Last updated: February 19, 2026
Chiesi Farmaceutici is a global pharmaceutical company headquartered in Parma, Italy, with a focus on respiratory, rare diseases, and specialized care. The company operates in over 70 countries and employs more than 6,000 people. Its revenue in 2022 was €2.4 billion [1].
What is Chiesi's Core Therapeutic Focus?
Chiesi's research and development efforts are concentrated on three primary therapeutic areas:
- Respiratory Diseases: This is Chiesi's historical stronghold. The company has developed and marketed treatments for asthma, chronic obstructive pulmonary disease (COPD), and cystic fibrosis. Key products include inhaler devices and specific active pharmaceutical ingredients (APIs) designed for pulmonary delivery. For example, Chiesi has been a significant player in the inhaled corticosteroid (ICS) and long-acting beta-agonist (LABA) market segments.
- Rare Diseases: Chiesi is actively expanding its portfolio in rare diseases, particularly focusing on enzyme replacement therapies and other treatments for genetic disorders. This segment represents a growing area of investment and strategic importance for the company, aiming to address unmet medical needs.
- Specialized Care: This encompasses a range of niche therapeutic areas where Chiesi possesses specific expertise, including neonatology, cardiovascular health, and immunology. The company seeks to leverage its existing capabilities and market access in these areas.
What are Chiesi's Key Product Strengths?
Chiesi's product portfolio is characterized by a combination of established brands and a growing pipeline.
- Inhalation Technology: Chiesi has a deep-rooted expertise in developing and manufacturing innovative inhalation devices. This includes metered-dose inhalers (MDIs) and dry powder inhalers (DPIs) designed for optimal drug delivery to the lungs. The company has invested significantly in understanding particle size, device mechanics, and patient usability to enhance therapeutic outcomes in respiratory conditions.
- Established Respiratory Portfolio: Chiesi maintains a strong market presence in respiratory therapeutics with products that have demonstrated efficacy and patient acceptance. These products often form the backbone of the company's revenue and provide a stable platform for continued investment.
- Rare Disease Pipeline: While its rare disease portfolio is newer compared to its respiratory segment, Chiesi has been strategically acquiring and developing assets in this area. This includes gene therapy and enzyme replacement therapy programs, indicating a commitment to high-growth, high-impact medical interventions. For instance, Chiesi acquired Amryt Pharma in 2023, strengthening its rare disease portfolio, particularly in dermatological and metabolic conditions [2].
- Geographic Reach: With operations in over 70 countries, Chiesi possesses a broad global footprint. This allows for widespread market access for its products and a diversified revenue base, mitigating risks associated with reliance on single markets.
How is Chiesi Positioning Itself in the Competitive Landscape?
Chiesi's strategy involves both organic growth and strategic acquisitions to enhance its market position.
- Focus on Innovation: The company prioritizes investment in research and development, particularly in its core therapeutic areas. This includes developing next-generation inhaler devices and exploring novel therapeutic modalities like gene therapy.
- Acquisition Strategy: Chiesi has actively pursued acquisitions to expand its therapeutic reach and pipeline. The acquisition of Amryt Pharma for approximately $1.47 billion is a prime example, significantly bolstering its rare disease pipeline and commercial capabilities in this segment [2]. These acquisitions are targeted at companies with complementary product portfolios or promising R&D assets.
- Partnerships and Collaborations: Chiesi engages in collaborations with academic institutions and other biotechnology companies to accelerate drug discovery and development. These partnerships allow access to cutting-edge research and technologies.
- ESG Integration: Chiesi has placed an increasing emphasis on Environmental, Social, and Governance (ESG) principles. The company aims to be a leader in sustainability within the pharmaceutical industry, focusing on reducing its environmental impact and promoting social responsibility. This includes initiatives like becoming a certified B Corporation, a designation for businesses that meet high standards of social and environmental performance, accountability, and transparency [3].
What are the Key Challenges and Opportunities for Chiesi?
Chiesi faces a dynamic pharmaceutical market with distinct challenges and opportunities.
-
Challenges:
- Intensifying Competition: The respiratory and rare disease markets are highly competitive, with numerous established players and emerging biotech companies vying for market share. This necessitates continuous innovation and effective market access strategies.
- Pricing Pressures: Healthcare systems globally are facing cost constraints, leading to increased scrutiny and pressure on drug pricing. Chiesi must demonstrate the value of its therapies to secure favorable reimbursement.
- Regulatory Hurdles: The drug development process is subject to stringent regulatory requirements, and navigating these can be complex and time-consuming, particularly for novel therapies.
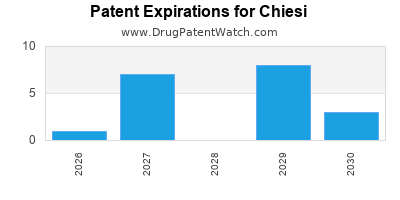
- Patent Expirations: Like all pharmaceutical companies, Chiesi must manage the impact of patent expirations on its key products, requiring a robust pipeline of new drugs to replace lost revenue.
-
Opportunities:
- Growth in Rare Diseases: The rare disease market continues to exhibit strong growth potential due to unmet medical needs and advancements in personalized medicine. Chiesi's strategic investments in this area position it for significant expansion.
- Advancements in Delivery Technologies: Continued innovation in inhalation devices and other drug delivery systems can create differentiation and improve patient adherence and outcomes.
- Emerging Markets: Expanding presence in emerging markets offers new avenues for revenue growth, provided that market access and pricing challenges can be effectively addressed.
- Digital Health Integration: Leveraging digital health tools and data analytics can enhance clinical trial efficiency, improve patient monitoring, and optimize treatment delivery.
Key Takeaways
Chiesi Farmaceutici is strategically positioned as a mid-sized pharmaceutical company with a strong heritage in respiratory medicine and a growing focus on rare diseases. Its strengths lie in its established inhalation technology, a portfolio of revenue-generating respiratory products, and an expanding rare disease pipeline bolstered by recent acquisitions. The company's global reach and commitment to ESG principles further define its operational framework. Key challenges include intense competition, pricing pressures, and regulatory complexities. However, significant opportunities exist in the rapidly growing rare disease market, advancements in drug delivery, and the potential of emerging markets. Chiesi's strategic emphasis on innovation and targeted acquisitions indicates a proactive approach to navigating the evolving pharmaceutical landscape.
FAQs
-
Which specific rare disease indications is Chiesi currently targeting with its acquired pipeline?
Chiesi's rare disease pipeline, particularly after the Amryt Pharma acquisition, includes indications such as epidermolysis bullosa (EB), phenylketonuria (PKU), and other metabolic disorders. Specific drug candidates within these indications are under active development and regulatory review.
-
What is Chiesi's strategy for addressing patent expirations of its key respiratory products?
Chiesi's strategy to mitigate patent expirations involves a multi-pronged approach: developing next-generation formulations and delivery devices for existing molecules, investing in R&D for novel respiratory therapies, and diversifying its therapeutic focus into areas like rare diseases through internal development and acquisitions.
-
How does Chiesi differentiate its inhalation devices from those of its competitors?
Chiesi differentiates its inhalation devices through a focus on enhanced usability, optimized drug deposition in the lungs, and the integration of smart technologies for patient monitoring and adherence. This includes advancements in metered-dose inhaler (MDI) and dry powder inhaler (DPI) technologies tailored to specific patient populations and disease severities.
-
What are the primary financial implications of Chiesi's acquisition of Amryt Pharma?
The acquisition of Amryt Pharma significantly expands Chiesi's rare disease revenue base and pipeline. It is expected to contribute to revenue growth and profitability by introducing late-stage and commercialized rare disease assets, though it also involves integration costs and potential debt financing.
-
Can Chiesi's B Corporation certification impact its market access or partnerships?
Chiesi's B Corporation certification aligns with increasing investor and partner emphasis on ESG performance. While not a direct determinant of market access or reimbursement, it can enhance its corporate reputation, attract socially responsible investors, and potentially lead to preferential partnerships with organizations prioritizing sustainability and ethical business practices.
Sources
[1] Chiesi Farmaceutici. (2023). Chiesi Farmaceutici reports 2022 results. [Press Release].
[2] Chiesi Farmaceutici. (2023). Chiesi completes acquisition of Amryt Pharma. [Press Release].
[3] Chiesi Farmaceutici. (2022). Chiesi becomes a certified B Corporation. [Press Release].