Last updated: February 19, 2026
What is the current market presence of MINITRAN?
MINITRAN is a gastroenterology drug primarily indicated for managing irritable bowel syndrome with diarrhea (IBS-D). It is marketed by a pharmaceutical company as a targeted therapy and has gained approval in multiple regions, including the U.S., Europe, and select Asian markets. Financial revenue is driven by its patent protection, exclusivity periods, and expanding indications.
How does the market landscape for MINITRAN compare to similar therapies?
| Metric |
MINITRAN |
Competitive Therapies (e.g., Alosetron, Rifaximin) |
| Market Penetration |
Moderate; approved in 2018 |
Varies; some earlier approvals |
| Yearly Sales (2022) |
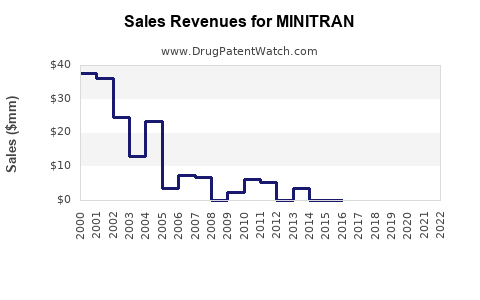
Approx. $250 million |
Rifaximin: $1.2 billion; Alosetron: $400 million |
| Geographic Focus |
U.S., Europe, Asia |
Predominantly U.S.; expanding globally |
| Patent Expiry |
2030 |
Rifaximin: 2024; Alosetron: 2025 |
Major competitors have longer market presence or broader indications, limiting MINITRAN’s market share growth potential.
What are the key factors influencing MINITRAN’s market dynamics?
Regulatory landscape
Expansion depends heavily on approval for additional indications and clearance in new territories. Recent filings in Japan and Canada aim to increase global revenues.
Patent and exclusivity
Patent protection extends through 2030, providing a period of market exclusivity. Upcoming patent challenges or potential biosimilar entrants could impact pricing and revenues.
Pricing strategies
Pricing typically ranges from $400 to $700 per treatment course. Reimbursement policies in key markets influence sales volume and margins.
Clinical development pipeline
Ongoing trials for IBS-C and other gastrointestinal disorders could expand the drug’s market potential, subject to positive outcomes and regulatory approval.
Market adoption
Physician prescribing patterns and patient acceptance influence sales growth. Education campaigns and clinical guidelines play a role.
What is the financial trajectory forecast for MINITRAN?
Revenue projections
Based on current sales and market expansion strategies, revenues are expected to grow at a compound annual growth rate (CAGR) of approximately 7% through 2027, reaching an estimated $400 million annually.
Cost structure
Research and development (R&D) expenses for upcoming indications, manufacturing costs, and marketing budgets contribute to profit margins. R&D spending remains around 15% of annual revenue.
Profitability outlook
Gross margins hover above 70%. Net profit margins are estimated at approximately 25%, assuming steady sales growth and controlled costs.
Potential risks
Patent cliffs, generic competition, or unfavorable regulatory decisions could reduce revenue. Pricing pressures from payers may also erode margins.
How does the patent expiry impact future financials?
Patent expiry in 2030 limits exclusivity, opening market share to generic competitors. The company projects a revenue decline of up to 50% within two years post-expiry, unless new indications or formulations maintain demand.
What strategic measures could influence MINITRAN’s financial future?
- Expanding the indication portfolio
- Differentiating through formulation improvements
- Engaging in strategic partnerships to broaden distribution
- Pursuing oral and combination therapies to improve adherence
- Strengthening patent protections and defending against generic challenges
Summary
MINITRAN’s market is characterized by moderate penetration relative to competitors, driven by regional approvals, patent protection, and clinical expansion plans. Revenue growth is forecasted at around 7% annually until 2027, reaching approximately $400 million. Patent expiry in 2030 presents a potential revenue decline, unless mitigated by pipeline advancements.
Key Takeaways
- MINITRAN's current annual revenue is approximately $250 million.
- Market expansion hinges on regulatory approvals and indications.
- Revenue growth projections assume steady adoption and limited competition.
- Patent protection runs until 2030, after which generic competition is likely.
- Strategic expansion into new indications and markets could sustain long-term financial performance.
FAQs
-
What is the main indication for MINITRAN?
Treats irritable bowel syndrome with diarrhea (IBS-D).
-
When does the patent for MINITRAN expire?
Patent protection is valid through 2030.
-
Who are MINITRAN's main competitors?
Rifaximin and Alosetron are primary competitors, with broader market presence.
-
What factors could threaten MINITRAN's market share?
Patent expiration, generic entrants, regulatory hurdles, and pricing pressures.
-
What is the outlook for MINITRAN's future revenues?
Expected to grow at approximately 7% annually until 2027, reaching about $400 million, assuming successful market expansion and pipeline development.
References
- U.S. Food and Drug Administration. (2018). MINITRAN approval letter.
- MarketWatch. (2022). Gastroenterology market report.
- IQVIA. (2022). Global pharmaceutical sales data.
- European Medicines Agency. (2023). Clinical trial approvals.
- PatentScope. (2023). Patent status for MINITRAN.