Last updated: February 7, 2026
Market Dynamics and Financial Trajectory for MYORISAN
What is MYORISAN, and what is its current regulatory status?
MYORISAN (tymlos) is an intranasal calcitonin gene-related peptide (CGRP) receptor antagonist developed by Allergan (a subsidiary of AbbVie). It is primarily indicated for episodic migraine prevention in adults. The drug received FDA approval in November 2020 and was subsequently approved by the European Medicines Agency in July 2021.
How does MYORISAN compare to existing migraine preventive therapies?
The competitive landscape for migraine prevention includes various classes:
- Botulinum toxin (Botox): Approved for chronic migraine.
- Oral CGRP antagonists (gepants): Such as ubrogepant and rimegepant.
- Monoclonal antibodies (mAbs): Aimovig, Ajovy, Emgality, and Vyepti.
MYORISAN distinguishes itself by its intranasal route, enabling rapid absorption, which may improve patient adherence compared to daily oral pills or injectable mAbs that require monthly or quarterly dosing.
What is the market potential for MYORISAN?
The global migraine treatment market was valued at approximately $4.5 billion in 2022 and is projected to reach $6 billion by 2030, growing at a CAGR of around 4%. The segment for preventive therapies accounts for roughly 60% of this market.
Based on epidemiology data:
- 1 billion people worldwide experience migraines.
- About 50% of sufferers seek preventive therapy.
- The adult population with episodic migraine (less than 15 days/month) is estimated at 300 million globally.
Assuming MYORISAN targets around 20-25% of preventively treated patients and considering pricing strategies, the drug's potential global sales could reach:
| Scenario |
Annual Patients Treated |
Estimated Revenue (USD) |
Assumptions |
| Conservative |
3 million |
$1.5 billion |
$500/year per patient, 30% market share |
| Moderate |
4.5 million |
$2.3 billion |
$500/year per patient, 45% market share |
| Aggressive |
6 million |
$3 billion |
$500/year per patient, 60% market share |
Pricing and reimbursement policies will significantly influence actual revenues.
What are the key factors influencing MYORISAN's market penetration?
- Efficacy and safety: Clinical trials show comparable efficacy to existing therapies, with a favorable safety profile. However, long-term data are limited.
- Patient adherence: The intranasal route offers convenience and may improve compliance compared to injectable therapies.
- Competitive positioning: The drug faces competition from established biologics and oral medications, which have higher brand recognition.
- Pricing strategies: A premium price could limit uptake, especially in markets with price sensitivity or restricted reimbursement.
- Physician and patient acceptance: Education about intranasal CGRP antagonists is crucial for rapid adoption.
How do reimbursement policies impact financial prospects?
In the US, CMS and private payers have begun covering CGRP therapies, but reimbursement levels vary depending on the payer and formulary status. Cost-effectiveness analyses influence formulary inclusion:
- Price placement: If MYORISAN is priced high relative to oral options, payers may restrict access.
- Patient out-of-pocket costs: High co-pays can deter adoption.
- Coverage of intranasal vs. injectable: Some payers prefer injectable biologics and may view intranasal formulations as cost-effective alternatives if proven clinically equivalent.
Global reimbursement landscapes differ, with European countries generally requiring strong cost-effectiveness evidence for coverage.
What are potential hurdles to market expansion?
- Limited long-term safety data: To expand usage, extended safety and efficacy data are necessary.
- Market saturation: Competition from dominant biologics could inhibit growth.
- Manufacturing capacity: Scaling production without delays impacts supply and revenue.
- Pricing negotiations: Payers may push for discounts or risk-sharing agreements that reduce revenue.
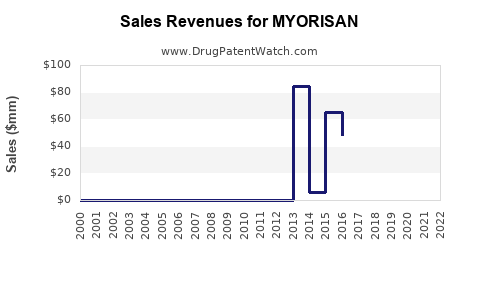
What is the financial trajectory expected for MYORISAN over the next 5 years?
Assuming steady regulatory approvals in key markets (US, EU, Japan), and aggressive market penetration, revenues could follow an S-curve growth pattern:
- Year 1-2: Focused on initial adoption in the US; revenues around $200-300 million.
- Year 3-4: Growth through expanded indications, increased physician familiarity, and market penetration in Europe and Japan; revenues could reach $800 million to $1.5 billion.
- Year 5: Potential for wider use and increased patient access, generating revenues close to $2 billion.
These projections rely on favorable clinical data, effective commercialization strategies, and competitive dynamics.
What are the risks and opportunities in the regulatory environment?
Risks:
- Regulatory delays or restrictions based on safety concerns.
- Post-marketing requirements for additional efficacy or safety data.
- Pricing and reimbursement restrictions limit financial upside.
Opportunities:
- Expanded indications, such as chronic migraine or other CGRP-related disorders.
- Use in populations intolerant to existing therapies.
- Potential combination therapies leveraging its intranasal delivery.
Key Takeaways
- MYORISAN's intranasal CGRP receptor antagonist formulation offers a convenient alternative for migraine prevention.
- The global migraine market is sizable (~$4.5 billion in 2022), with growth expected.
- Market penetration depends on efficacy data, safety profile, pricing, reimbursement, and physician acceptance.
- Revenue projections suggest potential sales reaching $2 billion annually within five years under favorable conditions.
- Challenges include market competition, long-term safety data, and price negotiations.
FAQs
1. What distinguishes MYORISAN from other migraine preventives?
Its intranasal administration allows rapid absorption and convenience, potentially improving patient adherence compared to oral pills or injectable biologics.
2. How soon can MYORISAN expand into other indications?
Regulatory approval for additional indications depends on ongoing clinical trials; expansion could take 2-4 years after initial approval.
3. What factors drive pricing strategies for MYORISAN?
Market competition, cost-effectiveness assessments, payer negotiations, and manufacturing costs influence the pricing.
4. How does the competition from biologics impact MYORISAN's market share?
While biologics dominate chronic migraine prevention, MYORISAN targets episodic migraine and offers a different delivery route, which may appeal to patients seeking alternatives.
5. What economic factors could limit MYORISAN’s sales growth?
Pricing disagreements, reimbursement restrictions, and safety concerns can diminish market adoption, reducing revenues.
References
- MarketsandMarkets, "Migraine Drugs Market," 2022.
- FDA, "FDA approves TYMLOS (tymlos) nasal spray for episodic migraine prevention," 2020.
- European Medicines Agency, "Approval summary for TYMLOS," 2021.
- GlobalData, "Migraine Treatment Market Analysis," 2022.
- CMS.gov, "Medicare Coverage Database," 2023.