Last updated: April 26, 2026
NORCO Market Dynamics and Financial Trajectory
What is NORCO and how does the market define its use?
NORCO is a brand of hydrocodone/acetaminophen (opioid analgesic + non-opioid analgesic) used for treatment of pain. In market terms, NORCO sits in the intersection of:
- High-value chronic/pain management purchasing behavior (hospital formularies, outpatient prescribing, PBM controls)
- Strict opioid access and utilization management (state prescribing oversight, payer restrictions, dose caps, step edits)
- Label- and abuse-deterrence-driven competitive filtering (how payers and providers route patients to specific opioid products)
This category’s commercial trajectory is largely determined by utilization, payer policy, and litigation/legal and regulatory overhang, rather than by incremental clinical differentiation.
How do regulations and payer policy shape demand?
Opioid formulary access is constrained through:
- Prior authorization, step therapy, and quantity limits applied at PBM and plan level
- Prescriber reporting requirements and controlled-substance scheduling
- Risk-mitigation requirements and tighter monitoring in many jurisdictions
For hydrocodone/acetaminophen products, payer access is also influenced by broader opioid risk frameworks that favor:
- Lower-risk formulations or products with less favorable abuse-risk profiles (depending on plan rules)
- Non-opioid alternatives when clinically appropriate
- Shorter initial supply and narrower dose ranges, which compress realized demand for any single brand
In this environment, NORCO’s market share is typically less about therapeutic advantage and more about contracting and access.
What are NORCO’s core competitive dynamics?
NORCO competes inside the hydrocodone/acetaminophen class and more broadly in opioid analgesics. Competitive pressure comes from four vectors:
| Competitive vector |
Mechanism |
Typical impact on brand economics |
| Class substitution |
Prescribers switch between hydrocodone/acetaminophen SKUs and strengths |
Share erosion and payer-mandated routing |
| Abuse-deterrent and formulary routing |
Plans prefer products aligned with their risk policies |
Reduced access or narrower covered populations |
| Generics |
Therapeutic equivalence drives price pressure after exclusivity windows |
Gross-to-net deterioration via rebates/concessions |
| Litigation/regulatory headlines |
Market access and investor sentiment shift toward “safer” commercial paths |
Higher cost of doing business; contracting volatility |
This competitive mix means NORCO’s financial trajectory depends on how successfully it maintains access versus generic substitution and how it manages net price under contracting pressure.
How does NORCO’s financial trajectory typically evolve through the product life cycle?
Across opioid analgesics that move from branded dominance into heavy generic penetration, the financial path usually follows:
- Early branded growth: higher gross revenues from broader access and fewer generic substitutes
- Mid-cycle contracting: PBM and large-account contracting tighten; rebates increase
- Late-cycle compression: realized net revenue declines as generics gain share
- Ongoing volatility: risk management policies and litigation exposure can alter formulary standing and discount requirements
NORCO is structurally exposed to this pattern because hydrocodone/acetaminophen is a mature category where generic competition is the norm. In such markets, “growth” tends to be offset by:
- Share loss to generics
- Higher rebate intensity
- Lower average selling price (ASP)
- Working-capital and compliance costs
What demand signals matter most for NORCO’s sales performance?
For hydrocodone/acetaminophen brands, the demand engine is not only prescriptions. Key leading indicators include:
| Indicator |
Why it matters |
What it changes |
| Covered lives and formulary tier placement |
Determines how often patients can access without extra steps |
Access volume and claim conversion |
| PBM contracting terms |
Drives gross-to-net and realized price |
Margin compression or stabilization |
| Prescription quantity limits |
Controls daily dose supply and refills |
Units and refill rate |
| Switching behavior across strengths |
Patients move to other strengths or alternatives |
Mix and ASP |
| Regulatory enforcement intensity |
Increases monitoring and reduces “low-justification” prescribing |
Utilization and adherence to guidelines |
These factors collectively set the unit and net revenue trajectory.
What does “financial trajectory” look like in this segment?
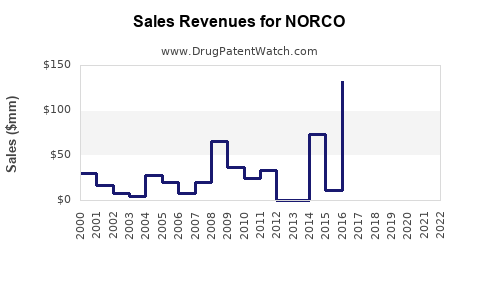
In opioid analgesic markets, company-reported revenue trajectories for branded products typically show:
- Sharp decline after generic substitution
- Lower but more stable decline rates once access stabilizes under established PBM contracts
- Margin pressure from gross-to-net as competition intensifies
- Event-driven volatility tied to litigation headlines, regulatory action, and formulary updates
Because NORCO competes in a heavily scrutinized and legally sensitive category, financials can also swing due to changing payer rules and contracting strategies even when underlying pain prevalence remains steady.
How do litigation and regulatory risk translate into commercial outcomes?
Opioid market participants price and manage legal/regulatory exposure through:
- Revised forecast assumptions for utilization
- Higher commercial compliance and monitoring costs
- Contracting adjustments to reduce execution risk under tight access rules
- Concentration risk management (avoiding overreliance on one opioid product line)
Commercial outcomes flow through in two ways:
- Access changes (formularies may shift away from specific branded products)
- Economic changes (rebates and discounts rise to maintain volume)
For NORCO, this translates into a financial trajectory that is often defined by how much net revenue remains after gross-to-net and share effects.
What is the investment-relevant view of market dynamics for NORCO?
From a business and investment standpoint, NORCO’s market dynamics map to three decision variables:
| Decision variable |
What to monitor |
What “good” looks like |
| Formulary retention |
Tier placement, PA/step edit additions, quantity-limit changes |
Stable or improved access and fewer restrictions |
| Net price resilience |
Rebates intensity and ASP trajectory |
Smaller gross-to-net deterioration |
| Competitive substitution |
Generic share movement and switch rate |
Slower loss of prescriptions and mix stabilization |
In this category, “trajectory” is usually a function of access retention and net price defense more than market expansion.
How does NORCO’s category positioning affect timing and durability?
Hydrocodone/acetaminophen durability is typically limited by:
- Generic substitution
- Evolving opioid prescribing guidelines
- Payer utilization management
As a result, even if NORCO holds share for a period, financial durability depends on whether:
- Net revenue does not keep declining at the same pace as unit demand
- Mix holds up through strength-specific prescribing patterns
- Contracting supports margin despite competition
What are the practical implications for R&D or portfolio strategy?
For portfolio decision-makers, the NORCO pattern implies:
- If the goal is near-term revenue, opioid brands in mature categories rely on access and contracting rather than differentiation
- If the goal is long-term value, the competitive and regulatory headwinds make risk-managed pipeline planning essential
- If the goal is capital allocation, investments should be calibrated to gross-to-net compression and share substitution risk
Key Takeaways
- NORCO operates in a mature opioid analgesic market where demand is driven primarily by formulary access, payer controls, and prescribing constraints, not clinical differentiation.
- Financial trajectory is typically shaped by generic substitution, gross-to-net pressure, and access volatility under strict opioid management policies.
- The most decision-relevant metrics for NORCO’s performance are covered access (tier/PA), net price resilience (rebates/ASP), and switching behavior to generics and alternative opioids.
- Litigation and regulatory risk translate into commercial execution costs and contracting changes, which affect both revenue realization and margin structure.
FAQs
-
What does NORCO compete against most directly?
Hydrocodone/acetaminophen branded equivalents (by strength) and competing opioid analgesics, with substantial substitution to generics driven by therapeutic equivalence and payer policy.
-
What drives NORCO sales more: prescriptions or formulary access?
Formulary and payer access drives claim conversion and refill utilization; prescriptions reflect demand, but access determines whether prescriptions turn into reimbursed volume.
-
Why does generic competition matter disproportionately for opioid brands?
Because it compresses ASP and typically increases rebate intensity, forcing higher incentives to maintain share within payer utilization management frameworks.
-
How do quantity limits and prior authorization affect NORCO’s financials?
They reduce unit velocity (initial fill and refills) and can lower net revenue per covered member even if prescription counts look stable.
-
What should be monitored for early signs of NORCO trajectory change?
PBM tier placement changes, PA/step therapy additions, quantity-limit tightening, and ASP and rebate trend direction.
References
[1] US Food and Drug Administration. “Drug Safety and Availability: Opioid Analgesics.” FDA website.
[2] Centers for Disease Control and Prevention. “CDC Guideline for Prescribing Opioids for Pain.” CDC.
[3] IQVIA Institute for Human Data Science. Market trends and therapeutic category reporting on analgesics and opioid utilization (latest available reports).
[4] Generic and branded opioid reimbursement and formulary management coverage in US managed care publications (PBM formulary and utilization management analyses).