Last updated: February 19, 2026
Penciclovir, a nucleoside analog antiviral, exhibits a mature market characterized by established generics and ongoing competition within its therapeutic niches. The drug's primary indications, recurrent herpes labialis and treatment of herpes simplex virus (HSV) infections in immunocompromised patients, define its commercial landscape. While patent expiries have led to significant generic penetration, the drug's efficacy and established treatment protocols ensure continued demand. Financial performance is largely driven by sales volume of both branded and generic formulations across major global markets.
What is the Current Market Size and Growth Projection for Penciclovir?
The global market size for penciclovir is not publicly reported as a distinct entity, as it is often aggregated with other antiviral medications, particularly acyclovir and its derivatives. However, industry analyses of topical antivirals and oral antiviral treatments for herpes infections indicate a stable, albeit slow-growing, market. The market is primarily driven by the prevalence of HSV infections, which remain widespread.
- Prevalence of HSV: Herpes simplex virus infections affect a significant portion of the global population. Estimates suggest that between 60% and 95% of adults worldwide have been infected with HSV-1 or HSV-2 [1]. Recurrent herpes labialis (cold sores) affects an estimated 20-40% of the population annually [2].
- Market Drivers:
- High prevalence of HSV infections.
- Patient preference for topical treatments for symptomatic relief of cold sores.
- Continued use in immunocompromised populations for managing HSV outbreaks.
- Market Restraints:
- Patent expiries leading to generic competition and price erosion.
- Development of newer antiviral agents with potentially improved efficacy or novel mechanisms of action.
- Limited efficacy against latent viral reservoirs.
- Growth Projection: The market for penciclovir is expected to experience low single-digit compound annual growth rate (CAGR) over the next five to seven years. This growth will be primarily fueled by emerging markets and sustained demand in developed markets due to its established safety profile and efficacy for specific indications. Growth will be tempered by generic competition and the absence of significant new clinical indications.
What are the Key Therapeutic Areas and Competitive Landscape for Penciclovir?
Penciclovir's primary therapeutic applications are focused on managing herpes simplex virus infections.
- Primary Indications:
- Recurrent Herpes Labialis (Cold Sores): Penciclovir is available as a topical cream (e.g., Denavir) for the treatment of cold sores. Its efficacy lies in shortening the healing time and reducing symptom duration when applied early in the course of an outbreak.
- Herpes Simplex Virus (HSV) Infections in Immunocompromised Patients: Oral formulations of penciclovir are used for the treatment of HSV infections in individuals with weakened immune systems, such as those undergoing chemotherapy or with HIV/AIDS.
- Competitive Landscape:
- Generics: The market for penciclovir is heavily influenced by generic competition. Following patent expiries, numerous pharmaceutical companies have introduced generic versions of penciclovir creams and oral tablets, leading to significant price reductions.
- Direct Competitors (Topical):
- Acyclovir (e.g., Zovirax cream): The most prominent competitor, also a nucleoside analog, widely used for cold sores.
- Valacyclovir (e.g., Valtrex oral, but available in topical formulations): While primarily an oral prodrug, its active metabolite acyclovir competes in the topical space.
- Docosanol (e.g., Abreva): A non-nucleoside analog that inhibits viral entry into cells, also used for cold sores.
- Direct Competitors (Oral - Immunocompromised):
- Acyclovir (oral): The historical standard of care for many HSV infections.
- Valacyclovir (oral): A prodrug of acyclovir with improved oral bioavailability, often preferred due to less frequent dosing.
- Famciclovir (oral): Another nucleoside analog, similar in mechanism and efficacy to penciclovir and valacyclovir.
- Market Positioning: Penciclovir holds a position as a well-established antiviral. For topical use, it competes with other antivirals based on cost, physician prescribing habits, and patient experience. In immunocompromised patients, it is one of several effective nucleoside analog options, with the choice often dictated by cost, tolerability, and physician familiarity.
What is the Intellectual Property and Patent Expiration Status for Penciclovir?
The primary patents protecting penciclovir have long expired, leading to a highly genericized market.
- Original Patents: The foundational patents for penciclovir and its formulations were filed in the late 1980s and early 1990s. Key patents typically have a term of 20 years from the filing date, with potential extensions for regulatory delays.
- Patent Expirations:
- The core composition of matter patents for penciclovir expired in the early 2010s.
- Formulation and method of use patents may have had staggered expiry dates, but most, if not all, have now expired.
- Current IP Landscape:
- No Active Compound Patents: There are no active patents covering the penciclovir molecule itself that prevent generic manufacturing.
- Potential for Niche Patents: Pharmaceutical companies may hold patents on specific novel formulations, delivery systems, or combination therapies involving penciclovir. However, these are unlikely to significantly impact the broad market for standard formulations.
- Generic Approvals: Regulatory bodies like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) have approved numerous generic versions of penciclovir products.
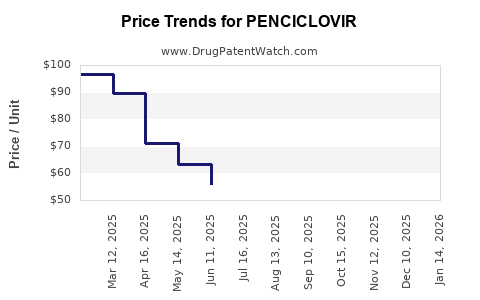
- Impact of Expirations: The expiration of key patents has resulted in intense price competition from generic manufacturers, significantly lowering the cost of penciclovir treatment and impacting the profitability of branded versions.
What are the Key Manufacturers and Market Share Distribution for Penciclovir?
The manufacturing and distribution of penciclovir are dominated by generic pharmaceutical companies.
- Key Generic Manufacturers:
- Teva Pharmaceutical Industries
- Mylan N.V. (now Viatris)
- Sun Pharmaceutical Industries
- Aurobindo Pharma
- Dr. Reddy's Laboratories
- Other regional and international generic drug manufacturers.
- Branded Manufacturer (Historical):
- SmithKline Beecham (now part of GlaxoSmithKline) was the original developer and marketer of branded penciclovir (e.g., Denavir). However, the branded product has a significantly diminished market presence due to generic competition.
- Market Share Distribution:
- Generic Dominance: Generic manufacturers collectively hold the vast majority of the market share for penciclovir globally.
- Fragmented Market: The market is fragmented, with no single generic manufacturer holding a dominant position. Market share is distributed among numerous players based on manufacturing capacity, distribution networks, and pricing strategies.
- Regional Variations: Market share can vary geographically, with some generic companies having stronger footholds in specific regions (e.g., North America, Europe, Asia).
- Impact of Pricing: Pricing strategies are a critical determinant of market share in the generic penciclovir market. Companies offering the lowest prices tend to capture larger volumes.
What is the Financial Trajectory and Revenue Generation for Penciclovir?
The financial trajectory of penciclovir is characterized by declining revenues for branded products and stable, volume-driven revenue for generic formulations.
- Branded Product Revenue: Revenue from branded penciclovir products has significantly decreased since patent expiries. Sales are now primarily from a small patient base that continues to use the branded product or from specific niche markets where generic availability or physician preference is lower. GlaxoSmithKline's reported sales for Denavir (penciclovir cream) were minimal in recent years, reflecting its transition to a largely genericized product.
- Generic Product Revenue: The financial performance of penciclovir is now primarily driven by the aggregate sales volume of generic formulations worldwide.
- Volume-Driven: Revenue is generated through high-volume sales at lower profit margins per unit.
- Price Sensitivity: The market is highly price-sensitive, with continuous pressure to reduce manufacturing costs and offer competitive pricing.
- Global Reach: Revenue is derived from sales across numerous countries, with contributions from both developed and emerging markets.
- Estimated Global Generic Market Value: While precise figures are elusive due to aggregation, the global market for topical antiviral creams and oral antivirals for HSV infections is estimated to be in the hundreds of millions of U.S. dollars annually. Penciclovir, as a component of this market, contributes a fraction of this total.
- Profitability:
- Branded: Historically, branded penciclovir offered higher profit margins.
- Generic: Profitability for generic manufacturers relies on efficient manufacturing, economies of scale, and cost control. Margins per unit are low, but overall profitability can be achieved through significant sales volumes.
- Future Financial Outlook: The financial trajectory for penciclovir is expected to remain stable with slow, incremental growth. This growth will be driven by increasing global populations and continued demand for cost-effective antiviral treatments. Significant revenue spikes are unlikely without the discovery of new therapeutic indications or novel delivery systems that can command premium pricing.
What are the Regulatory Considerations and Market Access Challenges for Penciclovir?
Penciclovir faces a standard regulatory pathway for generic drugs but encounters specific market access challenges related to its therapeutic class and competitive landscape.
- Regulatory Approvals:
- Generic Approvals: Manufacturers seeking to market generic penciclovir must demonstrate bioequivalence to the reference listed drug (RLD). This involves submitting Abbreviated New Drug Applications (ANDAs) to regulatory agencies such as the U.S. FDA or Marketing Authorization Applications (MAAs) to the EMA.
- Manufacturing Standards: Facilities must adhere to Good Manufacturing Practices (GMP) to ensure product quality and safety.
- Labeling: Generic labels must be comparable to the RLD's label, though they may be updated to reflect post-approval safety information.
- Market Access Challenges:
- Payer Scrutiny: While penciclovir is generally considered an established and cost-effective treatment, payers (insurance companies, government health programs) continuously evaluate drug costs. The availability of multiple generic options intensifies pressure on pricing.
- Formulary Placement: Generic penciclovir products compete for favorable placement on formularies. Lower-cost generics are typically preferred.
- Physician Prescribing Habits: While generics offer cost savings, physicians may maintain prescribing habits based on familiarity, perceived efficacy, or established patient responses to specific brands or generic manufacturers.
- Competition with Other Antivirals: Penciclovir competes with acyclovir and valacyclovir. Payer policies may favor one over the other based on cost-effectiveness analyses or preferred drug lists.
- Emerging Markets: Market access in emerging markets can be complicated by infrastructure challenges, varying regulatory landscapes, and local pricing sensitivities.
- Post-Market Surveillance: Like all approved drugs, penciclovir is subject to post-market surveillance for adverse events and pharmacovigilance. Manufacturers must report any significant safety concerns to regulatory authorities.
Key Takeaways
- Penciclovir operates in a mature, genericized market primarily driven by demand for topical treatment of recurrent herpes labialis and management of HSV in immunocompromised individuals.
- The market size is stable with low single-digit growth projections, influenced by the high prevalence of HSV infections but constrained by intense price competition from generics and the lack of new clinical indications.
- Original patents have expired, leading to a landscape dominated by numerous generic manufacturers. Branded product revenue is negligible.
- Key competitors include other nucleoside analogs like acyclovir and valacyclovir, as well as non-nucleoside antivirals such as docosanol.
- Financial performance is volume-driven for generics, with profitability dependent on efficient manufacturing and cost control, rather than high profit margins per unit.
- Regulatory hurdles primarily involve demonstrating bioequivalence for generic approval. Market access is influenced by payer scrutiny, formulary placement, and physician prescribing patterns, all within a highly cost-sensitive environment.
Frequently Asked Questions
What is the primary difference in mechanism of action between penciclovir and docosanol for cold sores?
Penciclovir is a nucleoside analog that, after intracellular phosphorylation, inhibits viral DNA polymerase. Docosanol is a saturated fatty alcohol that is thought to work by interfering with viral entry into host cells and the fusion of the viral envelope with the host cell membrane.
How does the efficacy of penciclovir compare to valacyclovir for treating HSV infections in immunocompromised patients?
Both penciclovir and valacyclovir are effective antiviral agents for HSV infections in immunocompromised patients. Valacyclovir is a prodrug of acyclovir and generally has better oral bioavailability than acyclovir, leading to less frequent dosing. Clinical trials have shown comparable efficacy between penciclovir, valacyclovir, and famciclovir in managing and preventing HSV recurrences in various immunocompromised settings, with the choice often based on dosing convenience, tolerability, and cost.
Are there any ongoing clinical trials investigating new uses or improved formulations of penciclovir?
Publicly available information does not indicate significant ongoing clinical trials for novel indications or major formulation improvements for penciclovir. Research in antiviral therapies has largely shifted towards newer drug classes or different viral targets, with penciclovir's therapeutic niche remaining well-defined by its established indications.
What is the typical cost difference between branded penciclovir (if still available) and its generic versions?
Branded penciclovir products, if still available, would typically be significantly more expensive than their generic counterparts. Generic medications are priced much lower due to competition among multiple manufacturers after patent expiration. For example, a prescription for a branded topical penciclovir could cost upwards of $70-$100 USD, while a generic equivalent might range from $15-$30 USD, depending on the pharmacy and insurance coverage.
What are the common side effects associated with topical penciclovir?
The most common side effects associated with topical penciclovir (cream) are generally mild and localized. These include application site reactions such as mild burning, stinging, or itching. Headache has also been reported, although less frequently. Systemic side effects are rare with topical application.
What is the recommended duration of treatment for recurrent herpes labialis using topical penciclovir?
Topical penciclovir for recurrent herpes labialis is typically recommended to be applied as soon as the first signs of a cold sore appear (e.g., tingling, itching, redness). It is usually applied multiple times a day (e.g., every 2 hours while awake for 4 days) to shorten healing time and reduce symptom duration. Treatment should continue for the full duration prescribed by a healthcare professional, even if symptoms improve earlier.
Citations
[1] The World Health Organization. (2023). Herpes simplex virus. Retrieved from https://www.who.int/news-room/fact-sheets/detail/herpes-simplex-virus
[2] National Institute of Allergy and Infectious Diseases. (n.d.). Genital Herpes. Retrieved from https://www.niaid.nih.gov/diseases-conditions/genital-herpes