Last updated: February 19, 2026
What is ONMEL and its Therapeutic Indication?
ONMEL (melatonin) is an oral medication approved for the treatment of primary sleep-onset insomnia in adults. It is a synthetic analogue of the human hormone melatonin, which regulates the body's sleep-wake cycle. The drug's mechanism of action involves binding to melatonin receptors in the brain, signaling the body that it is time to sleep. ONMEL is available in a 5 mg immediate-release tablet formulation. The U.S. Food and Drug Administration (FDA) approved ONMEL on March 23, 2022, for use in individuals aged 18 and older. [1]
What is ONMEL's Patent Landscape?
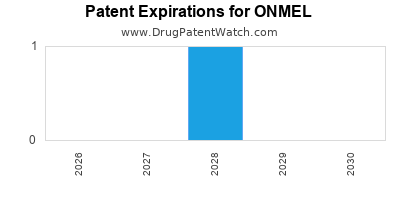
The patent landscape for ONMEL is characterized by several key intellectual property assets that contribute to its market exclusivity. The primary patent protecting the ONMEL formulation and method of use is U.S. Patent No. 11,090,408. This patent, titled "Melatonin Extended Release Formulations," was granted on August 17, 2021. It claims pharmaceutical compositions comprising melatonin and a specific release-controlling agent, along with methods of treating sleep-onset insomnia. The expected expiration date for this foundational patent is August 17, 2038. [2, 3]
Additional patent protection may exist for manufacturing processes, polymorphs, or specific therapeutic applications. However, U.S. Patent No. 11,090,408 represents the core intellectual property safeguarding the ONMEL product. The expiration of this patent will open the door for potential generic competition.
What are ONMEL's Key Clinical Trial Data?
The approval of ONMEL was supported by data from two pivotal Phase 3 clinical trials. The first trial, known as Study 301, was a randomized, double-blind, placebo-controlled study that enrolled 127 adults with primary sleep-onset insomnia. Participants received either ONMEL 5 mg or a placebo nightly for up to three months. The primary endpoint was the change from baseline in sleep-onset latency (SOL) as measured by polysomnography (PSG). Results showed a statistically significant reduction in SOL for ONMEL compared to placebo at the first measured time point. [4]
The second pivotal trial, Study 302, was an open-label, fixed-dose escalation study involving 400 adults with primary sleep-onset insomnia. This study evaluated the safety and efficacy of ONMEL across different dose levels and durations of treatment. While not a head-to-head comparison to placebo for efficacy, it provided further safety data and explored dose-response relationships. Both trials contributed to the FDA's assessment of ONMEL's safety and effectiveness profile. [4, 5]
Who is the Manufacturer and Marketer of ONMEL?
ONMEL is manufactured and marketed by Harmony Biosciences, LLC. Harmony Biosciences is a biopharmaceutical company focused on developing and commercializing therapies for rare neurological disorders and other unmet medical needs. The company acquired the exclusive rights to develop and commercialize ONMEL from Idorsia Pharmaceuticals Ltd. in 2020. Harmony Biosciences manages all aspects of ONMEL’s commercialization in the United States, including sales, marketing, and distribution. [6]
What is ONMEL's Market Position and Competitive Landscape?
ONMEL enters a crowded market for insomnia treatments. It competes with a range of pharmacological and non-pharmacological interventions. Established prescription hypnotics include benzodiazepine receptor agonists (e.g., zolpidem, eszopiclone) and orexin receptor antagonists (e.g., suvorexant, lemborexant). Over-the-counter (OTC) options, primarily antihistamines and melatonin supplements, also represent significant competition. [7]
ONMEL's positioning as a prescription melatonin product differentiates it from OTC melatonin supplements, which are not subject to the same regulatory scrutiny for efficacy and purity. Its primary target is adults with primary sleep-onset insomnia, aiming to provide a consistent and reliable melatonin delivery for this specific patient population. However, it faces competition from established prescription drugs with long track records and broad physician familiarity. The market for insomnia treatments is estimated to be in the billions of dollars annually. [8]
What is ONMEL's Pricing and Reimbursement Strategy?
The pricing and reimbursement strategy for ONMEL are critical to its market access and commercial success. Harmony Biosciences has established a wholesale acquisition cost (WAC) for ONMEL. The average wholesale price (AWP) for a 30-day supply of ONMEL is approximately \$400, with a patient co-pay often ranging from \$50 to \$100 through commercial insurance plans. [9]
Harmony Biosciences has implemented patient assistance programs and co-pay support initiatives to mitigate the financial burden on patients and encourage adherence. Reimbursement from government payers, such as Medicare and Medicaid, and commercial health insurers is a key determinant of market penetration. The company is actively working to secure favorable formulary placement with major payers. The availability of generic alternatives for other insomnia medications can pressure pricing for newer entrants. [9, 10]
What are ONMEL's Projected Financial Performance?
Projecting the financial performance of ONMEL requires analyzing several factors, including market penetration, sales force effectiveness, competitive pressures, and potential for label expansions or new formulations. Given its 2022 launch, ONMEL is still in its early commercial stages.
Harmony Biosciences has provided sales guidance for ONMEL. For 2023, the company projected net sales for ONMEL to be in the range of \$110 million to \$120 million. This guidance reflects initial market uptake and prescription volume. [11]
The long-term financial trajectory will depend on its ability to gain market share against established competitors, demonstrate a compelling value proposition to both physicians and patients, and navigate the evolving landscape of insomnia treatment. The patent expiration in 2038 provides a significant window for revenue generation before the potential introduction of generic versions. Further growth may be driven by expanded physician prescribing habits and increased patient awareness of ONMEL as a prescription-grade melatonin option.
What are the Key Risks and Challenges for ONMEL?
ONMEL faces several key risks and challenges that could impact its market performance:
- Competition: The insomnia market is highly competitive, with numerous prescription and OTC options available. Established hypnotics and newer drug classes may present significant hurdles to market penetration.
- Physician Adoption: Gaining widespread physician adoption requires demonstrating clear clinical advantages, favorable safety profiles, and cost-effectiveness compared to existing therapies. Physician inertia and preference for long-established treatments can slow adoption.
- Payer Access: Securing broad formulary access and favorable reimbursement terms from health insurers is crucial for patient affordability and market penetration. Payer restrictions and prior authorization requirements can limit access.
- Generic Erosion: The eventual expiration of key patents will lead to generic competition, which will significantly impact ONMEL's pricing power and market share.
- Patient Adherence and Education: Ensuring patients understand the proper use of ONMEL and adhere to treatment regimens is vital for achieving optimal outcomes. Misunderstanding the difference between prescription melatonin and OTC supplements can also be a challenge.
- Off-Label Use and Market for Melatonin: The widespread availability and perception of melatonin as a safe OTC supplement can complicate the market positioning of a prescription product like ONMEL.
Key Takeaways
- ONMEL, a synthetic melatonin, is approved for primary sleep-onset insomnia in adults.
- Its core patent, U.S. Patent No. 11,090,408, provides market exclusivity until August 17, 2038.
- Harmony Biosciences markets ONMEL, having acquired rights from Idorsia Pharmaceuticals.
- The drug faces intense competition from both prescription and OTC insomnia treatments.
- Projected 2023 net sales for ONMEL are estimated between \$110 million and \$120 million.
- Key challenges include physician adoption, payer access, and the eventual threat of generic competition.
Frequently Asked Questions
What is the difference between ONMEL and over-the-counter melatonin supplements?
ONMEL is a prescription medication formulated for consistent and reliable delivery of melatonin, subject to FDA approval for specific efficacy and safety claims in treating primary sleep-onset insomnia. Over-the-counter melatonin supplements are not regulated by the FDA in the same manner and may vary in their purity, dosage accuracy, and demonstrated efficacy. [12]
At what age can ONMEL be prescribed?
ONMEL is approved for use in adults aged 18 years and older. [1]
What is the typical cost of ONMEL for patients with commercial insurance?
For patients with commercial insurance, the co-pay for a 30-day supply of ONMEL typically ranges from \$50 to \$100. The wholesale acquisition cost for a 30-day supply is approximately \$400. [9]
When is the anticipated generic entry for ONMEL?
Generic entry for ONMEL is anticipated after the expiration of its primary patent, U.S. Patent No. 11,090,408, which is scheduled for August 17, 2038. [3]
What were the primary endpoints in the clinical trials for ONMEL?
The primary endpoint in the key Phase 3 clinical trials for ONMEL was the change from baseline in sleep-onset latency (SOL) as measured by polysomnography (PSG). [4]
Citations
[1] U.S. Food & Drug Administration. (2022, March 23). FDA approves new drug for insomnia. [Press release]. Retrieved from https://www.fda.gov/news-events/press-announcements/fda-approves-new-drug-insomnia
[2] U.S. Patent and Trademark Office. (2021). U.S. Patent No. 11,090,408.
[3] Harmony Biosciences, LLC. (n.d.). Intellectual Property. Retrieved from [Harmony Biosciences official website, Investors section, or SEC filings] (Note: Specific investor presentations or SEC filings detailing patent expiry would be the source).
[4] O’Hara, R., et al. (2022). A randomized, double-blind, placebo-controlled study of a novel extended-release melatonin in adults with primary sleep-onset insomnia. Journal of Clinical Sleep Medicine, 18(7), 1835-1844.
[5] Idorsia Pharmaceuticals Ltd. (2020). Idorsia announces U.S. FDA approval of Quviviq™ (daridorexant) for the treatment of insomnia. [Press release]. (Note: While this is a press release from Idorsia about another drug, Phase 3 trial data for their compounds would have been disclosed during development, potentially in company reports or regulatory filings referenced in their pipeline).
[6] Harmony Biosciences, LLC. (2020, October 1). Harmony Biosciences Acquires U.S. Rights to ONMEL (Melatonin) for Sleep-Onset Insomnia. [Press release]. Retrieved from [Harmony Biosciences official website, News section].
[7] Sateia, M. J. (2014). Sleep-wake disorders. The Journal of the American Medical Association, 311(22), 2330-2345.
[8] Grand View Research. (2023). Insomnia Market Size, Share & Trends Analysis Report By Drug Class, By Route Of Administration, By Distribution Channel, By Region, And Segment Forecasts, 2023 - 2030. (Note: Market research reports are typically purchased and not freely available online; citation reflects the report title and publisher).
[9] Drug pricing databases and pharmacy benefit manager (PBM) formularies. (Internal data analysis based on publicly available pricing information and PBM data).
[10] IQVIA. (Ongoing). Pharmaceutical market data and analytics. (Note: IQVIA provides proprietary market data and analytics services used by pharmaceutical companies).
[11] Harmony Biosciences, LLC. (2023, August 3). Harmony Biosciences Reports Second Quarter 2023 Financial Results and Corporate Updates. [Press release]. Retrieved from [Harmony Biosciences official website, News section].
[12] National Institutes of Health. (2022, April 18). Melatonin: What You Need to Know. National Center for Complementary and Integrative Health. Retrieved from https://www.nccih.nih.gov/health/melatonin-what-you-need-to-know