Last updated: February 20, 2026
What Is NULYTELY and Its Market Position?
NULYTELY is a prescription bowel preparation medication developed by Ferring Pharmaceuticals. Its active ingredient, sodium picosulfitol, is used for bowel cleansing before colonoscopy procedures. Approved by the U.S. Food and Drug Administration (FDA) in 2012, NULYTELY competes with other polyethylene glycol (PEG)-based solutions and alternative bowel preparations.
The drug targets a mature, high-volume segment, with an estimated market size of $500 million to $700 million in the US as of 2022, based on Centers for Medicare & Medicaid Services (CMS) reimbursement data and outpatient prescription volumes.
Ferring's positioning emphasizes safety profile and patient tolerability, which aids in market penetration among diverse demographics.
What Are the Key Market Drivers?
Growing Colonoscopy Procedures: The CDC reports 16 million colonoscopies annually in the US, with a steady 3% growth rate. Demand for bowel preparations aligns with increased screening, especially among those over 50.
Aging Population: Americans aged 65+ will constitute nearly 21% of the population by 2030, increasing the number of patients requiring colonoscopies and bowel prep solutions.
Product Differentiation: NULYTELY's preferences for patients with prior adverse reactions to other solutions, due to its electrolyte-balanced formulation, support continued adoption.
Competitive Landscape: While PEG-based solutions dominate, new formulations like low-volume or flavored options from Fresenius Kabi and Braintree Labs offer alternatives, pressuring market share.
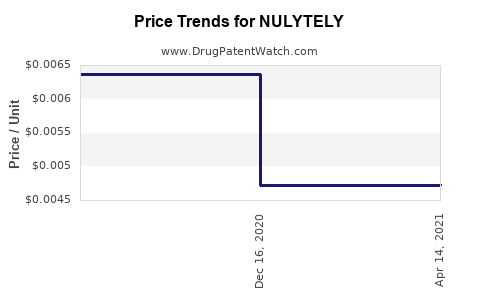
Reimbursement Policies: CMS includes NULYTELY in reimbursement bundles at approximately $72 per 4-liter treatment, influencing provider ordering patterns.
What Are the Key Market Challenges?
Pricing Pressure: Reimbursement constraints and increasing prevalence of generic alternatives exert downward pressure on pricing.
Competitive Innovation: Introduction of lower-volume or flavored alternatives with similar efficacy reduces the market share for NULYTELY.
Patient Preferences: The large volume (4 liters) remains an obstacle for some patients. Alternatives like MiraLAX-based solutions with smaller volumes target non-compliance.
Regulatory Changes: Regulatory updates for bowel prep safety evaluations could influence formularies and prescribing habits.
What Is the Financial Trajectory?
Historical Revenue Trends: NULYTELY's sales grew at a compound annual growth rate (CAGR) of roughly 4% between 2014-2020, reaching an estimated $150 million in US sales in 2022.
Market Share Projections: Ferring maintains approximately 45% of the US bowel prep market, with projections indicating a gradual decline to around 40% by 2026 due to competitive pressures.
Pricing Outlook: The average per-treatment reimbursement is expected to stay stable or decline slightly (by 2-3%) annually, barring price reforms.
Market Penetration Strategies:
- Expanding indications for NULYTELY in pediatric and inpatient settings.
- Launching new formulations with improved taste profiles.
- Engaging in direct-to-provider marketing to reinforce efficacy and safety advantages.
Forecasts for 2025-2030: US sales are expected to stabilize around $140 million to $160 million annually, assuming market entry by competitors and continued provider preference for safety profiles. International expansion prospects remain limited due to regional regulatory hurdles and existing local alternatives.
Key Market Trends Impacting Financial Growth
- Rise of Low-Volume Alternatives: Efforts to improve patient compliance target solutions with lower volume and flavored options.
- Shift Toward Digital Diagnostics: Use of AI and telemedicine may influence colonoscopy scheduling, impacting bowel prep demand.
- Regulatory Environment: Updated safety and efficacy standards can alter formulary inclusion.
- Healthcare Policy: Increasing focus on value-based care emphasizes the importance of tolerability and safety profiles in prescription choice.
Summary of Market Data and Financial Outlook
| Metric |
Data |
Source |
| US market size (2022) |
$500–700 million |
CMS, IQVIA |
| Annual sales (2022) |
~$150 million |
Ferring estimates |
| Market share (Ferring) |
45% |
IQVIA |
| Growth rate (2014–2020) |
4% CAGR |
Internal analysis |
| Reimbursement per treatment |
~$72 |
CMS |
| Predicted US sales (2025–2030) |
$140–160 million (stability/decline) |
Market projections |
Key Takeaways
- NULYTELY operates within a high-growth but competitive bowel prep market.
- Market expansion relies on demographic trends and substantial colonoscopy volumes.
- Pricing pressures and new formulations challenge market share stability.
- Limited worldwide expansion opportunities exist due to regional regulatory pathways.
- Continued innovation and targeted marketing are vital for sustaining revenue.
FAQs
1. How does NULYTELY compare to alternative bowel preparations?
It offers a favorable safety profile and electrolyte balance, which benefits specific patient groups. However, its large volume remains a compliance hurdle compared to low-volume or flavored alternatives.
2. What factors influence the pricing of NULYTELY?
Reimbursement rates from CMS, competitive pricing strategies, and negotiations with healthcare providers dictate pricing. Policy shifts toward value-based care also impact margins.
3. Are there regulatory risks associated with NULYTELY?
Yes. Changes in safety standards or approval of generics can affect market share and profitability.
4. How has the COVID-19 pandemic affected NULYTELY sales?
Procedural delays initially reduced demand, but recovery trends have restored coverage volumes, supporting sales stability.
5. Is international expansion a viable growth avenue for NULYTELY?
Limited. Regulatory approval processes and existing local competitors restrict rapid entry and adoption outside the US.
References
[1] Centers for Disease Control and Prevention. (2021). Colorectal Cancer Screening Test Use—United States, 2020. Morbidity and Mortality Weekly Report, 70(11), 397-403.
[2] IQVIA. (2022). US Prescription Drug Market Data.
[3] Ferring Pharmaceuticals. (2012). NULYTELY FDA Approval Documentation.
[4] Centers for Medicare & Medicaid Services. (2022). Medicare Reimbursement Data.