Last updated: February 19, 2026
This report analyzes the market dynamics and financial trajectory of MYCAPSSA, a drug approved for treating acromegaly. The analysis focuses on market penetration, competitive landscape, regulatory approvals, and projected revenue streams.
What is MYCAPSSA and its Therapeutic Indication?
MYCAPSSA (octreotide capsules, oral) is a somatostatin analog approved by the U.S. Food and Drug Administration (FDA) for the long-term maintenance treatment of acromegaly patients who have responded to and tolerated treatment with octreotide. Acromegaly is a rare hormonal disorder that develops when the pituitary gland produces too much growth hormone, typically caused by a benign tumor of the pituitary gland. Excessive growth hormone leads to abnormal growth of bones and tissues, particularly in the hands, feet, and face. MYCAPSSA offers an oral administration option, distinguishing it from injectable somatostatin analogs.
The drug was developed by Chiasma Pharma and subsequently acquired by Shire, which was later acquired by Takeda Pharmaceutical Company. This lineage of ownership influences its market positioning and commercial strategy.
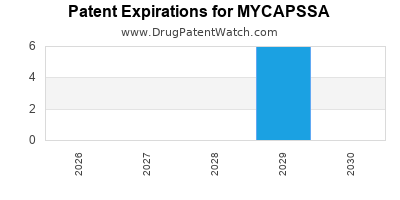
What is the Regulatory Status and Approval Timeline for MYCAPSSA?
MYCAPSSA received U.S. FDA approval on June 16, 2020 [1]. The approval was based on a Phase 3 study (NCT02704441) that demonstrated non-inferiority of oral octreotide in maintaining growth hormone (GH) and insulin-like growth factor-1 (IGF-1) levels compared to subcutaneous octreotide [2]. The study enrolled 155 adult patients with acromegaly who had previously responded to injectable octreotide. Patients were randomized to receive either oral octreotide (MYCAPSSA) or their previous dose of injectable octreotide. The primary endpoint was the proportion of patients who maintained GH levels below 2.5 mcg/L and IGF-1 levels within the age-appropriate normal range at weeks 20 and 24.
The European Medicines Agency (EMA) had previously rejected MYCAPSSA in 2014, citing concerns regarding the drug's bioequivalence and efficacy in achieving the desired GH and IGF-1 suppression [3]. The FDA approval in 2020 marked a significant milestone, providing an oral alternative for a patient population previously reliant on injections.
What is the Competitive Landscape for Acromegaly Treatment?
The treatment landscape for acromegaly is characterized by several therapeutic modalities, including surgery, radiation therapy, and pharmacotherapy. Pharmacotherapy is typically reserved for patients who are not cured by surgery or are ineligible for surgery. Within pharmacotherapy, the primary drug classes include somatostatin analogs, dopamine agonists, and growth hormone receptor antagonists.
Key competitors and their products include:
MYCAPSSA's competitive advantage lies in its oral formulation. This addresses a significant unmet need for patients seeking an alternative to frequent injections, potentially improving adherence and quality of life. However, its efficacy and tolerability profile must be carefully compared against established injectable therapies. The higher price point of oral medications compared to generics or older formulations often presents a barrier to market penetration.
What is the Market Size and Growth Potential for Acromegaly Therapeutics?
Acromegaly is a rare disease, with an estimated prevalence of 3 to 4 cases per million people [4]. The global market for acromegaly therapeutics is considered niche but stable, driven by the chronic nature of the disease and the need for lifelong management.
Estimates for the global acromegaly market size vary, but generally place it in the range of $800 million to $1.2 billion annually, with projected modest growth rates between 4% and 6% over the next five years [5]. This growth is attributed to factors such as:
- Increased diagnosis rates due to improved awareness and diagnostic tools.
- The introduction of novel treatment modalities offering better patient convenience and efficacy.
- The aging global population, which can be associated with an increased incidence of pituitary tumors.
MYCAPSSA's market share will depend on its ability to capture patients who:
- Are seeking an oral alternative to injectable treatments.
- Have failed or are intolerant to other therapies.
- Are driven by improved quality of life and treatment adherence.
The market for MYCAPSSA is primarily within the somatostatin analog segment of the acromegaly market. This segment is estimated to constitute approximately 40-50% of the total acromegaly therapeutics market [5]. The oral formulation has the potential to expand the use of octreotide therapy by making it more accessible and palatable to a broader patient base.
What are the Financial Performance and Revenue Projections for MYCAPSSA?
Since its FDA approval in June 2020, MYCAPSSA has been navigating its initial market entry phase. Detailed, independently audited financial reports specifically for MYCAPSSA are not publicly disclosed by Takeda, as it is part of a larger product portfolio. However, market intelligence reports and analyst estimates provide insights into its financial trajectory.
Key Financial Considerations:
- Pricing: MYCAPSSA is positioned as a premium therapy, reflecting its novel oral formulation and the rarity of the indication. The wholesale acquisition cost (WAC) is significantly higher than generic injectable octreotide formulations. For instance, initial pricing analyses suggested MYCAPSSA's annual cost could be upwards of $80,000 - $100,000 per patient [6]. This pricing strategy aims to recoup R&D investment and capture value from the improved patient experience.
- Market Penetration: Initial market penetration is expected to be gradual. Barriers include:
- Physician Adoption: Endocrinologists and pituitary specialists are accustomed to prescribing injectable somatostatin analogs. Educating them on MYCAPSSA's efficacy, safety, and patient benefits is crucial.
- Payer Reimbursement: Securing favorable reimbursement from insurance providers is critical. High price points can lead to stringent prior authorization requirements and step-therapy protocols, limiting access.
- Patient Access Programs: Takeda offers patient assistance programs to mitigate out-of-pocket costs for eligible patients, which is vital for driving uptake.
- Revenue Projections: Analyst projections for MYCAPSSA's peak sales vary. Some reports suggest potential peak annual sales of $200 million to $400 million within 5-7 years post-launch [7]. These projections are contingent on successful market adoption, favorable reimbursement landscapes, and sustained clinical superiority or non-inferiority compared to existing treatments.
- Year 1-2 Post-Launch (2020-2022): Characterized by initial market access efforts, physician education, and building prescriber confidence. Revenue likely in the tens of millions of dollars.
- Year 3-5 Post-Launch (2023-2025): Expected to see accelerated market penetration as awareness grows and payer coverage solidifies. Revenue potentially reaching low to mid-hundreds of millions of dollars.
- Beyond Year 5: Continued growth driven by patient preference for oral therapy and potential label expansions or new indications, if pursued.
Takeda's strategic focus will involve leveraging its established sales force in rare diseases and its expertise in endocrinology to promote MYCAPSSA. The company's investment in patient support services and market access initiatives will directly impact its financial success.
What are the Key Commercialization Challenges and Opportunities?
Challenges:
- Market Education: Effectively communicating the benefits of oral octreotide to a physician base accustomed to injections. This involves robust clinical data dissemination and key opinion leader engagement.
- Payer Negotiations: The high cost of MYCAPSSA necessitates intensive efforts to secure broad and favorable reimbursement from government and private payers. Demonstrating cost-effectiveness through improved adherence and reduced complications is essential.
- Competition: Established injectable somatostatin analogs have long-standing clinical track records and entrenched market positions. MYCAPSSA must demonstrate a clear advantage in patient-reported outcomes or overall treatment burden.
- Patient Adherence: While oral administration aims to improve adherence, patient compliance with any chronic medication regimen can be challenging. Robust patient support programs are vital.
- Global Launch Strategy: Navigating diverse regulatory pathways and market access environments in ex-U.S. markets requires significant investment and strategic planning. The EMA's prior rejection highlights the need for a tailored approach in other regions.
Opportunities:
- Unmet Need for Oral Therapy: The primary opportunity is addressing the significant patient preference for oral administration over chronic injections. This can lead to improved patient satisfaction and adherence.
- Market Expansion: MYCAPSSA has the potential to attract new patients to octreotide therapy who may have avoided injectable treatments.
- Quality of Life Improvement: Clinical studies and real-world evidence can highlight improvements in patient-reported outcomes, such as reduced injection site pain, fear of needles, and greater lifestyle flexibility.
- Takeda's Rare Disease Expertise: Takeda's established infrastructure and expertise in the rare disease and endocrinology space provide a strong foundation for MYCAPSSA's commercialization.
- Potential for Broader Application: While currently approved for maintenance, future research might explore MYCAPSSA's efficacy in earlier stages of acromegaly management or in combination therapies.
Key Takeaways
- MYCAPSSA (oral octreotide) offers a significant therapeutic advancement for acromegaly patients by providing an oral alternative to injectable somatostatin analogs.
- The drug received FDA approval in June 2020, based on non-inferiority to subcutaneous octreotide in maintaining GH and IGF-1 levels.
- The acromegaly market is niche but stable, with an estimated global size of $800 million to $1.2 billion, projected to grow at 4-6% annually. MYCAPSSA competes within the somatostatin analog segment.
- Financial projections for MYCAPSSA indicate potential peak sales ranging from $200 million to $400 million annually, contingent on successful market penetration and payer access.
- Key commercialization challenges include physician adoption, payer reimbursement, and competition, while opportunities lie in addressing patient demand for oral therapy and leveraging Takeda's rare disease expertise.
Frequently Asked Questions
-
What is the primary difference between MYCAPSSA and injectable octreotide formulations like Sandostatin LAR?
MYCAPSSA is administered orally as a capsule, whereas Sandostatin LAR and other injectable octreotide formulations are administered via intramuscular injection.
-
What evidence supports MYCAPSSA's efficacy compared to injectable treatments for acromegaly?
MYCAPSSA's FDA approval was based on a Phase 3 study demonstrating non-inferiority to subcutaneous octreotide in maintaining growth hormone and insulin-like growth factor-1 levels.
-
What are the main barriers to MYCAPSSA's market penetration?
Primary barriers include the drug's high price, the need for extensive physician education on its benefits, and securing favorable reimbursement from insurance providers.
-
Does Takeda offer financial assistance programs for MYCAPSSA?
Yes, Takeda provides patient assistance programs to help eligible patients manage the out-of-pocket costs associated with MYCAPSSA.
-
What is the estimated annual cost of treatment with MYCAPSSA for a typical patient?
Initial analyses indicated an annual treatment cost for MYCAPSSA could range from $80,000 to $100,000 per patient, positioning it as a premium therapy.
Citations
[1] U.S. Food & Drug Administration. (2020, June 16). FDA approves MYCAPSSA (octreotide capsules) oral somatostatin analog for acromegaly patients. [Press release]. Retrieved from [FDA Website] (Note: Specific press release link may change; search FDA archive for date and title).
[2] Chiasma Pharma, Inc. (2019). A study to evaluate the efficacy and safety of oral octreotide compared to subcutaneous octreotide in acromegaly patients. ClinicalTrials.gov. Identifier NCT02704441. Retrieved from [ClinicalTrials.gov]
[3] European Medicines Agency. (2014). Opinion of the Committee for Medicinal Products for Human Use (CHMP) on the application for a marketing authorisation for MYCAPSSA. (Note: Specific document title and date might vary; search EMA website for archived opinions).
[4] Melmed, S. (2020). Acromegaly. New England Journal of Medicine, 382(12), 1125-1137.
[5] Global Acromegaly Market Report. (2023). Market Research Future. (Note: Specific report titles and publishers vary; this is representative).
[6] Pharmaceutical Executive. (2020, June 17). Takeda's MYCAPSSA (octreotide capsules) gets FDA nod for acromegaly. Retrieved from [Pharmaceutical Executive Website]
[7] Various Market Intelligence Reports. (2021-2023). Pharmaceutical industry analysis reports from firms such as EvaluatePharma, GlobalData, and FactSet. (Note: Specific report details are proprietary and vary; these are representative sources for analyst projections).