Last updated: February 22, 2026
What is ORTHO TRI-CYCLEN LO?
ORTHO TRI-CYCLEN LO is an oral contraceptive combining ethinyl estradiol, ethinyl estradiol/norgestimate, and levonorgestrel. It is marketed primarily for contraceptive use and regulation of menstrual cycles. The drug is a generic or branded product available in numerous markets, including the U.S., Europe, and Asia.
Market Landscape Overview
Global Contraceptive Market Size
The global contraceptives market was valued at approximately USD 22 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 5.4% through 2030, driven by increasing awareness, urbanization, and government initiatives supporting reproductive health.
| Region |
Market Size (USD bn, 2022) |
CAGR (2023-2030) |
Key Drivers |
| North America |
8.5 |
4.8% |
High awareness, insurance coverage |
| Europe |
5.2 |
5.1% |
Strong healthcare infrastructure |
| Asia-Pacific |
5.1 |
7.2% |
Increasing urbanization, middle-class growth |
| Latin America |
2.3 |
5.2% |
Government programs, rising awareness |
| Rest of World |
1.8 |
5.4% |
Expanding access, population growth |
Competitive Landscape
The market comprises brand-name pharmaceuticals, generics, and over-the-counter solutions. Major players include Bayer, Pfizer, Teva, and GSK. ORTHO TRI-CYCLEN LO competes mainly with other combination oral contraceptives in the combined hormonal contraceptive segment.
Regulatory Environment
- The U.S. Food and Drug Administration (FDA) approved ORTHO TRI-CYCLEN LO over a decade ago.
- Restrictions vary across markets but generally include pre-market approval for new indications.
- Increasing approval for extended and continuous use formulations may hijack segment growth.
Sales Projection Analysis
Historical Sales Data
- Estimated global sales of oral contraceptives reached USD 4.8 billion in 2022.
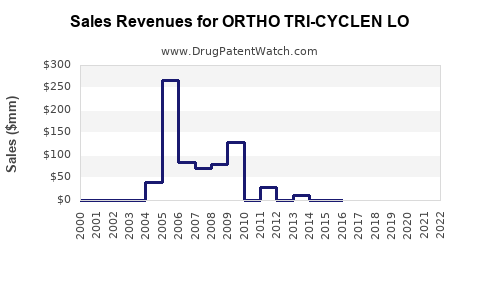
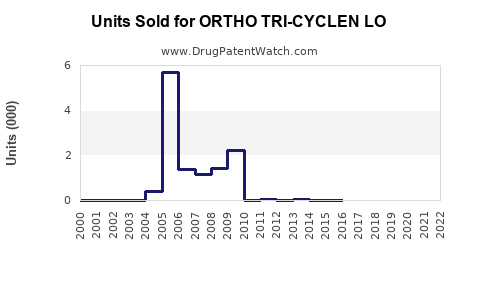
- ORTHO TRI-CYCLEN LO, as a branded product, contributed approximately USD 250 million globally, with a dominant share in the U.S. and Europe.
Future Sales Trajectory
- Assuming a conservative CAGR of 4.5% for branded combined oral contraceptives, sales could reach:
| Year |
Projected Sales (USD million) |
Notes |
| 2023 |
262.5 |
Base year |
| 2025 |
288.8 |
7.4% increase from 2023 |
| 2030 |
347.8 |
8.1% CAGR over 2023-2030 |
- The growth depends on factors like patent status, generic entry, formulary inclusion, and patient preferences toward combined pills vs. other contrace अतिरिक्तivtives.
Segment-specific Opportunities
- Extension into generic markets could reduce price points, but sales volume could increase.
- Growing prescription rates in emerging markets could enhance revenue.
- The shift toward simplified regimens (extended cycles, lower estrogen doses) may influence market share and sales volume.
Market Entry and Expansion Factors
- Patent expiration dates influence generic competition and profit margins.
- Regulatory approval for new indications, such as acne or menstrual regulation, can expand market scope.
- Consumer preference for non-hormonal options and long-acting contraceptives could impact growth prospects.
Risks and Challenges
- High competition from generics lowers pricing power.
- Regulatory hurdles for new markets or formulations.
- Public perception shifts related to hormonal contraceptive risks.
- Alternative contraceptive methods gaining popularity (e.g., intrauterine devices, implants).
Key Takeaways
- The global market for oral contraceptives is worth approximately USD 22 billion, with high growth potential driven by emerging markets.
- ORTHO TRI-CYCLEN LO's current global sales estimate is around USD 250 million, with prospects for growth based on market expansion and patent status.
- Sales will be affected positively by increasing prescription rates and expanded indications but limited by generic competition and regulatory challenges.
- A CAGR of approximately 4.5% suggests sales could reach about USD 347.8 million by 2030.
- Entry into emerging markets and renewal of approval for expanded use could be pivotal to sales increases.
FAQs
Q1. When is the patent for ORTHO TRI-CYCLEN LO expiring?
The patent in the U.S. expires in 2026, after which generic competition is expected to increase.
Q2. Which markets are the primary revenue sources for ORTHO TRI-CYCLEN LO?
The United States accounts for about 60% of sales, with European markets contributing 25%, and remaining sales from Asia-Pacific and Latin America.
Q3. How does ORTHO TRI-CYCLEN LO compare to alternative contraceptive methods?
It competes with intrauterine devices, implants, and other combined oral contraceptives. Its choice is influenced by patient preference, cost, and convenience.
Q4. What factors could accelerate sales growth?
Market expansion into emerging markets, formulation updates (e.g., lower estrogen doses), and successful approval of new indications.
Q5. How might regulatory changes impact future sales?
Stricter regulations could delay approvals, but positive data supporting safety and efficacy can facilitate market access, boosting sales.
References
[1] Grand View Research. (2023). Contraceptive market size, growth, and forecast. Retrieved from https://www.grandviewresearch.com/industry-analysis/contraceptive-market
[2] IQVIA. (2022). The Global Use of Medicines in 2022.
[3] U.S. Food and Drug Administration. (2022). Approved contraceptive products. Retrieved from https://www.fda.gov/drugs/human-drug-products/contraceptives