiClick in December 2020, following litigation that challenged its patents. The availability of generics significantly impacts market share and pricing power.
How is ProAir RespiClick Priced and What is its Reimbursement Status?
Pricing for prescription respiratory medications is complex, influenced by manufacturer list prices, negotiated rebates with payers, and generic entry.
- List Price: The manufacturer's list price for ProAir RespiClick has historically been in the range of $300-$500 for a 10.1 mg canister containing 200 actuations.
- Net Price: After rebates and discounts negotiated with pharmacy benefit managers (PBMs) and insurance companies, the net price realized by the manufacturer is substantially lower.
- Generic Pricing: Generic albuterol DPIs enter the market at significantly lower price points, typically 60-80% less than the branded product's net price.
- Reimbursement: ProAir RespiClick is generally covered by most commercial insurance plans and Medicare Part D. Coverage levels and co-pays vary significantly depending on the specific plan and formulary. The availability of generic alternatives often leads payers to steer patients towards generics through higher co-pays or prior authorization requirements for the branded product.
What are the Sales Performance Trends for ProAir RespiClick?
Prior to generic entry, ProAir RespiClick demonstrated strong sales growth. For example, in 2019, its net sales were reported to be over $600 million. Following the launch of generic alternatives in late 2020, sales have experienced a significant decline.
| Year |
Net Sales (USD Millions) |
Notes |
| 2019 |
600+ |
Pre-generic competition |
| 2020 |
580+ |
Impact of early generic entry towards year-end |
| 2021 |
< 300 |
Significant impact from generic competition |
| 2022 |
< 200 |
Continued erosion due to generic market share |
Data is approximate and based on publicly reported financial statements and industry analyses. Specific figures vary by reporting source.
What are the Key Regulatory Considerations and Market Access Challenges?
- FDA Approval Pathways: Approval of new inhaler devices and drug formulations requires rigorous clinical trials to demonstrate safety and efficacy.
- Generic Drug Approval (ANDA): Generic versions must demonstrate bioequivalence to the reference listed drug (RLD). The approval of a generic albuterol DPI for ProAir RespiClick was contingent on meeting these requirements.
- Market Access: Gaining and maintaining formulary placement with payers is essential. This involves demonstrating clinical utility and cost-effectiveness. Post-generic entry, market access for branded products becomes more challenging as payers favor lower-cost alternatives.
- Post-Market Surveillance: Ongoing monitoring for adverse events and product quality is required.
What is the Impact of Physician Prescribing Patterns and Patient Adherence?
Physician preference plays a significant role in the initial prescription of ProAir RespiClick. Factors influencing this include familiarity with the device, perceived patient benefits, and marketing efforts by the manufacturer. Patient adherence is also critical; the ease of use of the RespiClick device can contribute to better adherence compared to more complex inhalers. However, cost can become a barrier to adherence for patients with high co-pays, leading them to switch to generics or forgo treatment.
What are the Future Market Prospects for Albuterol Sulfate as a Rescue Inhaler?
Despite the rise of long-acting therapies, albuterol sulfate remains the cornerstone of rescue medication for asthma and COPD. The market for short-acting bronchodilators is expected to remain substantial, driven by the high prevalence of these conditions. However, the market share for branded albuterol products like ProAir RespiClick is likely to continue to decline as generic penetration increases. The focus for branded products will shift towards specific patient populations or niche indications where their unique device or formulation offers a demonstrable advantage, or through lifecycle management strategies.
Key Takeaways
ProAir RespiClick, a leading albuterol sulfate dry powder inhaler, has seen its market trajectory significantly altered by the expiration of key patents and the subsequent introduction of generic competitors. While the product benefited from robust sales and market exclusivity for a considerable period, its financial performance has been substantially impacted since late 2020. The competitive landscape is characterized by numerous albuterol formulations and alternative bronchodilators, with pricing and reimbursement strategies playing a critical role in market access. The increasing prevalence of respiratory diseases supports an ongoing demand for rescue inhalers, but the future market for branded products like ProAir RespiClick will likely be constrained by the widespread availability and cost-effectiveness of generics.
Frequently Asked Questions
-
What is the primary indication for ProAir RespiClick?
ProAir RespiClick is indicated for the treatment or prevention of bronchospasm in adults and children aged 4 years and older with reversible bronchospasm due to asthma and for the treatment of bronchospasm in adults and children 12 years of age and older with other forms of COPD.
-
How does the RespiClick device differ from a standard metered-dose inhaler (MDI)?
The RespiClick is a dry powder inhaler (DPI) that uses a breath-actuated mechanism. It delivers medication as a powder that is inhaled by the patient. Unlike many MDIs, it does not require the use of a propellant and is designed to deliver a consistent dose without the need for a spacer device.
-
What is the typical co-pay for a generic albuterol DPI compared to ProAir RespiClick?
A generic albuterol DPI typically has a significantly lower co-pay, often ranging from $0-$10. The co-pay for ProAir RespiClick can vary widely depending on the insurance plan, but it is generally higher, often in the $25-$75 range or more, especially without a preferred formulary tier.
-
Will there be any further patent extensions or exclusivities for ProAir RespiClick?
Based on publicly available patent information, the primary patents and regulatory exclusivities that provided market protection for ProAir RespiClick have expired or are nearing expiration, paving the way for generic competition. Significant future extensions are unlikely without new patent filings related to novel formulations or uses.
-
What factors are driving the growth in the overall respiratory inhaler market?
Key drivers include the increasing global incidence of asthma and COPD, rising rates of air pollution and allergens, aging populations more susceptible to respiratory illnesses, and ongoing innovation in inhaler technology for improved drug delivery and patient adherence.
Citations
[1] Global Respiratory Inhalers Market Size, Share & Trends Analysis Report by Product Type (Metered-dose Inhalers (MDIs), Dry Powder Inhalers (DPIs), Nebulizers, Smart Inhalers), By Disease Type (Asthma, COPD, Other Respiratory Diseases), By End-user (Hospitals, Clinics, Home Care), By Region, And Segment Forecasts, 2023 - 2030. (n.d.). Grand View Research. Retrieved from https://www.grandviewresearch.com/industry-analysis/respiratory-inhalers-market
[2] Teva Pharmaceuticals USA, Inc. v. Sandoz, Inc. (2020). United States District Court for the District of Delaware.
[3] Sunovion Pharmaceuticals Inc. (2019). Annual Report on Form 10-K. U.S. Securities and Exchange Commission.
[4] United States Patent and Trademark Office. (n.d.). Patent Search. Retrieved from USPTO.gov