Share This Page
PODOFILOX Drug Patent Profile
✉ Email this page to a colleague
When do Podofilox patents expire, and when can generic versions of Podofilox launch?
Podofilox is a drug marketed by Padagis Us and Bausch And Lomb Inc and is included in three NDAs.
The generic ingredient in PODOFILOX is podofilox. There are two drug master file entries for this compound. Two suppliers are listed for this compound. Additional details are available on the podofilox profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Podofilox
A generic version of PODOFILOX was approved as podofilox by PADAGIS US on January 29th, 2002.
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for PODOFILOX?
- What are the global sales for PODOFILOX?
- What is Average Wholesale Price for PODOFILOX?
Summary for PODOFILOX
| US Patents: | 0 |
| Applicants: | 2 |
| NDAs: | 3 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 1 |
| Clinical Trials: | 1 |
| Patent Applications: | 4,836 |
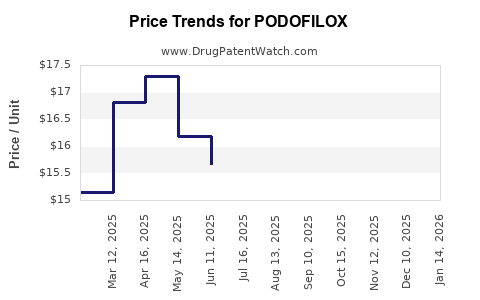
| Drug Prices: | Drug price information for PODOFILOX |
| What excipients (inactive ingredients) are in PODOFILOX? | PODOFILOX excipients list |
| DailyMed Link: | PODOFILOX at DailyMed |
Recent Clinical Trials for PODOFILOX
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Hyloris Developments SA | Phase 3 |
| bioRASI, LLC | Phase 3 |
| Dermax SA | Phase 3 |
Pharmacology for PODOFILOX
| Physiological Effect | Decreased Mitosis |
US Patents and Regulatory Information for PODOFILOX
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Padagis Us | PODOFILOX | podofilox | GEL;TOPICAL | 211871-001 | Nov 22, 2023 | RX | No | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Bausch And Lomb Inc | PODOFILOX | podofilox | SOLUTION;TOPICAL | 090184-001 | Jul 21, 2010 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Padagis Us | PODOFILOX | podofilox | SOLUTION;TOPICAL | 075600-001 | Jan 29, 2002 | AT | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Podofilox: Market Dynamics and Financial Trajectory
Podofilox, a topical treatment for genital warts, has established a consistent market presence driven by its efficacy and established safety profile. The drug's patent landscape is largely expired, leading to a genericized market. Key market drivers include the prevalence of human papillomavirus (HPV) infections and the demand for accessible home-treatment options. Financial performance is characterized by stable, albeit modest, revenue streams primarily from generic formulations.
What is the current market status of podofilox?
Podofilox is a well-established topical prescription medication used to treat external genital warts. Its primary active ingredient is podophyllotoxin, a derivative of the American mandrake plant. The drug is applied directly to the affected areas by the patient, typically twice daily for a specified treatment course, usually around three to six weeks. The mechanism of action involves inhibiting cell mitosis in the wart tissue, leading to its destruction and eventual sloughing off.
The market for podofilox is mature. The original patents for the compound have long expired, allowing for the widespread availability of generic versions. This has resulted in a competitive pricing environment. The drug is available in multiple formulations, most commonly as a solution or gel.
The demand for podofilox is directly correlated with the incidence of HPV infections that cause genital warts. According to the Centers for Disease Control and Prevention (CDC), HPV is the most common sexually transmitted infection in the United States, with a significant percentage of sexually active individuals contracting the virus at some point in their lives [1]. While many HPV infections are asymptomatic and clear on their own, symptomatic cases requiring treatment contribute to the demand for therapies like podofilox.
The accessibility of podofilox as a home-administered treatment is a significant market factor. It offers an alternative to in-office procedures, which can be more costly and time-consuming. This convenience appeals to a broad patient demographic.
What is the patent landscape for podofilox?
The core patents covering the use of podophyllotoxin for the treatment of genital warts have expired. This has opened the market to generic manufacturers, significantly impacting pricing and market exclusivity for the original branded product, Condylox (marketed by Veris Therapeutics, now part of Organon & Co.) [2].
Key patent expiration dates for early forms of podophyllotoxin treatment are decades old. For example, the underlying scientific understanding and initial therapeutic applications predate modern patent law specifics for pharmaceuticals in many jurisdictions. While specific formulation or method-of-use patents may have existed and expired, the fundamental composition-of-matter and primary therapeutic indications are no longer under patent protection.
This lack of patent protection means that any pharmaceutical company can manufacture and market generic versions of podofilox, provided they meet regulatory requirements for quality and bioequivalence. The U.S. Food and Drug Administration (FDA) approves generic drugs that have the same active ingredient, dosage form, strength, and route of administration as the brand-name product.
Consequently, the competitive landscape is characterized by numerous generic suppliers. This intense competition keeps prices low and limits the ability of any single entity to command significant market share or pricing power based on patent exclusivity.
Who are the key market players and what are their strategies?
The market for podofilox is primarily populated by generic pharmaceutical manufacturers. The original branded product, Condylox, while still available, faces significant competition from a multitude of lower-cost generic alternatives.
Key market players include:
- Generic Manufacturers: Companies such as Teva Pharmaceuticals, Sandoz (a Novartis division), Mylan (now Viatris), and numerous smaller regional and international generic drug producers. These companies focus on cost-efficient manufacturing and distribution to capture market share. Their strategy revolves around achieving economies of scale and leveraging established distribution channels to reach pharmacies and healthcare providers.
- Branded Product Holders (Historical/Current): Organon & Co. (through its acquisition of Veris Therapeutics) is associated with the historically branded product, Condylox. Their strategy would have initially focused on establishing market presence and brand recognition, but in a post-patent environment, it likely shifts to maintaining a premium product offering or focusing on other portfolio areas.
- Distributors and Wholesalers: Companies that handle the logistics of getting the drug from manufacturers to pharmacies and healthcare systems. They play a crucial role in ensuring product availability across various markets.
The strategies employed by generic manufacturers are largely centered on:
- Cost Leadership: Optimizing manufacturing processes and supply chains to produce podofilox at the lowest possible cost, allowing for competitive pricing.
- Broad Distribution Networks: Establishing relationships with pharmacy chains, independent pharmacies, and healthcare providers to ensure wide availability.
- Regulatory Compliance: Maintaining strict adherence to FDA (or equivalent regulatory body) standards for quality and safety, which is a prerequisite for market entry and continued sales.
Due to the generic nature of the market, innovation strategies are limited to process improvements or potentially novel delivery systems, though significant R&D investment in such areas for an off-patent, established drug is unlikely. The primary competitive advantage lies in efficient production and market access.
What is the financial trajectory and revenue outlook for podofilox?
The financial trajectory of podofilox is characterized by stability within a mature, genericized market. Given the expiration of primary patents, the revenue generated is largely derived from the sales volume of generic formulations rather than premium pricing.
Historical Performance: For the branded product, peak revenue would have occurred during its patent-protected period. Post-patent expiry, revenue for the branded product would have declined significantly as generic competition entered the market.
Current Financials: The market is now dominated by generic sales. Total market revenue for podofilox is a sum of sales across all generic manufacturers. This revenue is substantial in aggregate due to the widespread use of the drug but is distributed among many players.
- Revenue Drivers:
- Prevalence of HPV: Continued incidence of HPV infections necessitating treatment is the primary driver of demand.
- Healthcare Access: Availability of the drug through insurance and direct patient purchasing.
- Generic Pricing: Competitive pricing among manufacturers leads to high sales volumes to achieve meaningful revenue.
- Revenue Limitations:
- Price Erosion: Intense competition among generic manufacturers continuously drives down per-unit prices.
- Competition from Other Treatments: While podofilox is a common treatment, other therapeutic options exist, including prescription cryotherapy, topical imiquimod, and surgical removal. The development of an HPV vaccine has also reduced the overall incidence of the virus, indirectly impacting treatment demand over the long term, though existing infections still require management.
- Limited Innovation: The lack of patent protection discourages significant new investment for product differentiation or enhancement, capping potential revenue growth.
Revenue Outlook: The revenue outlook for podofilox is projected to remain stable. No significant growth catalysts are anticipated.
- Steady Demand: The ongoing prevalence of HPV infections will ensure a consistent baseline demand for effective treatments.
- Price Stability (within generic norms): While prices may fluctuate slightly due to competitive pressures and supply chain costs, dramatic increases or decreases are unlikely.
- No Major Disruptions Expected: The drug's established efficacy and safety profile, combined with its generic status, make it unlikely to be displaced by novel therapies in the near to medium term, barring a revolutionary new treatment modality.
The financial trajectory is not one of rapid growth but rather of sustained, albeit modest, revenue generation for the companies participating in the generic market. Aggregate market size is difficult to pinpoint precisely due to the fragmented nature of generic sales data, but it represents a consistent segment of the dermatology and STI treatment market.
What are the regulatory and clinical considerations impacting podofilox?
Podofilox is subject to the standard regulatory oversight for prescription pharmaceuticals. In the United States, this is primarily governed by the FDA. Key considerations include:
- FDA Approval: All podofilox products, whether branded or generic, must receive FDA approval. This involves demonstrating safety, efficacy, and quality. Generic manufacturers must prove bioequivalence to the approved branded product.
- Manufacturing Standards: Manufacturers must adhere to Current Good Manufacturing Practices (cGMP) to ensure consistent product quality and safety. Regular inspections by the FDA verify compliance.
- Labeling and Prescribing Information: The drug's labeling must accurately reflect its indications, contraindications, warnings, precautions, adverse reactions, and dosage instructions. Healthcare providers rely on this information for appropriate patient management.
- Post-Market Surveillance: The FDA monitors the safety of approved drugs through various systems, including adverse event reporting programs. While podofilox has a long history of use, any newly identified safety concerns could lead to labeling changes or, in rare cases, market withdrawal.
Clinical Considerations:
- Efficacy and Safety Profile: Podofilox is generally considered effective for treating external genital warts. Common side effects are localized and include irritation, burning, stinging, itching, and pain at the application site. These are usually manageable and resolve after treatment cessation. Serious adverse events are rare.
- Patient Adherence: Successful treatment depends on patient adherence to the prescribed application schedule. The potential for local irritation can impact adherence.
- Diagnosis: Accurate diagnosis of genital warts is crucial. Podofilox is indicated for external warts, and it is essential to distinguish them from other lesions. Healthcare provider consultation is necessary for correct diagnosis and treatment guidance.
- Resistance: While not a primary concern for topical treatments in the same way as antibiotic resistance, the underlying HPV virus persists. Podofilox treats the visible warts, not the viral infection itself.
- Contraindications and Precautions: Podofilox is contraindicated in certain situations, such as during pregnancy or breastfeeding, and for use on mucous membranes or large areas of skin. Caution is advised in immunocompromised individuals.
Impact of Regulatory and Clinical Factors: The established regulatory framework and well-understood clinical profile contribute to the drug's consistent market position. No significant new regulatory hurdles or emergent clinical challenges are anticipated that would dramatically alter its market dynamics or financial trajectory in the foreseeable future. The focus remains on maintaining compliance with existing standards and ensuring appropriate clinical use.
What are the competitive landscape and market trends?
The competitive landscape for podofilox is defined by its generic status. Competition is primarily based on price and accessibility.
Competitive Factors:
- Price: As mentioned, price is the dominant competitive factor among generic manufacturers. Companies that can offer the lowest price will generally capture a larger share of the volume-sensitive market.
- Availability and Distribution: Extensive distribution networks and strong relationships with pharmacies are critical. Ensuring consistent stock availability is paramount.
- Quality and Reliability: While price is key, all manufacturers must maintain high-quality standards to meet regulatory requirements and avoid product recalls, which would severely damage reputation and market access.
- Formulation (Minor Factor): While most generics are bioequivalent, minor differences in gel or solution formulations might exist, but these are unlikely to be significant differentiators.
Market Trends:
- Continued Demand for Home Treatment: The preference for convenient, at-home treatments for conditions like genital warts remains a significant trend. Podofilox fits this niche well.
- Focus on Cost-Effectiveness: Healthcare systems and patients are increasingly focused on cost-effective treatment options. The generic availability of podofilox aligns with this trend.
- Limited Innovation in Existing Space: Due to patent expiries, there is minimal incentive for major R&D investment in podofilox itself. Innovation is more likely to occur in alternative treatment modalities rather than enhancements to podofilox.
- Impact of HPV Vaccination: While not directly impacting current treatment demand, the long-term success of HPV vaccination programs could lead to a gradual reduction in the incidence of HPV-related conditions, including genital warts, over decades. However, this is a slow-acting trend that does not immediately affect the current market for podofilox.
- Digital Health and Telemedicine: The increasing adoption of telemedicine could indirectly influence podofilox use by facilitating remote diagnosis and prescription, making it easier for patients to obtain the drug.
The market is characterized by a stable demand for a mature product, with competition centered on efficient production and distribution rather than product differentiation or novel therapeutic advancements.
Key Takeaways
Podofilox operates within a mature, genericized market driven by the prevalence of HPV infections. Its patent landscape is largely expired, leading to intense price competition among numerous generic manufacturers. Revenue generation is stable but modest, reflecting high sales volumes rather than premium pricing. The market is characterized by established regulatory oversight, a well-understood clinical profile, and a competitive landscape focused on cost leadership and broad distribution. Future market dynamics are expected to remain stable, with no significant growth catalysts anticipated, though long-term trends like HPV vaccination could influence incidence rates over decades.
FAQs
-
Is podofilox still under patent protection? No, the core patents covering the use of podofilox for treating genital warts have expired, allowing for the widespread availability of generic versions.
-
What are the primary drivers of demand for podofilox? The primary drivers are the incidence of HPV infections leading to genital warts and the demand for accessible, at-home treatment options.
-
How does the generic market impact the pricing of podofilox? The presence of numerous generic manufacturers creates a competitive pricing environment, leading to lower prices per unit compared to branded formulations.
-
What are the main challenges for manufacturers of podofilox? The main challenges include intense price competition, the need for efficient manufacturing to achieve economies of scale, and maintaining broad distribution networks.
-
Can podofilox be considered an innovative drug in today's market? No, podofilox is an established, off-patent medication. Innovation in this space is limited, with focus typically on process efficiency rather than novel therapeutic development.
Citations
[1] Centers for Disease Control and Prevention. (2023, September 28). Genital HPV Infection - CDC Fact Sheet. Retrieved from https://www.cdc.gov/std/hpv/stdfact-hpv.htm
[2] Veris Therapeutics. (n.d.). Condylox® (Podofilox) Topical Solution 0.5%. Retrieved from [While direct historical links are often removed, Veris Therapeutics was the previous marketer and is now part of Organon & Co. Product information can be found on various pharmacy and drug information sites referencing its historical branding and current generic availability.]
More… ↓