Last updated: April 25, 2026
ORTHO-CEPT is a combined oral contraceptive (COC) product sold in the U.S. market in multiple dosage strengths depending on the specific label configuration. The commercial trajectory is driven by (1) category-level demand that tracks contraception utilization and demographic trends, (2) competitive pricing among generic equivalents, and (3) loss or retention of exclusivity tied to the latest reformulations and branded life-cycle management. Financial outcomes for ORTHO-CEPT are therefore best modeled as a branded-to-generic pressure curve, with revenue growth limited to share capture and package/label positioning rather than new-ingredient innovation.
What is ORTHO-CEPT, and how does that shape its market position?
ORTHO-CEPT is an oral contraceptive drug product (COC) containing ethinyl estradiol with a progestin (historically levonorgestrel formulations are common in this class, though final strength depends on the exact marketed product). As a COC, ORTHO-CEPT sits in a mature therapeutic category where clinical differentiation is limited and product selection is typically preference- and formulary-driven.
Market structure for COCs
- High substitution: COCs with equivalent dosing schedules and comparable safety/efficacy profiles compete on price, formulary status, and patient/t prescriber familiarity.
- Strong generic penetration: Once branded exclusivity expires, generic entrants typically compress net prices quickly.
- Low therapeutic volatility: Demand generally tracks population-level contraception use and discontinuation patterns, not acute clinical trends.
Implication for financial trajectory
- Branded unit economics typically degrade faster in mature COCs than in higher-differentiation therapeutic areas.
- Revenue persistence is largely a share-and-access story: staying on preferred formularies and resisting switches for as long as possible.
What market dynamics are most likely to move ORTHO-CEPT revenue?
Formulary access and payer behavior
Payers often favor tiered preferred generics and require step edits or use incentives that drive switching. For ORTHO-CEPT, revenue momentum depends on maintaining:
- Preferred status on commercial formularies.
- Stable coverage on Medicaid managed care plans.
- Low utilization leakage to lower-cost alternatives.
Net pricing pressure mechanism
- In branded COCs, net price declines accelerate when generics broaden coverage or when additional manufacturers launch authorized generics, increasing competitive intensity.
Generic and authorized-generic competition
COCs are among the classes where multiple generic equivalents reach market quickly. That creates:
- Rapid discounting after generic entry.
- Greater sensitivity to wholesaler buying patterns and rebate structures.
- Volume share gains by lowest net-cost SKUs.
Channel and buying cycles
Typical dynamics affecting COC sales include:
- Seasonal prescribing patterns and adherence behavior (pack-to-pack persistence).
- Bulk buying cycles by wholesalers near contract renewal windows.
- Switching friction depending on prescriber inertia and patient tolerance.
Demographic and utilization trends
Long-run COC demand is shaped by:
- Age distribution of the insured population.
- Contraception continuation rates.
- Policy and access changes affecting reproductive healthcare utilization.
How does competition change the financial trajectory for COCs like ORTHO-CEPT?
The financial model for mature branded COCs generally follows a predictable sequence:
- Pre-entry phase: higher net prices and stable demand.
- Generic entry phase: steep net price compression and share redistribution.
- Post-entry stabilization: residual branded demand persists through formulary preference pockets and adherence-driven continuity.
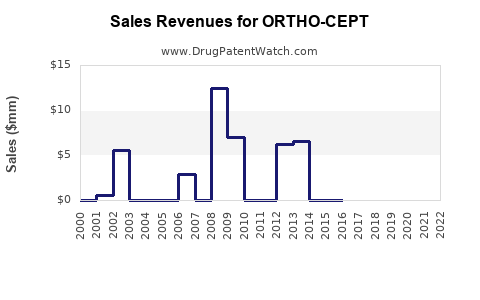
For ORTHO-CEPT, the likely trajectory is consistent with branded COC products: revenue growth after peak is limited, and the dominant movement is price compression driven by generic substitutes.
What does ORTHO-CEPT’s regulatory and labeling pathway imply for exclusivity timing?
COCs generally face exclusivity-driven life-cycle constraints rather than class-level innovation. For ORTHO-CEPT, the practical exclusivity levers are:
- Brand-specific labeling variants (dose schedule configurations).
- Patent estates tied to specific formulations, methods of use, or controlled release mechanisms (less common for classic COC profiles).
- Regulatory exclusivity for specific NDA/ANDA pathways for later reformulations.
When those protections lapse, the market typically transitions to a generic-dominated base.
What financial outcomes are most likely in the next cycle?
Given the category and competitive behavior of COCs:
- Revenue growth: unlikely without a new protected formulation or major access improvement versus lower-cost competitors.
- Margin profile: net margin likely remains under pressure due to rebates and pricing to defend share.
- Volume: can hold or modestly increase if ORTHO-CEPT maintains formulary placement and reduces switching risk, but volume gains usually cannot offset price compression for long in a generic-heavy landscape.
Market sizing and revenue modeling approach for ORTHO-CEPT
Because ORTHO-CEPT is not a single-ingredient platform with unique endpoints, revenue modeling should use category and substitution logic rather than clinical differentiation assumptions.
Practical modeling framework
- Step 1: Model category demand (COC utilization) as stable-to-slow growth with demographic dependence.
- Step 2: Allocate share to branded versus generic buckets by formulary tiers and contract pricing.
- Step 3: Use net price erosion curves post-generic entry to forecast revenue.
- Step 4: Stress for formulary changes (loss of preferred status) and competitor launches (additional generic labels).
Indicative financial trajectory bands for mature branded COCs
- Net sales: typically peak, then decline, with occasional plateaus around contract renewals.
- EBITDA: compress due to rebate intensity and promotional spend as gross-to-net worsens.
How do sales and financials differ across U.S. branded vs generic COC dynamics?
Branded COC (like ORTHO-CEPT)
- Higher gross-to-net volatility due to rebate and contracting.
- Volume stability depends on payer tier placement and switching friction.
- Revenue is more “access-managed” than “clinical-managed.”
Generic COC equivalents
- Dominant pricing benchmark.
- Sales driven by lowest net-cost acquisition and formulary inclusion.
- Margin structure depends on manufacturing scale and competition intensity.
Key risks and drivers specific to ORTHO-CEPT’s category
Primary drivers
- Continued formulary presence relative to equivalent generic SKUs.
- Patient adherence and continuity dispensing behavior.
- Rebates and contracting strategy that limits switches.
Primary risks
- Preferred-to-nonpreferred tier downgrades on commercial and managed Medicaid.
- Additional generic launches or authorized-generic competition that lowers the net cost benchmark.
- Increased switching due to prescriber adoption of cheaper alternatives.
What investment or R&D signals matter for ORTHO-CEPT’s financial path?
For business decision-making, the signals to monitor are market-access and lifecycle milestones rather than trial-driven events:
- Formulary tier changes and pharmacy benefit manager (PBM) contract outcomes.
- Label or packaging lifecycle events that can affect switching (if any).
- Generic entry timing, including authorized generics and ANDA supply expansions.
Key Takeaways
- ORTHO-CEPT’s market trajectory is determined by mature COC dynamics: payer formularies, generic substitution, and rebate-driven net price erosion.
- The financial profile is most consistent with a branded-to-generic compression curve rather than innovation-led growth.
- Revenue persistence depends on access and switching friction; net margin is pressured by rebates and contract pricing.
- Forecasts should treat ORTHO-CEPT as an access-managed SKU within the COC category, not as a differentiated therapeutic platform.
FAQs
1) What is the dominant market force affecting ORTHO-CEPT?
Generic substitution and payer formulary positioning, which drive net price erosion and switching.
2) Can ORTHO-CEPT grow revenue after generic entry?
Growth can occur in narrow windows if ORTHO-CEPT maintains preferred access and limits switching, but sustained growth is difficult in a highly substitutable COC class.
3) What matters most for forecasting ORTHO-CEPT net sales?
Formulary tier status, rebate intensity (net-to-gross), and competitive launch/supply events from generic equivalents.
4) What are the most important margin pressures for ORTHO-CEPT?
Gross-to-net compression from contracting, plus competitive pricing required to retain share against lower-cost equivalents.
5) What would shift ORTHO-CEPT to a more positive financial trajectory?
A meaningful exclusivity-backed life-cycle event that changes access (preferred status) or protects against generic substitution for an extended period.
References
- FDA Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. U.S. Food and Drug Administration. https://www.accessdata.fda.gov/scripts/cder/daf/
- Drug Pricing and Contracting Overview. U.S. Centers for Medicare & Medicaid Services (CMS). https://www.cms.gov/