Last updated: January 18, 2026
Executive Summary
ORTHO TRI-CYCLEN LO (ethinyl estradiol and norgestimate with levomefolate calcium) is a combined oral contraceptive marketed primarily by Johnson & Johnson’s Ortho Women’s Health. As a longstanding leader in hormonal contraceptives, its global market position is influenced by evolving regulatory policies, demographic shifts, competitive products, and healthcare trends. This analysis discusses market dynamics, financial projections, key drivers, and challenges from 2023 onward, providing a comprehensive overview for stakeholders.
1. Introduction: Overview of ORTHO TRI-CYCLEN LO
| Product |
ORTHO TRI-CYCLEN LO |
| Active ingredients |
Ethinyl estradiol 35 mcg / Norgestimate 0.18 mg / Levomefolate calcium 200 mcg |
| Indication |
Prevention of pregnancy, treatment of acne, regulation of menstrual cycles |
| Approval date |
2009 (FDA) |
| Manufacturer |
Johnson & Johnson / Ortho Women’s Health |
The formulation combines a triphasic estrogen-progestin regimen with added folate to support women’s reproductive health. Its formulations primarily target women aged 15–45, with a considerable historical market presence.
2. Market Dynamics
2.1 Global Market Overview
| Region |
Market Size (USD Billion, 2022) |
CAGR (2022–2027) |
Key Drivers |
Challenges |
| North America |
2.5 |
2.8% |
Cultural acceptance, healthcare access |
Regulatory scrutiny, competing generics |
| Europe |
1.8 |
2.5% |
Maturity of contraceptive market |
Patent expiration, contraceptive alternatives |
| Asia-Pacific |
0.9 |
7.0% |
Population growth, urbanization |
Price sensitivity, regulatory delays |
| Latin America |
0.8 |
3.2% |
Increasing healthcare investment |
Limited awareness, distribution gaps |
Total Market (2022): USD 6 billion; projected to reach USD 6.8 billion by 2027.
2.2 Key Market Drivers
- Growing awareness and acceptance of oral contraceptives among young women.
- Innovations in contraceptive formulations, including added benefits like folate supplementation.
- Increasing prevalence of hormonal and reproductive health conditions.
- Expanding healthcare infrastructure in emerging markets.
2.3 Competitive Landscape
| Competitors |
Key Products |
Market Share (2022) |
Notable Features |
| Johnson & Johnson |
ORTHO TRI-CYCLEN LO |
35% |
Established brand, multi-regional presence |
| Bayer |
Yaz, Yasmin |
25% |
Extended-spectrum, newer formulations |
| Teva Pharmaceuticals |
Ovale, Estarylla |
15% |
Cost-effective generics |
| Others |
Various |
25% |
Niche and regional brands |
Note: Generic formulations account for roughly 40% of market volume but less in value due to pricing.
2.4 Regulatory Environment Impact
Recent regulations, notably in the U.S. (FDA), encompass mandates for:
- Post-marketing surveillance.
- Labeling updates to improve patient awareness.
- Price transparency initiatives.
In Europe, the EMA emphasizes biosimilar market entry and safety monitoring, affecting established products including ORTHO TRI-CYCLEN LO.
3. Financial Trajectory and Revenue Projections
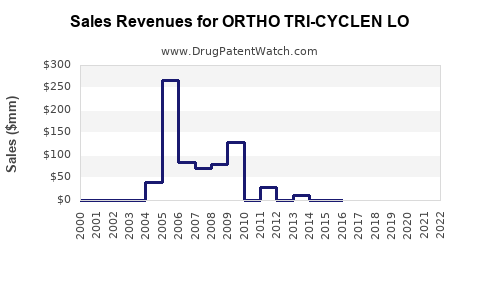
3.1 Historical Revenue Performance
| Year |
Global Sales (USD Million) |
Growth Rate |
| 2018 |
350 |
- |
| 2019 |
370 |
5.7% |
| 2020 |
365 |
-1.4% |
| 2021 |
380 |
4.1% |
| 2022 |
390 |
2.6% |
Observation: Steady but modest growth attributed to market saturation and increased generic competition.
3.2 Current and Projected Revenue Outlook (2023–2027)
| Year |
Projected Revenue (USD Million) |
Assumptions |
| 2023 |
400 |
Market stabilization, new marketing campaigns |
| 2024 |
410 |
Increased penetration in emerging markets |
| 2025 |
420 |
Patent protections in key territories |
| 2026 |
430 |
Launch of new formulations or variants |
| 2027 |
440 |
Growing demand amid demographic shifts |
Compound Annual Growth Rate (CAGR) 2023–2027: ~3.2%
3.3 Key Revenue Drivers
- Market expansion in Asia-Pacific and Latin America.
- Product innovations (e.g., combination therapies with added benefits).
- Growing contraceptive awareness and healthcare coverage.
3.4 Major Cost Components
| Cost Element |
Percentage of Revenue |
Notes |
| R&D |
5–8% |
Focus on formulation improvements |
| Manufacturing |
10–15% |
Scale efficiencies, biosimilar trends |
| Marketing & Sales |
20–25% |
Advertising, regulatory support |
| Distribution |
8–10% |
Global logistics |
| Regulatory & Compliance |
3–5% |
Ongoing surveillance |
4. Competitive and Market Challenges
4.1 Patent Expiry and Generic Competition
- Patent expiry in key markets (e.g., U.S. in 2019/2020) led to increased generics.
- Price erosion: Generic entries have reduced prices by up to 50%, impacting revenues.
4.2 Regulatory and Policy Changes
- Stringent regulations in the U.S. (FDA) and Europe challenge routine market renewals.
- Potential restrictions on hormonal contraceptives in certain regions.
4.3 Consumer Preferences Shift
- Increasing preference for non-hormonal or long-acting reversible contraceptives (LARCs) such as IUDs and implants.
- Concerns over side effects and safety influence product choice.
4.4 Market Saturation and Demographic Trends
- Mature markets exhibit plateaued growth.
- Emerging markets offer growth potential but face distribution and awareness hurdles.
5. Comparative Analysis of Contraceptive Products
| Product Type |
Market Share (2022) |
Advantages |
Limitations |
| Combination Oral Contraceptives |
45% |
Ease of use, familiarity |
Side effects, daily compliance |
| Progestin-only Pills |
10% |
Suitable for certain populations |
Slightly less effective |
| Devices (IUDs, Implants) |
20% |
Long-term efficacy |
Insertions require medical procedure |
| Natural Family Planning |
10% |
No chemicals |
Less reliable, user-dependent |
| Others |
15% |
Niche options |
Limited market penetration |
Implications: ORTHO TRI-CYCLEN LO faces competition not only from generics but also from LARCs, which are gaining favor.
6. Strategic Outlook
| Strategic Area |
Focus Points |
Potential Actions |
| Market Penetration |
Emerging markets, targeted marketing |
Local partnerships |
| Product Innovation |
Additional benefits, reduced side effects |
R&D investments |
| Pricing Strategies |
Competitive pricing, value-based models |
Cost optimization |
| Regulatory Navigation |
proactive compliance |
Engagement with authorities |
| Portfolio Diversification |
Adjunct health benefits |
Develop combination products |
7. Key Takeaways
- Moderate growth trajectory: Johnson & Johnson’s ORTHO TRI-CYCLEN LO is projected to grow at approximately 3.2% CAGR from 2023 to 2027, driven by market expansion and product innovation, especially in emerging markets.
- Pricing and patent challenges: Patent expiries and intensifying generic competition pressure margins; strategic pricing and patent protections are critical.
- Market shift towards LARCs: Increasing preference for long-acting contraceptives may limit oral contraceptive market share, requiring product diversification.
- Regulatory risks: Post-market surveillance and compliance remain pivotal, particularly amid evolving policies.
- Competitiveness: Maintaining brand loyalty and differentiating through added health benefits are key to sustaining revenues.
8. FAQs
Q1: What is the main revenue driver for ORTHO TRI-CYCLEN LO?
A: The primary driver is the global demand for safe, effective oral contraceptives in developed and emerging markets, with growth fueled by increasing awareness and healthcare infrastructure expansion.
Q2: How does patent expiry impact the financial trajectory?
A: Patent expiry enables generic manufacturers to enter the market, leading to significant price reductions and revenue erosion for the branded product, necessitating innovation and market expansion strategies.
Q3: What are the key regulatory considerations for ORTHO TRI-CYCLEN LO?
A: Compliance with FDA and EMA regulations regarding safety, labeling, post-marketing surveillance, and potential biosimilar entries influence market access and revenue.
Q4: How competitive is the oral contraceptive market?
A: It is highly competitive, with large pharmaceutical companies holding significant market shares, facing challenges from generics, alternative contraceptive methods, and shifting consumer preferences.
Q5: What strategies should Johnson & Johnson pursue to maintain a competitive advantage?
A: Focus on innovation, expansion into underserved markets, strategic pricing, product diversification, and robust regulatory engagement.
References
- Grand View Research, "Oral Contraceptives Market Size, Share & Trends Analysis Report," 2022.
- Johnson & Johnson Annual Reports, 2018–2022.
- U.S. Food and Drug Administration (FDA), Drug Approvals and Labeling Database.
- European Medicines Agency (EMA), Contraceptive Market Regulations, 2022.
- World Health Organization (WHO), Family Planning Global Statistics, 2022.