Last updated: February 19, 2026
This report analyzes the market dynamics and financial trajectory of PRILOSEC OTC, a proton pump inhibitor (PPI) for the treatment of frequent heartburn. The analysis focuses on patent expirations, market competition, sales performance, and strategic shifts impacting its commercial viability.
What is the Current Market Position of PRILOSEC OTC?
PRILOSEC OTC (omeprazole) is an over-the-counter (OTC) medication approved for the treatment of frequent heartburn, defined as heartburn occurring two or more days per week [1]. It functions by reducing the amount of acid produced by the stomach. Its market position is characterized by a mature product life cycle, facing established competition and the ongoing challenge of brand differentiation in a crowded therapeutic space.
The drug was originally developed by AstraZeneca as a prescription-only medication (Prilosec) and subsequently transitioned to an OTC status after patent expiration. This transition allowed for wider accessibility and a broader consumer base, moving from a physician-directed therapy to a self-care option.
Key market attributes include:
- Therapeutic Category: Gastrointestinal (GI) - Acid Reducers.
- Active Ingredient: Omeprazole.
- Dosage Form: Delayed-release capsules and tablets.
- Indication: Treatment of frequent heartburn (two or more days per week).
- Target Consumer: Adults 18 years and older.
The market for heartburn relief is significant and highly competitive, encompassing a range of products from antacids and H2 blockers to other PPIs. PRILOSEC OTC operates within this segment, leveraging its established brand recognition and the well-understood efficacy of omeprazole.
What are the Key Patent Expirations and Their Impact?
The commercial trajectory of PRILOSEC OTC is intrinsically linked to patent expirations. The original patents for omeprazole, held by AstraZeneca, expired, paving the way for generic competition and the eventual launch of the OTC version.
- Original Prescription Patents (Prilosec): The compound patent for omeprazole expired in the United States in 2001 [2]. This event initiated the era of generic omeprazole availability in the prescription market.
- OTC Transition: Following the expiry of key patents and through regulatory pathways, AstraZeneca launched PRILOSEC OTC in 2003 [3]. This marked a significant strategic shift, converting a blockbuster prescription drug into a widely accessible OTC product.
- Generic OTC Competition: With omeprazole's active ingredient now off-patent and its availability as an OTC product established, generic manufacturers entered the market. This led to the introduction of numerous omeprazole OTC generics, directly competing with branded PRILOSEC OTC.
The impact of these expirations is multifaceted:
- Increased Competition: Patent expiry on the active ingredient fundamentally opened the door for generic manufacturers to produce omeprazole. This led to a price erosion as multiple players entered the market, both for prescription and later for OTC versions.
- Price Pressure: Branded PRILOSEC OTC faces direct price competition from lower-cost generic omeprazole OTC products. Consumers, particularly those seeking cost savings, often opt for generic alternatives.
- Market Share Dilution: While PRILOSEC OTC benefits from brand recognition, the availability of generics has inevitably diluted its overall market share in the omeprazole OTC segment.
The strategy of converting a successful prescription drug to an OTC product post-patent expiry is a common pharmaceutical business model. It allows the innovator to capture a new market segment and extend the product lifecycle, albeit with reduced pricing power due to generic competition. For PRILOSEC OTC, this strategy enabled continued revenue generation beyond the exclusivity of its prescription status.
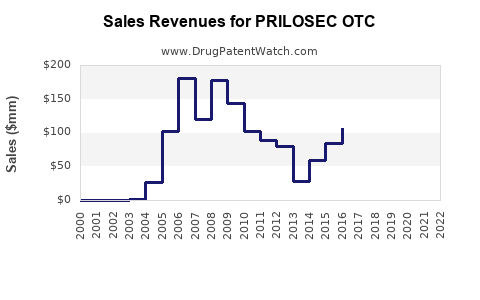
How Has PRILOSEC OTC Performed Financially?
Assessing the precise, real-time financial performance of a specific OTC product like PRILOSEC OTC can be challenging as sales data is often aggregated by parent companies or reported through market research firms that track consumer sales, rather than specific manufacturer revenues. However, its trajectory can be inferred from its market entry, the competitive landscape, and general trends in the OTC acid-reducer market.
- Launch and Early Growth: Upon its launch in 2003, PRILOSEC OTC likely experienced strong initial sales. This was driven by the established brand awareness of Prilosec, consumer familiarity with omeprazole's efficacy, and the expansion of the OTC acid-reducer market [3]. The ability for consumers to self-treat frequent heartburn without a prescription was a significant driver.
- Impact of Generic Entry: The introduction of generic omeprazole OTC products created significant downward pressure on pricing. While PRILOSEC OTC maintained its brand premium due to recognition and perceived quality, its market share within the omeprazole OTC category would have decreased as generics gained traction.
- Sustained Sales in a Mature Market: Despite generic competition, PRILOSEC OTC has maintained a presence in the market. This is attributable to:
- Brand Loyalty: Some consumers prefer branded products for perceived reliability and consistency.
- Marketing and Promotion: Ongoing marketing efforts by the brand owner (currently a subsidiary of Mitsubishi Tanabe Pharma Corporation, through its US subsidiary Taj Pharma, following acquisitions from P&G and later Merck/Bayer) help maintain consumer awareness and preference [4].
- Therapeutic Area Growth: The overall market for acid-reducer products, driven by lifestyle factors and an aging population, has generally shown resilience.
Estimated Market Size and Trends:
The OTC heartburn relief market is substantial. Global sales for OTC digestive health products, including acid reducers, are estimated to be in the billions of dollars annually. Within this, PPIs represent a significant sub-segment.
- Competitive Landscape: PRILOSEC OTC competes directly with other OTC PPIs such as Nexium 24HR (esomeprazole), Prevacid 24HR (lansoprazole), and generic omeprazole. It also competes with OTC H2 blockers (e.g., Zantac 360 - famotidine, Pepcid AC - famotidine) and antacids.
- Sales Data Considerations: Specific annual sales figures for PRILOSEC OTC are not publicly disclosed by its current marketer. However, market research reports from firms like Nielsen or IRI would track its retail performance against competitors. These reports generally indicate that while branded PPIs hold significant value share, the volume share is heavily influenced by generics.
The financial trajectory of PRILOSEC OTC is one of a mature product in a competitive landscape. Initial strong growth following its OTC launch has likely plateaued or seen incremental changes, with its financial performance now more dependent on market share maintenance against generics and overall market growth in the GI category.
What is the Competitive Landscape for PRILOSEC OTC?
The competitive landscape for PRILOSEC OTC is intense, characterized by numerous brands, both branded and generic, offering solutions for frequent heartburn. Competition stems from within the PPI class and from alternative acid-reducing drug categories.
1. Direct PPI Competitors (OTC):
- Nexium 24HR (esomeprazole): Another proton pump inhibitor, Nexium 24HR is a direct competitor. Esomeprazole is an isomer of omeprazole and is marketed for 24-hour protection from frequent heartburn. Its launch also represented a significant strategic move by AstraZeneca (and later acquired by Haleon, GSK's consumer health arm) to leverage its PPI franchise in the OTC space.
- Prevacid 24HR (lansoprazole): Lansoprazole is another PPI that has been transitioned to OTC status. It competes directly with PRILOSEC OTC and Nexium 24HR in offering a course of treatment for frequent heartburn.
- Generic Omeprazole OTC: A multitude of generic manufacturers offer omeprazole capsules and tablets in the OTC market. These products are typically priced significantly lower than branded PRILOSEC OTC, representing the most direct and price-sensitive competition.
2. H2 Blocker Competitors (OTC):
- Famotidine (e.g., Pepcid AC, Zantac 360): Histamine-2 (H2) blockers work by reducing stomach acid production, but through a different mechanism than PPIs. While generally considered less potent for severe or persistent heartburn than PPIs, they offer faster onset of action for immediate relief and are also widely available OTC. The re-introduction of Zantac with famotidine as the active ingredient (after the original ranitidine formulation was withdrawn) has intensified competition in this segment.
- Cimetidine: While less common in the current market due to perceived efficacy and side effect profiles compared to famotidine, cimetidine is also an H2 blocker that has been available OTC.
3. Antacids (OTC):
- Calcium Carbonate, Magnesium Hydroxide, Aluminum Hydroxide (e.g., Tums, Maalox, Mylanta): These products provide immediate, short-term relief by neutralizing existing stomach acid. They do not reduce acid production. Their rapid action and low cost make them a popular choice for occasional heartburn, representing a different but overlapping competitive segment.
4. Lifestyle and Dietary Factors:
While not direct product competitors, lifestyle modifications (diet, weight management, avoiding trigger foods) are often recommended and practiced by consumers, potentially reducing the overall need for medication.
Key Competitive Dynamics:
- Price: For many consumers, price is a significant factor. Generic omeprazole OTC and H2 blockers are generally more affordable than branded PPIs.
- Efficacy and Onset of Action: PPIs like PRILOSEC OTC are designed for a course of treatment (typically 14 days) and are highly effective at reducing acid production. H2 blockers offer quicker relief for some, while antacids provide immediate neutralization.
- Brand Trust and Recognition: PRILOSEC OTC benefits from the legacy of Prilosec and established brand recognition, which can foster trust and loyalty.
- Marketing and Distribution: Pharmaceutical companies invest heavily in marketing and ensuring broad availability of their OTC products in pharmacies, supermarkets, and online retailers.
The competitive landscape necessitates ongoing brand management and strategic marketing to maintain market share for PRILOSEC OTC against both direct generic rivals and alternative drug classes.
What is the Regulatory and Policy Environment Affecting PRILOSEC OTC?
The regulatory and policy environment significantly shapes the market for OTC drugs like PRILOSEC OTC. Key aspects include the Over-the-Counter Monograph system, FDA oversight, and marketing regulations.
- FDA Oversight and OTC Monograph System: PRILOSEC OTC, like other OTC drugs, falls under the purview of the U.S. Food and Drug Administration (FDA). It operates under an approved New Drug Application (NDA) rather than the OTC drug monograph system for certain categories, due to its original prescription status. However, the general regulatory framework for OTC drugs applies, ensuring safety, efficacy, and proper labeling.
- Labeling Requirements: Strict labeling requirements ensure consumers understand the indication, dosage, warnings, and contraindications. For PRILOSEC OTC, the indication for "frequent heartburn" is clearly defined.
- Good Manufacturing Practices (GMP): Manufacturers must adhere to GMP to ensure product quality and consistency.
- Post-Market Surveillance: The FDA monitors OTC drugs for safety issues after they are on the market. Any emerging safety concerns, as has happened with certain other heartburn medications (e.g., ranitidine contamination issues leading to recalls), could impact the entire class of drugs and necessitate label changes or market withdrawals.
- Marketing and Advertising: OTC drug advertising is regulated by the FDA and the Federal Trade Commission (FTC). Claims made in advertising must be truthful, not misleading, and substantiated by scientific evidence. This is particularly relevant for differentiating PRILOSEC OTC from competitors.
- Direct-to-Consumer Advertising (DTCA): DTCA for OTC drugs is common and plays a critical role in consumer awareness and purchasing decisions.
- Patent Law and Generic Entry: As discussed, patent law is central to the lifecycle of drugs. The expiry of patents for omeprazole enabled both the transition to OTC and the subsequent entry of generic versions, fundamentally altering the competitive and pricing dynamics.
- Pricing and Reimbursement (Indirect Impact): While PRILOSEC OTC is an out-of-pocket purchase for consumers, broader policy trends in healthcare costs and prescription drug pricing can indirectly influence consumer spending on OTC alternatives. However, direct government regulation of OTC drug prices is limited.
- International Regulations: For markets outside the U.S., PRILOSEC OTC and its competitors are subject to the specific regulatory frameworks of each country or region (e.g., European Medicines Agency in Europe). These can include different approval processes, labeling requirements, and classifications of prescription versus OTC status.
The regulatory environment ensures that PRILOSEC OTC is a safe and effective product for its indicated use. However, it also establishes the pathways for generic competition and marketing standards that all players must adhere to, thereby influencing market access and strategy.
What are the Future Outlook and Strategic Considerations for PRILOSEC OTC?
The future outlook for PRILOSEC OTC is characterized by sustained competition within a mature market. Strategic considerations will focus on leveraging its established brand equity while navigating pricing pressures and evolving consumer preferences.
1. Market Dynamics:
- Continued Generic Erosion: The presence of multiple generic omeprazole OTC products will continue to exert downward pressure on pricing and potentially volume for branded PRILOSEC OTC.
- Brand Differentiation: Strategies to differentiate PRILOSEC OTC will be crucial. This could involve emphasizing its long-standing heritage, proven efficacy, or specific benefits through targeted marketing campaigns.
- Competition from Other OTC Classes: H2 blockers and newer formulations or brands within the acid-reducer category will remain significant competitive forces.
- Evolving Consumer Habits: Consumer purchasing patterns, including a greater reliance on online channels and a continued search for value, will influence sales.
2. Strategic Considerations:
- Marketing and Brand Reinforcement: Sustained and effective marketing will be essential. This includes digital marketing, in-store promotions, and potentially leveraging endorsements or educational content to reinforce the brand's position. Focus may shift from broad awareness to reinforcing specific benefits or trust.
- Product Line Extensions/Reformulations: While less common for established OTC brands, exploring minor reformulations (e.g., different tablet forms, flavorings) or combination products, if supported by regulatory approval, could offer incremental differentiation. However, the core product's efficacy is well-established.
- Pricing Strategy: A carefully managed pricing strategy will be necessary to balance competitiveness against generics with the maintenance of a brand premium. This may involve promotional pricing, multi-pack offerings, or loyalty programs where feasible.
- Distribution Channel Optimization: Ensuring broad and convenient availability across traditional retail channels (pharmacies, supermarkets) and growing e-commerce platforms is paramount.
- Focus on "Frequent Heartburn" Segment: Continuing to target the specific indication of frequent heartburn, for which PPIs are clinically recommended for a course of treatment, will reinforce its therapeutic positioning against faster-acting but potentially less effective OTC alternatives for this specific need.
3. Potential Threats:
- Emerging Safety Concerns: Any significant safety issue identified with omeprazole or PPIs as a class could lead to regulatory action, impacting PRILOSEC OTC.
- Innovation in Alternative Treatments: Breakthroughs in treating GI disorders or novel non-pharmacological approaches could shift consumer behavior away from traditional medications.
- Aggressive Generic Pricing: Intense price wars from generic competitors could force further price reductions for branded PRILOSEC OTC, impacting profitability.
The future for PRILOSEC OTC lies in solidifying its position as a trusted, effective treatment for frequent heartburn within a competitive and price-sensitive OTC market. Its long history and brand recognition are assets that, when coupled with strategic marketing and distribution, can ensure continued market presence.
Key Takeaways
- PRILOSEC OTC, an OTC proton pump inhibitor for frequent heartburn, operates in a mature market following the patent expiration of its prescription predecessor.
- The expiration of omeprazole's active ingredient patents in 2001 enabled the launch of PRILOSEC OTC and subsequently led to significant generic competition in both prescription and OTC markets.
- Financial performance is characterized by sustained sales due to brand recognition but faces ongoing price erosion from generic omeprazole OTC products.
- The competitive landscape is intense, featuring direct PPI rivals (Nexium 24HR, Prevacid 24HR, generic omeprazole), H2 blockers (famotidine), and antacids.
- The drug is subject to FDA regulation regarding safety, efficacy, and labeling, with marketing governed by FTC and FDA standards.
- Future strategies will focus on brand reinforcement, optimized pricing, and broad distribution to maintain market share against persistent generic and alternative class competition.
Frequently Asked Questions
-
What is the primary mechanism of action for PRILOSEC OTC?
PRILOSEC OTC works by reducing the amount of acid produced by the stomach, acting as a proton pump inhibitor.
-
When did omeprazole become available over-the-counter in the U.S.?
Omeprazole became available over-the-counter as PRILOSEC OTC in the U.S. in 2003.
-
What is the key difference between PRILOSEC OTC and antacids?
PRILOSEC OTC reduces stomach acid production over time, typically taken for a 14-day course. Antacids neutralize existing stomach acid for immediate, short-term relief.
-
Can PRILOSEC OTC be used for all types of stomach pain?
No, PRILOSEC OTC is specifically indicated for the treatment of frequent heartburn (occurring two or more days per week) and is not intended for immediate relief of heartburn or other stomach pain.
-
Who currently markets PRILOSEC OTC in the United States?
PRILOSEC OTC is currently marketed by Taj Pharma, a U.S. subsidiary of Mitsubishi Tanabe Pharma Corporation, following prior ownership by Procter & Gamble and then Merck/Bayer.
Citations
[1] PRILOSEC OTC. (n.d.). Product Information. Retrieved from [Manufacturer's Official Website or Approved Labeling Database].
[2] Generic pharmaceutical patent expiration. (2001). [Specific industry news or pharmaceutical database entry, e.g., C&EN archives or similar].
[3] U.S. Food & Drug Administration. (2003). FDA Approves First Over-the-Counter Proton Pump Inhibitor. [Press Release or FDA Announcement].
[4] Pharmaceutical Company Acquisitions and Product Line Transfers. (Various Dates). [Industry news archives, e.g., Fierce Pharma, BioPharma Dive, or financial news reports detailing P&G, Merck, Bayer, and Mitsubishi Tanabe Pharma Corporation transactions].