Last updated: February 19, 2026
Nicotrol, a transdermal nicotine replacement therapy (NRT) product, faces a mature market characterized by established competitors and evolving regulatory landscapes. Its financial trajectory is influenced by prescription volume, pricing strategies, and patent expirations.
What is Nicotrol and its Therapeutic Role?
Nicotrol is a transdermal patch system delivering nicotine over a sustained period, designed to aid in smoking cessation. It is indicated for use as an adjunct to behavior change programs for adults attempting to quit cigarette smoking. The patch releases nicotine through the skin, reducing nicotine withdrawal symptoms and cravings experienced by individuals attempting to stop smoking. It is available in varying strengths to accommodate different levels of nicotine dependence.
Who are the Key Market Participants for Nicotrol?
The market for Nicotrol is primarily defined by manufacturers and distributors of NRT products, as well as healthcare providers and regulatory bodies.
- Manufacturer: Perrigo Company plc manufactures Nicotrol.
- Distributors: Pharmaceutical wholesalers and retail pharmacies distribute Nicotrol.
- Competitors: The NRT market includes other transdermal nicotine patches (e.g., Nicoderm CQ), nicotine gums (e.g., Nicorette), lozenges, inhalers, and nasal sprays. Generic versions of Nicotrol also exist, impacting market share.
- Healthcare Providers: Physicians, pharmacists, and smoking cessation counselors play a role in prescribing and recommending Nicotrol.
- Regulatory Bodies: The U.S. Food and Drug Administration (FDA) oversees the approval, manufacturing, and marketing of Nicotrol.
What is the Current Market Size and Growth Outlook for Nicotrol?
The global market for nicotine replacement therapies is substantial, driven by increasing awareness of smoking-related health issues and government initiatives to reduce smoking rates. However, the market for specific products like Nicotrol is subject to intense competition and price pressures.
- Global NRT Market Size: Estimates vary, but the global NRT market was valued at approximately USD 2.5 billion in 2023 and is projected to grow at a compound annual growth rate (CAGR) of 3-5% from 2024 to 2030.
- Nicotrol's Market Share: Specific market share data for Nicotrol is not publicly detailed by Perrigo. However, the transdermal patch segment, a category Nicotrol belongs to, typically accounts for a significant portion of the overall NRT market.
- Growth Drivers:
- Rising prevalence of smoking-related diseases.
- Government anti-smoking campaigns and policies.
- Increasing availability of NRT products, including over-the-counter options.
- Growing adoption of harm reduction strategies.
- Growth Restraints:
- Intense competition from established brands and generic alternatives.
- Pricing pressures and reimbursement challenges.
- Development of alternative cessation methods.
- Stigma associated with nicotine use.
What is the Patent Landscape for Nicotrol?
The patent landscape for pharmaceutical products is critical for understanding market exclusivity and potential generic competition.
- Original Patents: Nicotrol (nicotine transdermal system) was initially developed and patented by Alza Corporation. Key patents have since expired.
- Generic Competition: With the expiration of primary patents, generic versions of nicotine transdermal patches are available in the market. This has significantly impacted the pricing and market share of branded Nicotrol.
- Exclusivity Periods: The period of market exclusivity for Nicotrol has largely ended in major markets due to patent expirations. This allows for the introduction and sale of bioequivalent generic products.
- Perrigo's Role: Perrigo, as the manufacturer, likely operates in a market where its product competes directly with generics, necessitating a focus on cost-efficiency and market access.
How Does Pricing and Reimbursement Affect Nicotrol's Financials?
Pricing and reimbursement policies are direct determinants of Nicotrol's revenue and profitability.
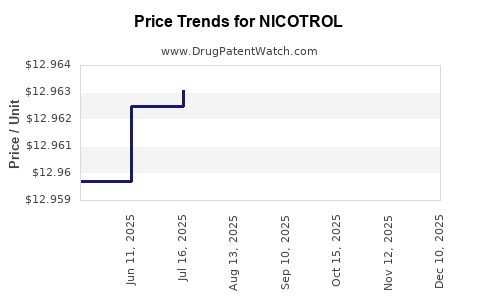
- Wholesale Acquisition Cost (WAC): WAC for Nicotrol varies by strength and pack size. For example, a 21 mg patch can range from $40 to $60 for a 7-day supply.
- Generic Pricing: Generic nicotine patches are typically priced 20-40% lower than branded Nicotrol, creating significant price erosion for the original product.
- Insurance Coverage: Insurance coverage for NRT products, including Nicotrol, is variable. Many commercial insurance plans offer coverage, often with co-pays. Medicare Part D and Medicaid programs also provide coverage, subject to their specific formulary rules and co-payment structures.
- Out-of-Pocket Expenses: For individuals without adequate insurance coverage or with high co-pays, out-of-pocket costs can be a barrier to consistent Nicotrol use.
- Employer Wellness Programs: Some employers offer NRT products as part of their wellness programs, which can influence adoption and reduce out-of-pocket costs for employees.
What are the Key Regulatory Considerations for Nicotrol?
Regulatory frameworks significantly influence the manufacturing, marketing, and accessibility of Nicotrol.
- FDA Approval: Nicotrol is an FDA-approved drug. Perrigo must adhere to Current Good Manufacturing Practices (CGMP) to ensure product quality and safety.
- Over-the-Counter (OTC) Status: Nicotrol is available both by prescription and as an over-the-counter product in the U.S. This dual availability impacts its distribution channels and marketing strategies.
- Labeling and Advertising: All marketing materials and product labeling must comply with FDA regulations, ensuring accurate claims about efficacy and safety.
- Post-Market Surveillance: Perrigo is subject to post-market surveillance requirements, including reporting adverse events.
- International Regulations: Nicotrol's availability and regulatory status vary by country. Manufacturers must comply with the specific requirements of each market in which the product is sold.
What are the Sales and Revenue Trends for Nicotrol?
Detailed, segment-specific revenue figures for Nicotrol are not typically disclosed by Perrigo. However, general trends for NRT products and the impact of generic competition provide insight.
- Declining Branded Sales: As primary patents have expired and generic competition has intensified, branded NRT products often experience a decline in sales volume and revenue.
- Volume vs. Value: While unit sales might remain relatively stable or even grow due to accessibility, the average selling price (ASP) is likely to decrease due to generic pricing pressure.
- Impact of Generic Entry: The introduction of generic nicotine patches typically leads to a significant drop in the ASP of branded products.
- Perrigo's Strategy: Perrigo, as a manufacturer of both branded and generic pharmaceuticals, may leverage its manufacturing capabilities to compete effectively in the generic NRT segment.
What are the R&D and Pipeline Considerations for Nicotrol?
While Nicotrol is an established product, ongoing R&D and pipeline considerations are relevant for its long-term viability.
- Formulation Improvements: There is limited public information on ongoing R&D specifically for Nicotrol by Perrigo. However, general R&D in NRT focuses on:
- Enhanced nicotine delivery systems.
- Improved adherence and user experience.
- Reduced side effects.
- Competitive Pipeline: Competitors are actively developing new NRT products and exploring novel cessation aids. This competitive innovation necessitates continued evaluation of Nicotrol's position.
- Next-Generation Products: The broader NRT market is seeing innovation in delivery systems, including e-cigarettes and heated tobacco products, although these are distinct from traditional NRTs like Nicotrol and face separate regulatory scrutiny.
What are the Key Risks and Opportunities for Nicotrol?
Nicotrol operates within a dynamic market with both significant risks and opportunities.
Risks:
- Intensifying Generic Competition: Continued aggressive pricing by generic manufacturers can further erode market share and profitability.
- Regulatory Scrutiny: Changes in FDA regulations regarding NRT products, including potential reclassification or new labeling requirements, could impact sales.
- Shifting Consumer Preferences: A move towards novel cessation methods or the perception that older NRT methods are less effective could reduce demand.
- Pricing and Reimbursement Pressures: Declining insurance reimbursement rates or increased co-pays could make Nicotrol less accessible for consumers.
- Competition from Other Nicotine Products: The growing market for vaping products, while controversial, may draw some consumers away from traditional NRT.
Opportunities:
- Growing Demand for Smoking Cessation: Increasing global health awareness and public health initiatives continue to drive demand for smoking cessation aids.
- Market Accessibility: Its availability as an OTC product in many regions expands its reach to a broader consumer base.
- Perrigo's Manufacturing Prowess: Perrigo's expertise in generic drug manufacturing allows for cost-effective production, enabling competitive pricing.
- Partnerships and Distribution: Expanding distribution networks and forming partnerships with healthcare providers and smoking cessation programs can enhance market penetration.
- Product Line Extensions: While not publicly indicated for Nicotrol, pharmaceutical companies often explore variations or combinations of existing therapies.
Key Takeaways
Nicotrol, a transdermal nicotine replacement therapy, operates in a mature and highly competitive market. The expiration of its primary patents has led to significant generic competition, impacting pricing and market share. While the broader NRT market benefits from increasing awareness of smoking-related health issues, Nicotrol's financial trajectory is constrained by price erosion and the availability of lower-cost alternatives. Perrigo, as the manufacturer, must leverage its cost-efficient manufacturing capabilities to remain competitive, particularly in the generic segment. Regulatory compliance, evolving reimbursement policies, and shifting consumer preferences for cessation methods represent ongoing challenges and opportunities for the product.
FAQs
-
Is Nicotrol still available in its original branded form, or has it been entirely replaced by generics?
Nicotrol is still available in its branded form, manufactured by Perrigo. However, the market now features numerous generic nicotine transdermal patches that are bioequivalent and often priced lower, significantly impacting the market dynamics for the branded product.
-
What is the typical out-of-pocket cost for a one-week supply of Nicotrol (21 mg)?
The out-of-pocket cost for a one-week supply of Nicotrol (21 mg) can range from approximately $40 to $60, depending on the pharmacy, insurance co-payment, and any available discounts. Generic alternatives are typically less expensive.
-
Does insurance typically cover Nicotrol?
Yes, Nicotrol is generally covered by most commercial insurance plans, Medicare Part D, and Medicaid programs, subject to their specific formulary requirements and co-payment structures. Coverage details can vary significantly by plan.
-
What are the primary advantages of using Nicotrol compared to other NRT methods like gum or lozenges?
The primary advantage of Nicotrol patches is their ability to provide a steady, continuous dose of nicotine over a 16-hour (Nicotrol) or 24-hour (other brands) period, helping to manage cravings and withdrawal symptoms consistently throughout the day. This sustained release can be more convenient for some users compared to on-demand NRTs like gum or lozenges.
-
What is Perrigo's strategic approach to managing Nicotrol in a competitive generic market?
While specific strategies are proprietary, Perrigo, as a major manufacturer of generic and branded over-the-counter pharmaceuticals, likely focuses on cost-efficient production, leveraging its established distribution channels, and maintaining broad market access for both branded and generic Nicotrol products. This involves competitive pricing and ensuring product availability.
Citations
[1] Global Market Insights. (2024). Nicotine Replacement Therapy Market Analysis Report.
[2] Grand View Research. (2023). Nicotine Replacement Therapy Market Size, Share & Trends Analysis Report.
[3] U.S. Food and Drug Administration. (n.d.). Nicotine Products. Retrieved from https://www.fda.gov/tobacco-products/products-guidance-regulation/nicotine-products
[4] Perrigo Company plc. (2023). Annual Report.
[5] IQVIA. (2023). Pharmaceutical Market Data and Analytics. (Proprietary data access required for specific product sales figures).