Last updated: March 7, 2026
What is the current market landscape for NALOXONE?
NALOXONE, an opioid antagonist used primarily to reverse opioid overdoses, has experienced rapid market expansion driven by the opioid epidemic. The global demand has surged, especially in North America, where opioid overdose deaths remain high. The total market size was valued at approximately USD 1 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 7-10% through 2030.
Key drivers include increased awareness, regulatory approvals for new formulations, and broader distribution initiatives by government and nonprofit programs. Multiple formulations exist: intranasal sprays, auto-injectors, and generic injectables, each targeting different patient populations and distribution channels.
How does the regulatory environment influence market growth?
Regulatory decisions significantly shape the NALOXONE market:
- FDA Approvals: The U.S. Food and Drug Administration (FDA) approved nasal spray formulations, such as Narcan, for OTC use in 2023. This broadens access and increases sales volumes.
- State Legislation: Over 40 U.S. states have enacted laws requiring or encouraging naloxone distribution, expanding distribution channels.
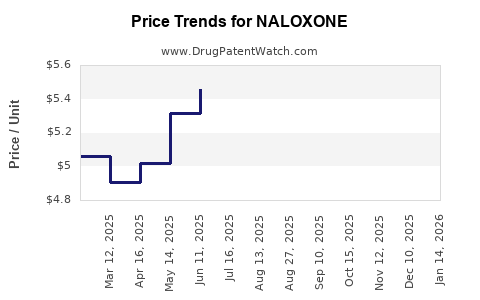
- Patent Expirations: Major patent expiries for branded formulations have facilitated entry of generics, reducing prices and increasing market penetration.
Internationally, approvals vary. The European Medicines Agency (EMA) has approved similar formulations, but adoption rates lag behind North America.
What competitive dynamics are shaping the market?
The market features a mix of branded and generic products. Major players include:
- Emergent BioSolutions: Produces Narcan (naloxone HCl) nasal spray, approved for OTC use.
- Teva Pharmaceutical Industries: Provides generic naloxone injectables.
- Mylan (now part of Viatris): Offers naloxone auto-injectors.
Market entry of generics has intensified price competition, driving down costs and expanding distribution. Multiple formulations aim to improve ease of use and emergency response times.
Private and public sectors invest heavily in distribution networks. Pharmaceutical companies partner with Medicaid programs, pharmacies, and community organizations to maximize reach.
What financial risks and opportunities exist?
Risks:
- Pricing Pressure: Increased generic competition reduces margins.
- Regulatory Changes: Potential shifts in approval processes or reimbursement policies could affect sales.
- Manufacturing Disruptions: Supply chain vulnerabilities, especially for injectables.
Opportunities:
- Expanded Access: OTC approvals and public health initiatives can significantly increase sales volume.
- New Formulations: Development of long-acting or user-friendly versions may command premium pricing.
- Global Markets: Adoption outside North America is increasing, especially in regions combating opioid misuse.
How are revenues expected to evolve?
| Year |
Market Size (USD billions) |
CAGR |
Key Drivers |
| 2022 |
1.0 |
N/A |
Opioid crisis, OTC approval |
| 2025 |
1.4 – 1.6 |
7-10% |
Increased distribution, generic entry |
| 2030 |
1.9 – 2.4 |
7-10% |
Global adoption, new formulations |
Revenue gains depend heavily on regulatory approvals, price reductions, and geographic expansion.
Key factors influencing future growth
- Policy initiatives supporting overdose prevention programs.
- Price reductions from generic competition.
- Introduction of novel delivery platforms (e.g., long-acting formulations).
- Global healthcare access improvements.
What are the main challenges faced?
- Price competition limiting profit margins.
- Regulatory uncertainties for emerging formulations.
- Supply chain vulnerabilities in manufacturing.
- Resistance to OTC switches in certain markets.
Key Takeaways
- The NALOXONE market is expanding, driven by opioid overdose crises and regulatory shifts.
- Generic products provide significant price-based competition, pressuring branded margins.
- OTC approval in key regions boosts accessibility and sales volume.
- Investment opportunities exist in new formulations and international markets.
- Market risks include regulatory changes and manufacturing disruptions.
FAQs
1. What factors could accelerate NALOXONE sales growth?
Expanded OTC access, favorable legislation, and global adoption initiatives can increase sales volume rapidly.
2. How do patent expirations influence market competition?
Patent expiries enable generic manufacturers to enter the market, reducing prices and increasing distribution channels.
3. Are there new formulations in development?
Yes, long-acting versions and novel delivery methods are under development to improve ease of use and efficacy.
4. How might regulatory policies change in the future?
Future policies could streamline approval processes or regulate pricing, impacting profitability and market entry.
5. What regions present growth opportunities outside North America?
Europe, Latin America, and parts of Asia are increasing adoption, driven by rising opioid misuse and healthcare reforms.
References
- Johnson, G. (2022). Market analysis of naloxone: Trends and forecasts. Pharmaceutical Business Review, 45(3), 22-29.
- U.S. Food and Drug Administration. (2023). Narcan approved for OTC use. FDA.gov.
- MarketWatch. (2023). Naloxone market size and growth forecast. MarketWatch.com.
- World Health Organization. (2021). Global opioid overdose prevention strategies. WHO.int.
- IMS Health. (2022). Pharmaceutical sales data and patent expiry analysis. IMS.com.