PLENVU Drug Patent Profile
✉ Email this page to a colleague
When do Plenvu patents expire, and what generic alternatives are available?
Plenvu is a drug marketed by Salix and is included in one NDA. There are eleven patents protecting this drug and one Paragraph IV challenge.
This drug has one hundred and sixty-five patent family members in thirty-seven countries.
The generic ingredient in PLENVU is ascorbic acid; polyethylene glycol 3350; potassium chloride; sodium ascorbate; sodium chloride; sodium sulfate. There are six drug master file entries for this compound. Four suppliers are listed for this compound. Additional details are available on the ascorbic acid; polyethylene glycol 3350; potassium chloride; sodium ascorbate; sodium chloride; sodium sulfate profile page.
DrugPatentWatch® Generic Entry Outlook for Plenvu
There have been four patent litigation cases involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
Indicators of Generic Entry
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for PLENVU?
- What are the global sales for PLENVU?
- What is Average Wholesale Price for PLENVU?
Summary for PLENVU
| International Patents: | 165 |
| US Patents: | 11 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Clinical Trials: | 8 |
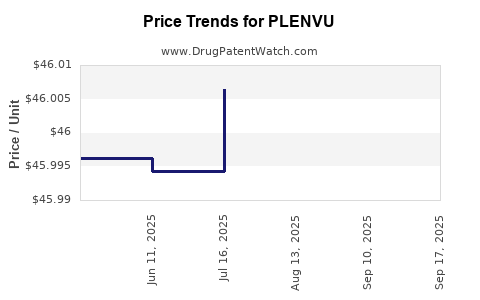
| Drug Prices: | Drug price information for PLENVU |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for PLENVU |
| What excipients (inactive ingredients) are in PLENVU? | PLENVU excipients list |
| DailyMed Link: | PLENVU at DailyMed |
Recent Clinical Trials for PLENVU
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Norgine | PHASE3 |
| Parexel | PHASE3 |
| ASST Rhodense | NA |
Paragraph IV (Patent) Challenges for PLENVU
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| PLENVU | For Oral Solution | ascorbic acid; polyethylene glycol 3350; potassium chloride; sodium ascorbate; sodium chloride; sodium sulfate | 140 g, 5.2 g, 2.2.g, 48.11 g, 9 g and 7.54 g per pouch | 209381 | 1 | 2018-12-06 |
US Patents and Regulatory Information for PLENVU
PLENVU is protected by eleven US patents.
International Patents for PLENVU
When does loss-of-exclusivity occur for PLENVU?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Argentina
Patent: 2500
Patent: SOLUCION DE LIMPIEZA DE COLON
Estimated Expiration: ⤷ Start Trial
Australia
Patent: 13314442
Patent: Compositions comprising PEG and ascorbate
Estimated Expiration: ⤷ Start Trial
Patent: 15228962
Patent: Method of cleansing the colon
Estimated Expiration: ⤷ Start Trial
Patent: 18204694
Patent: COMPOSITIONS COMPRISING PEG AND ASCORBATE
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 2015005270
Patent: composições compreendendo peg e ascorbato
Estimated Expiration: ⤷ Start Trial
Patent: 2016019914
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 84472
Patent: COMPOSITIONS (COMPOSITIONS COMPRISING PEG AND ASCORBATE)
Estimated Expiration: ⤷ Start Trial
Patent: 42089
Patent: METHODE DE NETTOYAGE DU COLON (METHOD OF CLEANSING THE COLON)
Estimated Expiration: ⤷ Start Trial
Patent: 16204
Patent: COMPOSITION POUR NETTOYAGE DU COLON COMPRENANT DE L'ASCORBATE ET DU POLYETHYLENEGLYCOL (PEG) (COLON CLEANSING COMPOSITION COMPRISING ASCORBATE AND PEG)
Estimated Expiration: ⤷ Start Trial
China
Patent: 5007915
Patent: Compositions comprising PEG and ascorbate
Estimated Expiration: ⤷ Start Trial
Patent: 6102738
Patent: 清洗结肠的方法 (Method of cleansing the colon)
Estimated Expiration: ⤷ Start Trial
Patent: 5154464
Patent: 清洗结肠的方法 (Method of cleaning colon)
Estimated Expiration: ⤷ Start Trial
Croatia
Patent: 0191007
Estimated Expiration: ⤷ Start Trial
Cyprus
Patent: 21668
Estimated Expiration: ⤷ Start Trial
Denmark
Patent: 95163
Estimated Expiration: ⤷ Start Trial
Patent: 73248
Estimated Expiration: ⤷ Start Trial
Patent: 77850
Estimated Expiration: ⤷ Start Trial
Eurasian Patent Organization
Patent: 8866
Patent: КОМПОЗИЦИЯ И РАСТВОР ДЛЯ ОЧИЩЕНИЯ ТОЛСТОЙ КИШКИ, ЛЕКАРСТВЕННОЕ СРЕДСТВО (ВАРИАНТЫ), НАБОР И СПОСОБ ОЧИЩЕНИЯ ТОЛСТОЙ КИШКИ (COLON CLEANSING COMPOSITION AND SOLUTION, MEDICAMENT (EMBODIMENTS), KIT AND METHOD OF CLEANSING THE COLON)
Estimated Expiration: ⤷ Start Trial
Patent: 7891
Patent: СПОСОБ ОЧИЩЕНИЯ ТОЛСТОЙ КИШКИ (METHOD OF CLEANSING THE COLON)
Estimated Expiration: ⤷ Start Trial
Patent: 1500320
Patent: КОМПОЗИЦИЯ (ВАРИАНТЫ) И РАСТВОР ДЛЯ ОЧИЩЕНИЯ ТОЛСТОЙ КИШКИ, ЛЕКАРСТВЕННОЕ СРЕДСТВО, НАБОР И СПОСОБ ОЧИЩЕНИЯ ТОЛСТОЙ КИШКИ (ВАРИАНТЫ)
Estimated Expiration: ⤷ Start Trial
Patent: 1600639
Patent: СПОСОБ ОЧИЩЕНИЯ ТОЛСТОЙ КИШКИ
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 95163
Patent: COMPOSITIONS COMPRENANT DU PEG ET DE L'ASCORBATE (COMPOSITIONS COMPRISING PEG AND ASCORBATE)
Estimated Expiration: ⤷ Start Trial
Patent: 16494
Patent: MÉTHODE DE NETTOYAGE DU CÔLON (METHOD OF CLEANSING THE COLON)
Estimated Expiration: ⤷ Start Trial
Patent: 73248
Patent: COMPOSITIONS COMPRENANT DU POLYÉTHYLÈNE GLYCOL ET UN MÉTAL ALCALIN OU DES SULFATES MÉTALLIQUES ALCALINOTERREUX DESTINÉS À ÊTRE UTILISÉS EN TANT QUE COMPOSITIONS DE NETTOYAGE DU CÔLON (COMPOSITIONS COMPRISING POLYETHYLENE GLYCOL AND ALKALI METAL OR ALKALINE EARTH METAL SULPHATES FOR USE AS COLON CLEANSING COMPOSITIONS)
Estimated Expiration: ⤷ Start Trial
Patent: 77850
Patent: PROCÉDÉ DE NETTOYAGE DU COLON (METHOD OF CLEANSING THE COLON)
Estimated Expiration: ⤷ Start Trial
Hong Kong
Patent: 06604
Patent: 含聚乙二醇和抗壞血酸鹽的組合物 (COMPOSITIONS COMPRISING PEG AND ASCORBATE)
Estimated Expiration: ⤷ Start Trial
Hungary
Patent: 43605
Estimated Expiration: ⤷ Start Trial
India
Patent: 04DEN2015
Estimated Expiration: ⤷ Start Trial
Ireland
Patent: 0130273
Patent: Compositions comprising PEG and ascorbate
Estimated Expiration: ⤷ Start Trial
Patent: 6410
Patent: Compositions comprising PEG and ascorbate
Estimated Expiration: ⤷ Start Trial
Israel
Patent: 7377
Patent: הרכבות המכילות peg ו-ascorbate (Compositions comprising peg and ascorbate)
Estimated Expiration: ⤷ Start Trial
Patent: 7170
Patent: שיטה לשטיפת מעי הגס (Method of cleansing the colon)
Estimated Expiration: ⤷ Start Trial
Patent: 9024
Patent: הרכבות המכילות peg ו-ascorbate (Compositions comprising peg and ascorbate)
Estimated Expiration: ⤷ Start Trial
Patent: 4374
Patent: תמיסה לניקוי של מעי הגס (A colon cleansing solution)
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 57675
Estimated Expiration: ⤷ Start Trial
Patent: 55391
Estimated Expiration: ⤷ Start Trial
Patent: 77396
Estimated Expiration: ⤷ Start Trial
Patent: 15527385
Patent: PEGとアスコルビン酸塩とを含む組成物
Estimated Expiration: ⤷ Start Trial
Patent: 17507965
Patent: 結腸を洗浄する方法
Estimated Expiration: ⤷ Start Trial
Patent: 20073579
Patent: PEGとアスコルビン酸塩とを含む組成物 (COMPOSITIONS COMPRISING ASCORBATE AND PEG)
Estimated Expiration: ⤷ Start Trial
Lithuania
Patent: 95163
Estimated Expiration: ⤷ Start Trial
Malaysia
Patent: 4758
Estimated Expiration: ⤷ Start Trial
Patent: 8550
Estimated Expiration: ⤷ Start Trial
Patent: 3378
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 9318
Estimated Expiration: ⤷ Start Trial
Patent: 2868
Estimated Expiration: ⤷ Start Trial
Patent: 6425
Estimated Expiration: ⤷ Start Trial
Patent: 1897
Estimated Expiration: ⤷ Start Trial
Patent: 15003031
Estimated Expiration: ⤷ Start Trial
Patent: 16011256
Estimated Expiration: ⤷ Start Trial
Patent: 21005225
Estimated Expiration: ⤷ Start Trial
Montenegro
Patent: 413
Estimated Expiration: ⤷ Start Trial
Netherlands
Patent: 11424
Estimated Expiration: ⤷ Start Trial
New Zealand
Patent: 5499
Estimated Expiration: ⤷ Start Trial
Patent: 3017
Estimated Expiration: ⤷ Start Trial
Patent: 3984
Estimated Expiration: ⤷ Start Trial
Poland
Patent: 95163
Estimated Expiration: ⤷ Start Trial
Patent: 73248
Estimated Expiration: ⤷ Start Trial
Patent: 77850
Estimated Expiration: ⤷ Start Trial
Portugal
Patent: 95163
Estimated Expiration: ⤷ Start Trial
Patent: 73248
Estimated Expiration: ⤷ Start Trial
Patent: 77850
Estimated Expiration: ⤷ Start Trial
San Marino
Patent: 01900318
Estimated Expiration: ⤷ Start Trial
Serbia
Patent: 979
Estimated Expiration: ⤷ Start Trial
Singapore
Patent: 201702005V
Estimated Expiration: ⤷ Start Trial
Patent: 201807427R
Estimated Expiration: ⤷ Start Trial
Patent: 202112138Q
Estimated Expiration: ⤷ Start Trial
Patent: 201501713X
Estimated Expiration: ⤷ Start Trial
Patent: 201606834Q
Estimated Expiration: ⤷ Start Trial
Slovenia
Patent: 95163
Estimated Expiration: ⤷ Start Trial
South Africa
Patent: 1501415
Estimated Expiration: ⤷ Start Trial
Patent: 1603747
Estimated Expiration: ⤷ Start Trial
Patent: 1605592
Estimated Expiration: ⤷ Start Trial
South Korea
Patent: 1771586
Estimated Expiration: ⤷ Start Trial
Patent: 2234803
Estimated Expiration: ⤷ Start Trial
Patent: 2257207
Estimated Expiration: ⤷ Start Trial
Patent: 150054990
Patent: PEG 및 아스코르베이트를 포함하는 조성물 (COMPOSITIONS COMPRISING PEG AND ASCORBATE)
Estimated Expiration: ⤷ Start Trial
Patent: 160130488
Patent: 대장 세척 방법 (METHOD OF CLEANSING THE COLON)
Estimated Expiration: ⤷ Start Trial
Patent: 170098980
Patent: PEG 및 아스코르베이트를 포함하는 조성물 (PEG COMPOSITIONS COMPRISING PEG AND ASCORBATE)
Estimated Expiration: ⤷ Start Trial
Patent: 190137925
Estimated Expiration: ⤷ Start Trial
Patent: 210063445
Estimated Expiration: ⤷ Start Trial
Patent: 230010844
Estimated Expiration: ⤷ Start Trial
Spain
Patent: 28459
Estimated Expiration: ⤷ Start Trial
Patent: 31412
Estimated Expiration: ⤷ Start Trial
Patent: 09386
Estimated Expiration: ⤷ Start Trial
Patent: 28719
Estimated Expiration: ⤷ Start Trial
Taiwan
Patent: 1416092
Estimated Expiration: ⤷ Start Trial
Patent: 2017562
Estimated Expiration: ⤷ Start Trial
Patent: 87238
Estimated Expiration: ⤷ Start Trial
Patent: 56623
Estimated Expiration: ⤷ Start Trial
Ukraine
Patent: 6546
Estimated Expiration: ⤷ Start Trial
Patent: 3659
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering PLENVU around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Malaysia | 193378 | ⤷ Start Trial | |
| Brazil | 112016019914 | ⤷ Start Trial | |
| South Africa | 201304866 | COLONOSCOPY-PREPARATION | ⤷ Start Trial |
| China | 115154464 | 清洗结肠的方法 (Method of cleaning colon) | ⤷ Start Trial |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for PLENVU
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 3141251 | SPC/GB20/075 | United Kingdom | ⤷ Start Trial | PRODUCT NAME: A MEDICINAL PRODUCT CONSISTING OF A COMBINATION OF A FIRST DOSE PHARMACEUTICAL COMPOSITION AND A SECOND DOSE PHARMACEUTICAL COMPOSITION, THE FIRST DOSE PHARMACEUTICAL COMPOSITION CONSISTING OF THE ACTIVE INGREDIENTS: POLYETHYLENE GLYCOL 3350, SODIUM SULPH; REGISTERED: IS IS/1/17/063/01 20171016; UK PL 20011/0040 20171016 |
| 3141251 | 301099 | Netherlands | ⤷ Start Trial | PRODUCT NAME: A MEDICINAL PRODUCT CONSISTING OF A COMBINATION OF A FIRST DOSE PHARMACEUTICAL COMPOSITION AND A SECOND DOSE PHARMACEUTICAL COMPOSITION, THE FIRST DOSE PHARMACEUTICAL COMPOSITION CONSISTING OF THE ACTIVE INGREDIENTS POLYETHYLENE GLYCOL, SODIUM SULPHATE, SODIUM CHLORIDE AND POTASSIUM CHLORIDE AND THE SECOND DOSE PHARMACEUTICAL COMPOSITION CONSISTING OF THE ACTIVE INGREDIENTS POLYETHYLENE GLYCOL, ASCORBIC ACID, SODIUM ASCORBATE, SODIUM CHLORIDE AND POTASSIUM CHLORIDE; NATIONAL REGISTRATION NO/DATE: RVG 120195 20171114; FIRST REGISTRATION: IS IS/1/17/083/01 20171016 |
| 3141251 | 122021000018 | Germany | ⤷ Start Trial | PRODUCT NAME: EIN ARZNEIMITTEL BESTEHEND AUS EINER KOMBINATION EINER PHARMAZEUTISCHEN ZUSAMMENSETZUNG FUER EINE ERSTE DOSIS UND EINER PHARMAZEUTISCHEN ZUSAMMENSETZUNG FUER EINE ZWEITE DOSIS, WOBEI DIE PHARMAZEUTISCHE ZUSAMMENSETZUNG FUER DIE ERSTE DOSIS AUS DEN WIRKSTOFFEN POLYETHYLENGLYCOL, NATRIUMSULFAT, NATRIUMCHLORID UND KALIUMCHLORID BESTEHT UND DIE PHARMAZEUTISCHE ZUSAMMENSETZUNG FUER DIE ZWEITE DOSIS AUS DEN; NAT. REGISTRATION NO/DATE: 98450.00.00 20180205; FIRST REGISTRATION: ISLAND IS/1/17/083/01 20171016 |
| 3141251 | C202130017 | Spain | ⤷ Start Trial | PRODUCT NAME: COMBINACION DE PRINCIPIOS ACTIVOS EN DOS DOSIS, EN LA QUE LA PRIMERA DOSIS CONSISTE EN LOS PRINCIPIOS ACTIVOS POLIETILENGLICOL, SULFATO DE SODIO, CLORURO DE SODIO Y CLORURO DE POTASIO, Y LA SEGUNDA DOSIS CONSISTE EN LOS PRINCIPIOS ACTIVOS POLIETILENGLICOL, ACIDO ASCORBICO, ASCORBATO DE SODIO, CLORURO DE SODIO Y CLORURO DE POTASIO.; NATIONAL AUTHORISATION NUMBER: 82959-SE/H/1801/001/DC; DATE OF AUTHORISATION: 20180525; NUMBER OF FIRST AUTHORISATION IN EUROPEAN ECONOMIC AREA (EEA): IS/1/17/083/01; DATE OF FIRST AUTHORISATION IN EEA: 20171016 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.