Share This Page
PLAN B Drug Patent Profile
✉ Email this page to a colleague
When do Plan B patents expire, and what generic alternatives are available?
Plan B is a drug marketed by Fdn Consumer and is included in two NDAs.
The generic ingredient in PLAN B is levonorgestrel. There are twenty drug master file entries for this compound. Thirty-five suppliers are listed for this compound. Additional details are available on the levonorgestrel profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Plan B
A generic version of PLAN B was approved as levonorgestrel by NOVEL LABS INC on February 22nd, 2013.
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for PLAN B?
- What are the global sales for PLAN B?
- What is Average Wholesale Price for PLAN B?
Summary for PLAN B
| US Patents: | 0 |
| Applicants: | 1 |
| NDAs: | 2 |
| Raw Ingredient (Bulk) Api Vendors: | 112 |
| Patent Applications: | 3,895 |
| What excipients (inactive ingredients) are in PLAN B? | PLAN B excipients list |
| DailyMed Link: | PLAN B at DailyMed |
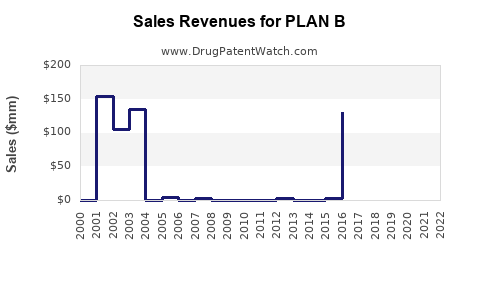
US Patents and Regulatory Information for PLAN B
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Fdn Consumer | PLAN B | levonorgestrel | TABLET;ORAL | 021045-001 | Jul 28, 1999 | DISCN | Yes | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Fdn Consumer | PLAN B | levonorgestrel | TABLET;ORAL | 021045-002 | Aug 24, 2006 | DISCN | Yes | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Fdn Consumer | PLAN B ONE-STEP | levonorgestrel | TABLET;ORAL | 021998-001 | Jul 10, 2009 | OTC | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Plan B: Market Dynamics and Financial Trajectory
Executive Summary
Plan B, a leading emergency contraceptive, has established a significant market presence driven by accessibility and demand. Its financial trajectory is characterized by consistent revenue streams, primarily from direct-to-consumer sales and over-the-counter accessibility. The market is influenced by regulatory policies, patent expirations of alternative methods, and evolving consumer behavior. Future growth is projected to remain steady, with potential impacts from the introduction of novel contraceptive technologies and shifts in healthcare policy.
What is Plan B and What is its Mechanism of Action?
Plan B is an emergency contraceptive pill containing 1.5 milligrams of levonorgestrel, a synthetic progestin. It is intended for use within 72 hours after unprotected intercourse or contraceptive failure to prevent pregnancy. Plan B works primarily by inhibiting or delaying ovulation. If ovulation has already occurred, it may also prevent fertilization by altering the cervical mucus, making it more difficult for sperm to reach the egg. It is not an abortifacient; it does not cause an abortion or harm an existing pregnancy.
What are the Key Patents and Intellectual Property Protecting Plan B?
The original patents covering the use of levonorgestrel as an emergency contraceptive have expired. The primary U.S. patent, U.S. Patent No. 4,297,358, which claimed methods of treating hormonal disorders using progestins including levonorgestrel, expired in 2001. Subsequent patents related to specific formulations, manufacturing processes, or delivery systems might exist for specific branded versions of levonorgestrel-based emergency contraception, such as Plan B One-Step. However, the core composition and method of use are no longer exclusively protected by foundational patents, allowing for the availability of generic versions.
- Original Composition Patents: Expired.
- Formulation & Manufacturing Patents: May exist for specific branded products, but are less likely to be broad.
- Generic Availability: Enabled by patent expiration.
What is the Current Market Size and Projected Growth for Plan B?
The global market for emergency contraceptives, of which Plan B is a significant component, is substantial. While specific figures for Plan B alone are proprietary, the broader emergency contraception market was valued at approximately USD 1.5 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 4.5% from 2023 to 2030. This growth is driven by increased awareness, wider availability, and a growing number of women seeking to prevent unintended pregnancies.
- 2022 Market Value (Emergency Contraceptives): ~USD 1.5 billion
- Projected CAGR (2023-2030): 4.5%
The market for Plan B is further characterized by its over-the-counter (OTC) status in many regions, which significantly boosts accessibility and sales volume compared to prescription-only alternatives.
Who are the Key Manufacturers and Competitors in the Plan B Market?
The market for levonorgestrel-based emergency contraceptives includes both the original branded product and numerous generic alternatives.
Primary Manufacturers/Marketers:
- Foundation Consumer Healthcare (for Plan B One-Step): Holds the rights to the leading branded product in the U.S.
- Generic Manufacturers: A multitude of pharmaceutical companies produce generic versions of 1.5mg levonorgestrel emergency contraceptive pills. Examples include Teva Pharmaceuticals, Lupin Pharmaceuticals, and Aurobindo Pharma.
Key Competitors:
- Other Emergency Contraceptives:
- Ulipristal Acetate (UPA) based products (e.g., ella): While often requiring a prescription in some regions, UPA offers a different mechanism of action and a potentially wider window of effectiveness (up to 120 hours).
- Copper Intrauterine Device (IUD): Highly effective when inserted within 5 days of unprotected sex and also serves as long-term contraception.
- Traditional Contraceptives: Availability and affordability of daily pills, condoms, and other methods influence the demand for emergency options.
- Advocacy for Comprehensive Contraception Access: Organizations promoting wider access to various contraceptive methods indirectly impact the demand for emergency products.
What are the Regulatory and Legal Factors Affecting Plan B?
Regulatory status is a critical determinant of Plan B's market accessibility and consumer reach. The decision to make Plan B available over-the-counter (OTC) has been a significant factor in its market penetration.
- U.S. FDA Approval and OTC Status:
- Plan B One-Step received FDA approval for OTC sales in 2006 for women aged 18 and over.
- In 2013, the FDA removed the age restriction, making it available OTC for all women of reproductive age. This decision significantly expanded its market.
- In 2020, the FDA granted full approval for Plan B One-Step without any restrictions on its sale or use by individuals of any age.
- International Regulatory Approvals: Similar regulatory pathways exist in other countries, with varying timelines for OTC availability.
- Labeling and Marketing Regulations: Manufacturers must adhere to strict guidelines regarding claims, usage instructions, and warnings.
- Legal Challenges and Policy Debates: Plan B has been subject to legal challenges and political debates concerning its accessibility, particularly regarding age restrictions and potential limitations on its sale. These debates can influence public perception and regulatory scrutiny.
- Generic Labeling: Generic versions must meet the same efficacy and safety standards as the branded product, but may have different packaging and marketing.
What is the Financial Performance and Revenue Model for Plan B?
The financial performance of Plan B is largely driven by its OTC status, high sales volume, and the presence of both branded and generic options.
- Revenue Streams:
- Direct-to-Consumer Sales: The primary driver, facilitated by OTC availability in pharmacies, supermarkets, and online retailers.
- Wholesale Distribution: Sales to pharmacies and healthcare providers.
- Brand vs. Generic Pricing: Plan B One-Step commands a premium price, while generic versions compete on cost, offering a wider price range for consumers.
- Pricing:
- Plan B One-Step: Typically ranges from $35 to $60 USD.
- Generic Levonorgestrel: Can range from $15 to $30 USD.
- Sales Volume: Driven by demand for emergency contraception and the ease of purchase. While specific sales figures for Plan B One-Step are proprietary, the aggregate market for emergency contraception indicates substantial unit sales.
- Profit Margins: Branded products likely have higher profit margins per unit, but generic versions contribute to overall market volume and accessibility, creating a dual revenue strategy.
- Market Penetration: High due to OTC accessibility, broad marketing, and social acceptance as a reproductive health option.
What are the Key Market Trends and Drivers for Plan B?
Several factors are shaping the current and future market for Plan B.
- Increased Awareness and Education: Public health campaigns and educational initiatives have improved understanding of emergency contraception and its proper use.
- OTC Accessibility: The removal of prescription requirements in many key markets has been a significant growth driver, simplifying access and reducing barriers.
- Generic Competition: The availability of lower-cost generic alternatives increases affordability and market penetration, though it can pressure margins for branded products.
- Shifts in Contraceptive Use Patterns: While not a primary method, Plan B remains a critical backup for contraceptive failures and unprotected sex. Trends in primary contraceptive use indirectly influence its demand.
- Social and Political Advocacy: Ongoing discussions and advocacy surrounding reproductive rights and access to contraception can influence public perception and policy.
- Technological Advancements: While Plan B's core technology is established, research into novel contraceptive methods, including longer-acting reversible contraceptives (LARCs) and potentially new emergency methods, could represent future competition.
- E-commerce Growth: Online sales channels are increasingly important for accessibility and discreet purchasing.
What are the Challenges and Risks Facing the Plan B Market?
Despite its established position, Plan B faces several challenges.
- Stigmatization and Misinformation: Though reduced, some societal stigma and misinformation about emergency contraception persist, potentially affecting consumer behavior.
- Competition from Novel Contraceptives: Development of more advanced or convenient emergency contraceptive options, or more effective long-term methods, could divert market share.
- Regulatory Uncertainty: While currently favorable in major markets, changes in regulatory policies or political shifts could impact availability or marketing.
- Reimbursement Policies: While largely OTC, insurance coverage and reimbursement policies for emergency contraception can vary, potentially affecting affordability for some consumers.
- Dependence on Unintended Pregnancy Rates: While a solution for unintended pregnancies, its demand is inherently linked to the occurrence of such events, which are influenced by broader societal factors and access to primary contraception.
- Supply Chain Disruptions: Like all pharmaceutical products, Plan B is subject to potential supply chain vulnerabilities.
What is the Future Outlook for Plan B?
The future outlook for Plan B is one of stable, albeit moderate, growth. Its position as a readily accessible emergency contraceptive is secure in the short to medium term.
- Continued OTC Dominance: Plan B and its generic equivalents are expected to maintain their strong presence due to established accessibility and consumer familiarity.
- Market Growth Drivers: Ongoing demand for emergency contraception, coupled with increased global awareness and accessibility initiatives, will sustain growth.
- Impact of Generics: The generic market will continue to expand, offering more affordable options and driving volume.
- Potential for Innovation: While the core levonorgestrel product is mature, further innovations in formulation or delivery could emerge, although significant market disruption from new emergency contraceptive mechanisms is not immediately anticipated.
- External Policy Influence: Future regulatory changes or policy shifts regarding reproductive health will remain the most significant external factor influencing the market.
Key Takeaways
Plan B is a mature, yet resilient, product in the emergency contraceptive market, driven by its OTC status and consistent demand. Patent expirations have fostered a competitive generic landscape, ensuring broad accessibility and price variation. The market's financial trajectory is characterized by steady revenue, underpinned by direct-to-consumer sales. Regulatory approvals and public health education have been critical for its widespread adoption. While facing competition from alternative methods and potential policy shifts, Plan B is projected to maintain its market position with stable growth.
Frequently Asked Questions
-
What is the primary revenue driver for Plan B? Direct-to-consumer sales, facilitated by its over-the-counter availability in pharmacies and retail outlets, are the primary revenue driver for Plan B.
-
How has the expiration of original patents impacted the Plan B market? The expiration of foundational patents has allowed for the introduction of numerous generic versions of levonorgestrel emergency contraceptives, increasing market competition and lowering prices for consumers.
-
Are there any significant new technologies expected to disrupt the Plan B market in the near future? While research into novel contraceptive technologies continues, no immediate disruptive technologies are widely anticipated to significantly alter the established market for levonorgestrel-based emergency contraception in the short to medium term.
-
What is the typical price range for Plan B and its generic alternatives? Branded Plan B One-Step typically ranges from $35 to $60 USD, while generic levonorgestrel emergency contraceptive pills can be purchased for $15 to $30 USD.
-
What role does regulatory status play in Plan B's market success? Plan B's over-the-counter regulatory status, particularly in key markets like the United States, is critical. It has removed prescription barriers, significantly increasing accessibility, sales volume, and market penetration.
Citations
[1] Global Emergency Contraceptives Market Size, Share & Trends Analysis Report by Product Type (Levonorgestrel, Ulipristal Acetate, Others), by Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), by Region, and Segment Forecasts, 2023 - 2030. (n.d.). Grand View Research. Retrieved from https://www.grandviewresearch.com/industry-analysis/emergency-contraceptives-market [2] U.S. Food and Drug Administration. (n.d.). FDA Approves Plan B One-Step for Over-the-Counter Use for All Women of Reproductive Age. Retrieved from https://www.fda.gov/news-events/press-announcements/fda-approves-plan-b-one-step-over-counter-use-all-women-reproductive-age
More… ↓
