MONOFERRIC Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Monoferric, and when can generic versions of Monoferric launch?
Monoferric is a drug marketed by Pharmacosmos and is included in one NDA. There are five patents protecting this drug.
This drug has sixty-three patent family members in twenty-five countries.
The generic ingredient in MONOFERRIC is ferric derisomaltose. There are twenty drug master file entries for this compound. One supplier is listed for this compound. Additional details are available on the ferric derisomaltose profile page.
DrugPatentWatch® Generic Entry Outlook for Monoferric
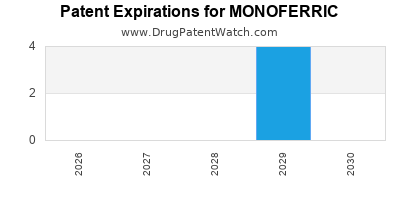
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be March 25, 2029. This may change due to patent challenges or generic licensing.
There has been one patent litigation case involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
Indicators of Generic Entry
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for MONOFERRIC?
- What are the global sales for MONOFERRIC?
- What is Average Wholesale Price for MONOFERRIC?
Summary for MONOFERRIC
| International Patents: | 63 |
| US Patents: | 5 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Clinical Trials: | 11 |
| Drug Prices: | Drug price information for MONOFERRIC |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for MONOFERRIC |
| What excipients (inactive ingredients) are in MONOFERRIC? | MONOFERRIC excipients list |
| DailyMed Link: | MONOFERRIC at DailyMed |
DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for MONOFERRIC
Generic Entry Date for MONOFERRIC*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
SOLUTION;INTRAVENOUS |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for MONOFERRIC
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| University of Saskatchewan | PHASE3 |
| Oregon Health and Science University | Early Phase 1 |
| Alberta Health Services, Calgary | Phase 3 |
US Patents and Regulatory Information for MONOFERRIC
MONOFERRIC is protected by five US patents.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of MONOFERRIC is ⤷ Start Trial.
This potential generic entry date is based on patent 12,030,962.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Pharmacosmos | MONOFERRIC | ferric derisomaltose | SOLUTION;INTRAVENOUS | 208171-003 | Jan 16, 2020 | RX | Yes | Yes | 11,633,489 | ⤷ Start Trial | ⤷ Start Trial | ||||
| Pharmacosmos | MONOFERRIC | ferric derisomaltose | SOLUTION;INTRAVENOUS | 208171-001 | Jan 16, 2020 | DISCN | Yes | No | 11,633,489 | ⤷ Start Trial | ⤷ Start Trial | ||||
| Pharmacosmos | MONOFERRIC | ferric derisomaltose | SOLUTION;INTRAVENOUS | 208171-003 | Jan 16, 2020 | RX | Yes | Yes | 10,414,831 | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| Pharmacosmos | MONOFERRIC | ferric derisomaltose | SOLUTION;INTRAVENOUS | 208171-002 | Jan 16, 2020 | DISCN | Yes | No | 8,815,301 | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
International Patents for MONOFERRIC
When does loss-of-exclusivity occur for MONOFERRIC?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Australia
Patent: 09342799
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 0924653
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 56580
Estimated Expiration: ⤷ Start Trial
China
Patent: 2361651
Estimated Expiration: ⤷ Start Trial
Patent: 8752395
Estimated Expiration: ⤷ Start Trial
Patent: 9700828
Estimated Expiration: ⤷ Start Trial
Croatia
Patent: 0170230
Estimated Expiration: ⤷ Start Trial
Patent: 0201903
Estimated Expiration: ⤷ Start Trial
Patent: 0250214
Estimated Expiration: ⤷ Start Trial
Cyprus
Patent: 18725
Estimated Expiration: ⤷ Start Trial
Patent: 23651
Estimated Expiration: ⤷ Start Trial
Denmark
Patent: 11053
Estimated Expiration: ⤷ Start Trial
Patent: 56075
Estimated Expiration: ⤷ Start Trial
Eurasian Patent Organization
Patent: 3917
Estimated Expiration: ⤷ Start Trial
Patent: 1190214
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 11053
Estimated Expiration: ⤷ Start Trial
Patent: 56075
Estimated Expiration: ⤷ Start Trial
Patent: 21910
Estimated Expiration: ⤷ Start Trial
Patent: 45137
Estimated Expiration: ⤷ Start Trial
Hong Kong
Patent: 64138
Estimated Expiration: ⤷ Start Trial
Hungary
Patent: 30866
Estimated Expiration: ⤷ Start Trial
Patent: 52617
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 26010
Estimated Expiration: ⤷ Start Trial
Patent: 12521369
Estimated Expiration: ⤷ Start Trial
Lithuania
Patent: 11053
Estimated Expiration: ⤷ Start Trial
Patent: 56075
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 11009936
Patent: UN COMPUESTO DE OLIGOSACARIDO DE HIERRO ESTABLE. (A STABLE IRON OLIGOSACCHARIDE COMPOUND.)
Estimated Expiration: ⤷ Start Trial
New Zealand
Patent: 5597
Patent: A STABLE IRON OLIGOSACCHARIDE COMPOUND
Estimated Expiration: ⤷ Start Trial
Philippines
Patent: 015500891
Patent: A STABLE IRON OLIGOSACCHARIDE COMPOUND
Estimated Expiration: ⤷ Start Trial
Poland
Patent: 11053
Estimated Expiration: ⤷ Start Trial
Patent: 56075
Estimated Expiration: ⤷ Start Trial
Patent: 21910
Estimated Expiration: ⤷ Start Trial
Portugal
Patent: 11053
Estimated Expiration: ⤷ Start Trial
Patent: 56075
Estimated Expiration: ⤷ Start Trial
Serbia
Patent: 700
Patent: STABILNO JEDINJENJE GVOŽĐA I OLIGOSAHARIDA (A STABLE IRON OLIGOSACCHARIDE COMPOUND)
Estimated Expiration: ⤷ Start Trial
Patent: 104
Patent: OLIGOSAHARID I POSTUPAK NJEGOVE PRIPREME (AN OLIGOSACCHARIDE AND A PROCESS FOR PREPARATION THEREOF)
Estimated Expiration: ⤷ Start Trial
Slovenia
Patent: 11053
Estimated Expiration: ⤷ Start Trial
Patent: 56075
Estimated Expiration: ⤷ Start Trial
South Africa
Patent: 1107431
Patent: A STABLE IRON OLIGOSACCHARIDE COMPOUND
Estimated Expiration: ⤷ Start Trial
South Korea
Patent: 1580348
Estimated Expiration: ⤷ Start Trial
Patent: 120016063
Estimated Expiration: ⤷ Start Trial
Spain
Patent: 17658
Estimated Expiration: ⤷ Start Trial
Patent: 40074
Estimated Expiration: ⤷ Start Trial
Patent: 09818
Estimated Expiration: ⤷ Start Trial
Ukraine
Patent: 3373
Patent: СТАБИЛЬНОЕ СОЕДИНЕНИЕ ФЕРРУМА ОЛИГОСАХАРИДА (STABLE IRON OLIGOSACCHARIDE COMPOUND)
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering MONOFERRIC around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Lithuania | 2411053 | ⤷ Start Trial | |
| China | 109700828 | ⤷ Start Trial | |
| Ukraine | 103373 | СТАБИЛЬНОЕ СОЕДИНЕНИЕ ФЕРРУМА ОЛИГОСАХАРИДА (STABLE IRON OLIGOSACCHARIDE COMPOUND) | ⤷ Start Trial |
| Cyprus | 1123651 | ⤷ Start Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for MONOFERRIC
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2411053 | C02411053/01 | Switzerland | ⤷ Start Trial | PRODUCT NAME: FERRUM (III) UT FERRI ISOMALTOSIDUM; REGISTRATION NO/DATE: SWISSMEDIC-ZULASSUNG 67001 26.08.2019 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Market Dynamics and Financial Trajectory for MONOFERRIC
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.