MIRENA Drug Patent Profile
✉ Email this page to a colleague
When do Mirena patents expire, and what generic alternatives are available?
Mirena is a drug marketed by Bayer Hlthcare and is included in one NDA. There are two patents protecting this drug.
This drug has forty-five patent family members in twenty countries.
The generic ingredient in MIRENA is levonorgestrel. There are twenty drug master file entries for this compound. Thirty-four suppliers are listed for this compound. Additional details are available on the levonorgestrel profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Mirena
A generic version of MIRENA was approved as levonorgestrel by NOVEL LABS INC on February 22nd, 2013.
Summary for MIRENA
| International Patents: | 45 |
| US Patents: | 2 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 104 |
| Clinical Trials: | 69 |
| Patent Applications: | 4,535 |
| Formulation / Manufacturing: | see details |
| Drug Prices: | Drug price information for MIRENA |
| What excipients (inactive ingredients) are in MIRENA? | MIRENA excipients list |
| DailyMed Link: | MIRENA at DailyMed |

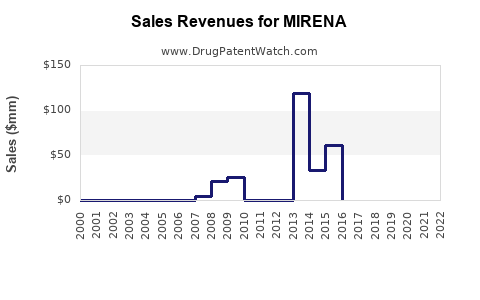
Recent Clinical Trials for MIRENA
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Arkansas Children's Hospital Research Institute | Phase 4 |
| Boston Children's Hospital | Phase 2 |
| Eastern Virginia Medical School | Phase 4 |
Pharmacology for MIRENA
| Drug Class | Progestin Progestin-containing Intrauterine System |
| Physiological Effect | Inhibit Ovum Fertilization |
Anatomical Therapeutic Chemical (ATC) Classes for MIRENA
US Patents and Regulatory Information for MIRENA
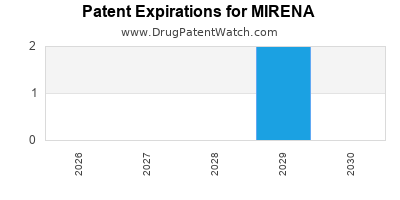
MIRENA is protected by two US patents.
Patents protecting MIRENA
Inserter
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: A METHOD OF POSITIONING AN INTRAUTERINE SYSTEM (IUS) BY DETERMINING A DEPTH OF THE UTERUS, HOLDING AN INSERTER HANDLE WITH ONE HAND, INSERTING THE IUS INTO THE UTERUS, AND RETRACTING A SLIDER ON THE HANDLE TO RELEASE THE IUS INTO THE UTERUS
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: A METHOD OF PREVENTING PREGNANCY BY PROVIDING AN INTRAUTERINE SYSTEM (IUS), HOLDING AN INSERTER HANDLE WITH ONE HAND, INSERTING THE IUS INTO THE UTERUS, AND MOVING A SLIDER IN THE HANDLE TO RELEASE THE IUS WITHIN THE UTERUS
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bayer Hlthcare | MIRENA | levonorgestrel | INTRAUTERINE DEVICE;INTRAUTERINE | 021225-001 | Dec 6, 2000 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Bayer Hlthcare | MIRENA | levonorgestrel | INTRAUTERINE DEVICE;INTRAUTERINE | 021225-001 | Dec 6, 2000 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for MIRENA
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Bayer Hlthcare | MIRENA | levonorgestrel | INTRAUTERINE DEVICE;INTRAUTERINE | 021225-001 | Dec 6, 2000 | ⤷ Try a Trial | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for MIRENA
See the table below for patents covering MIRENA around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Bulgaria | 101744 | ⤷ Try a Trial | |
| South Korea | 100393708 | ⤷ Try a Trial | |
| Japan | 5449366 | ⤷ Try a Trial | |
| South Korea | 101604507 | ⤷ Try a Trial | |
| Mexico | 2011002670 | UN INSERTADOR. (AN INSERTER.) | ⤷ Try a Trial |
| South Korea | 20110083598 | AN INSERTER | ⤷ Try a Trial |
| Denmark | 2349143 | ⤷ Try a Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for MIRENA
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1453521 | C 2015 029 | Romania | ⤷ Try a Trial | PRODUCT NAME: LEVONORGESTREL SI ETINILESTRADIOL; NATIONAL AUTHORISATION NUMBER: RO 7793/2015/001; DATE OF NATIONAL AUTHORISATION: 20150612; NUMBER OF FIRST AUTHORISATION IN EUROPEAN ECONOMIC AREA (EEA): SK. 17/0017/15-S; DATE OF FIRST AUTHORISATION IN EEA: 20150129 |
| 1453521 | C201630040 | Spain | ⤷ Try a Trial | PRODUCT NAME: ETINILESTRADIOL Y MEZCLA DE LEVONORGESTREL Y ETINILESTRADIOL; NATIONAL AUTHORISATION NUMBER: 80340; DATE OF AUTHORISATION: 20160122; NUMBER OF FIRST AUTHORISATION IN EUROPEAN ECONOMIC AREA (EEA): 17/0017/15-S; DATE OF FIRST AUTHORISATION IN EEA: 20150211 |
| 1453521 | 93156 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: LEVONORGESTREL ET ETHINYLESTRADIOL; FIRST REGISTRATION DATE: 20150211 |
| 1453521 | 300814 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: LEVONORGESTREL EN ETHINYLESTRADIOL; NATIONAL REGISTRATION NO/DATE: RVG 117453 20151211; FIRST REGISTRATION: SK 17/0017/15-S 20150211 |
| 1453521 | 122015000093 | Germany | ⤷ Try a Trial | PRODUCT NAME: LEVONORGESTREL UND ETHINYLESTRADIOL; NAT. REGISTRATION NO/DATE: 87675.00.00 20150720; FIRST REGISTRATION: SLOWAKEI 17/0017/15-S 20150129 |
| 1453521 | 132016000025143 | Italy | ⤷ Try a Trial | PRODUCT NAME: LEVONORGESTREL ED ETINILESTRADIOLO(SEASONIQUE); AUTHORISATION NUMBER(S) AND DATE(S): 17/0017/15-S, 20150211;042139016, 20150414 |
| 1453521 | 15C0050 | France | ⤷ Try a Trial | PRODUCT NAME: ETHINYLESTRADIOL ET MELANGE DE LEVONORGESTREL ET ETHINYLESTRADIOL; NAT. REGISTRATION NO/DATE: NL 42237 20150320; FIRST REGISTRATION: SK - 17/0017/15-S 20150129 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |


