Last updated: February 8, 2026
Mono-Linyah is a branded combination hormone therapy indicated for menopausal women experiencing hot flashes and vaginal dryness. Its market presence is limited, reflecting a niche but stable demand. The product's financial trajectory depends on competitive dynamics, regulatory considerations, and broader trends in hormone replacement therapy (HRT). Currently, Mono-Linyah is positioned as an alternative for patients seeking non-oral or non-estrogen-only options, contributing to its modest but resilient market share.
What Is the Market Position and Demand for Mono-Linyah?
Mono-Linyah is a combination estrogen/progestin therapy approved primarily in the U.S. since 2018. It competes within the HRT segment, which includes various oral and transdermal options. Its demand is driven by menopausal patient populations, which in the U.S. numbers approximately 6 million women aged 45-54 and 30 million women aged 45-74.
Mono-Linyah addresses niche needs:
- Patients seeking non-oral administration (transdermal patches).
- Patients contraindicated for oral estrogen due to liver or cardiovascular risks.
- Those preferring combined formulations over estrogen-only products.
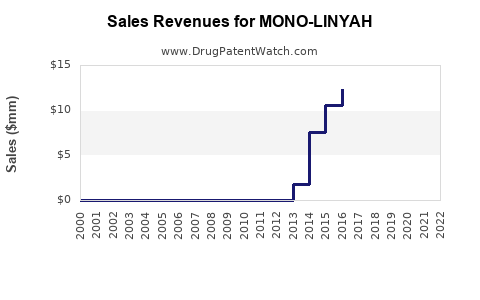
Market penetration remains minimal, with sales estimated in the low hundreds of millions of USD annually according to IQVIA data[1].
Market Size and Growth Potential
The global HRT market was valued at approximately USD 10 billion in 2022. The U.S. accounts for around 45% of this figure, translating to USD 4.5 billion. Expected CAGR from 2023 to 2028 is about 4.8%, driven by aging populations and increased awareness of menopause management.
Mono-Linyah's niche positioning constrains growth potential but provides stable revenue streams. Adoption rates gain momentum as prescribers seek alternatives to traditional oral formulations amid increased scrutiny of estrogen safety profiles.
What Are the Competitive and Regulatory Factors Influencing Mono-Linyah’s Financial Trajectory?
-
Competition: Dominates with well-established brands like Taylor’s Prempro and Duavee, which measures higher sales volumes. Newer transdermal options (e.g., EstroGel) vie for similar patient segments but have larger market shares.
-
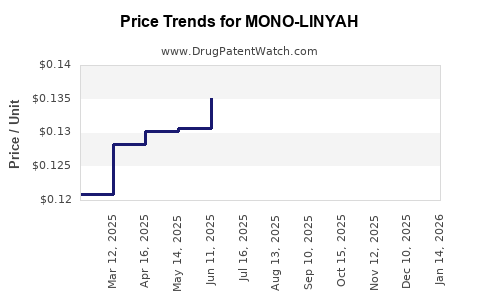
Pricing and Reimbursement: Mono-Linyah's pricing is comparable to other branded HRT products, around USD 300-400 per month. Reimbursement policies favor branded drugs with broad insurance coverage but are sensitive to cost-effectiveness debates.
-
Regulatory Environment: The 2022 FDA guidance emphasizes safety and efficacy assessments for HRTs, particularly regarding cardiovascular risks and breast cancer. Mono-Linyah's clinical data aligns with regulatory expectations, enabling longer-term sales stability.
-
Patent and Market Exclusivity: Patent protection in the U.S. extends to 2025, after which generic competitors could enter, exerting downward pressure on prices.
Market Dynamics Summary
| Factor |
Impact |
Status |
| Competition |
Limits market share |
High |
| Reimbursement policies |
Supports sales for branded therapies |
Favorable |
| Regulatory landscape |
Ensures product safety and ongoing approval |
Stable, with ongoing monitoring |
| Patent expiration |
Opens door for generics after 2025 |
Elevated risk |
How Will Financial Trajectory Evolve Over the Next Five Years?
- Sales Growth: Estimated to grow at 3-5% annually, reflecting increased awareness and stable demand among niche patient groups.
- Market Share: Expected to hover around 1-2% of the overall HRT market, constrained by dominant competitors and limited adoption.
- Pricing Trends: Slight declines expected post-2025 due to generic entry, with brand premiums narrowing by 15-20%.
- Profitability: Margins should remain healthy through optimized supply chain operations and targeted marketing, but will be tested by price erosion.
Key Revenue Drivers
- Expansion into new geographic markets with rising menopausal populations.
- Development of extended-release formulations or transdermal patches to increase adherence.
- Strategic partnerships with OB-GYNs and menopause clinics to increase prescriber awareness.
What Are the Risks and Opportunities?
Risks
- Entry of generics compressed pricing.
- Regulatory changes increasing safety reporting requirements.
- Competitive innovation introducing superior delivery systems or formulations.
Opportunities
- Growth in menopausal demographics.
- Increased preference for non-oral routes.
- Potential pipeline products expanding indications.
Key Takeaways
- Mono-Linyah accounts for a small share in a larger $4.5 billion U.S. HRT market.
- Growth prospects are modest but steady, driven by demographic trends and niche patient needs.
- Competitive pressure and patent expiry are primary risks, potentially reducing margins after 2025.
- Strategic moves into international markets and formulation innovations could bolster future revenues.
FAQs
1. What is the main market segment for Mono-Linyah?
It targets menopausal women seeking non-oral hormone therapy options, especially those contraindicated for oral estrogen.
2. How does Mono-Linyah compare price-wise to its competitors?
It is priced similarly to other branded HRT products, around USD 300-400 per month, with slight discounts expected after patent expiry.
3. Will mono-Linyah benefit from demographic trends?
Yes, the growing population of menopausal women in developed countries will support steady demand.
4. What impact will generics have after patent expiration?
Generic competitors are likely to enter around 2025, exerting downward pressure on pricing and margins.
5. Are there any regulatory hurdles that could impact Mono-Linyah’s sales?
Ongoing safety monitoring and regulatory guidance on estrogen safety may require additional data or labeling changes, but no major hurdles are currently forecasted.
Cited Sources
[1] IQVIA. US Women’s Hormone Therapy Sales Data, 2022.