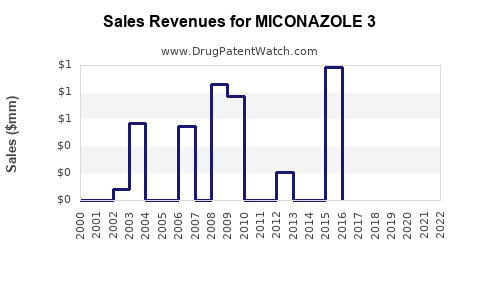
Last updated: August 13, 2025
Introduction
Miconazole 3% emerges as a prominent antifungal agent used primarily for the treatment of superficial fungal infections. Its broad-spectrum activity against dermatophytes, yeasts, and moulds positions it as a key asset within the topical antifungal market. As the pharmaceutical industry navigates evolving regulatory environments, competitive landscapes, and growing consumer demand for effective skin infection remedies, understanding the market dynamics and projecting the financial trajectory of Miconazole 3% is essential for stakeholders.
Market Overview and Clinical Applications
Miconazole 3% is indicated for the treatment of conditions such as tinea corporis, tinea cruris, tinea pedis, and candidiasis. Its efficacy, safety profile, and ease of topical application underpin its widespread acceptance among clinicians and patients. The global antifungal market, valued at approximately USD 11 billion in 2022, is projected to grow at a compound annual growth rate (CAGR) of 4-6% over the next five years, driven by increasing incidences of fungal infections and expanding dermatological healthcare awareness [1].
In particular, the topical antifungal segment is gaining traction owing to advantages like reduced systemic side effects and targeted therapy. Miconazole, being a well-established molecule, enjoys a dominant position within this segment, especially in over-the-counter (OTC) formulations.
Market Dynamics Influencing Miconazole 3%
1. Increasing Prevalence of Fungal Infections
The elevated rate of dermatophyte infections globally, fueled by climate factors, urbanization, and lifestyle changes, bolsters demand for effective antifungal treatments. The World Health Organization estimates that dermatophyte infections affect over 20-25% of the global population at some point in life [2]. This demographic trend sustains steady demand for Miconazole 3%, particularly in emerging markets.
2. Competitive Landscape and Market Penetration
Miconazole faces competition from other topical antifungals such as clotrimazole, terbinafine, and econazole. However, Miconazole’s longstanding clinical reputation, affordability, and availability as an OTC medication confer advantages. In developed markets, formulations combining Miconazole with corticosteroids or additional antifungal agents—e.g., Miconazole-Nitric oxide complexes—are expanding its therapeutic scope.
3. Formulation Innovations and Delivery Mechanisms
Innovations such as gels, creams, sprays, and long-acting formulations improve patient compliance and treatment efficacy. Additionally, nanoparticle-based delivery systems and bioadhesive formulations are under research, potentially enhancing absorption and providing competitive edge.
4. Regulatory and Patent Landscape
Most Miconazole formulations are off-patent, enabling significant OTC availability and price competition. Nonetheless, patent extensions for specific formulations or combination therapies could influence market exclusivity and pricing strategies temporarily, impacting revenue streams.
5. Regulatory Approvals and Market Variability
Regulatory authorities worldwide have approved Miconazole for OTC and prescription use, with some regional restrictions. The approval process for new formulations or combination therapies influences product lifecycle and market entry timing.
6. Impact of the COVID-19 Pandemic
The pandemic’s effects on healthcare utilization have temporarily slowed prescription rates but increased demand for OTC antifungals, especially in private and retail channels. Post-pandemic recovery is expected to restore growth momentum.
Financial Trajectory and Revenue Projections
Current Revenue Status
Existing data indicates that Miconazole-based products generate annual revenues exceeding USD 1 billion globally, with Europe and North America contributing approximately 50% of the revenue share [1]. The majority of these revenues are derived from OTC formulations, which benefit from widespread accessibility.
Growth Drivers and Forecasts
- Market Penetration in Emerging Economies: Rapid urbanization, rising healthcare awareness, and improving healthcare infrastructure facilitate increased OTC sales; forecasts suggest a 7-9% CAGR in emerging markets over 2023-2028.
- Formulation Diversification: The introduction of combination products with corticosteroids and other antifungal agents enhances therapeutic options, prompting premium pricing and expanding per-unit revenue.
- E-commerce and Digital Marketing: Rising online sales channels broaden reach, especially in non-traditional markets, potentially adding USD 200-300 million annually to revenues.
Potential Revenue Impact of Key Factors
| Factor |
Impact |
Estimated Effect |
| Patent expiry of certain formulations |
Price competition increases |
10-15% revenue decline in mature markets |
| New formulation approvals or combination therapies |
Market expansion and premium pricing |
15-25% revenue increase |
| Entry into untapped geographic markets |
Revenue diversification |
USD 100-200 million incremental annually |
| Regulatory restrictions or approval delays |
Market access limitations |
5-10% revenue decrease in affected regions |
Projected Financial Trajectory (2023-2028)
Assuming a blended CAGR of 5-6%, revenues could reach USD 1.5-2 billion globally by 2028. The trajectory depends critically on market penetration strategies, formulation innovations, and competitive responses.
Market Challenges and Risk Factors
- Price Competition and Genericization: As patents expire and generics proliferate, downward pressure on prices could constrain profitability.
- Regulatory Changes: Stricter quality standards and labeling requirements may delay product launches or increase compliance costs.
- Emerging Resistance: Rising resistance among dermatophytes to antifungal agents could diminish Miconazole’s effectiveness, necessitating formulation modifications or combination use.
- Market Saturation: Mature markets may experience saturation, limiting growth potentials unless new formulations or indications are introduced.
Conclusion and Future Outlook
Miconazole 3% maintains a robust position within the topical antifungal segment, driven by a high prevalence of fungal infections and consumer preference for OTC options. Its financial success depends on strategic formulation innovation, effective regulatory navigation, and expansion into underserved markets. Emerging trends such as nanoparticle delivery and combination therapies offer avenues for growth and margin enhancement.
Key Takeaways
- The global antifungal market, valued at over USD 11 billion, is projected to grow steadily, with Miconazole 3% poised to benefit from rising dermatophyte infection rates.
- Market dynamics favor Miconazole due to its established efficacy, affordability, OTC availability, and ongoing innovation.
- Revenue projections indicate a potential rise to USD 1.5-2 billion by 2028, contingent on strategic expansion and product development.
- Challenges include patent expirations, increasing competition, and resistance patterns, which require proactive adaptation.
- Stakeholders should focus on formulation diversification, geographic expansion, and regulatory compliance to sustain growth.
FAQs
1. How does Miconazole 3% compare to other antifungal agents in efficacy?
Miconazole 3% demonstrates comparable effectiveness to other topical antifungals like clotrimazole and terbinafine in treating superficial fungal infections, with a favorable safety profile. Its broad-spectrum activity and extensive clinical history make it a preferred option.
2. What are the main markets driving Miconazole sales?
The United States, Europe, and increasingly, Asia-Pacific, especially China and India, are primary markets driven by high dermatophyte infection prevalence and OTC product consumption.
3. How might formulation innovations impact Miconazole’s market position?
Innovative formulations such as gels, long-acting applications, or combination therapies can enhance patient compliance and treatment efficacy, potentially capturing market share from traditional creams.
4. What role does OTC availability play in Miconazole’s financial performance?
OTC availability broadens consumer access, promotes higher sales volumes, and sustains revenue growth, especially in markets where prescription requirements restrict access.
5. What are the primary risks to Miconazole’s future growth?
Key risks include patent expirations leading to generic competition, evolving resistance patterns, regulatory hurdles, and market saturation in developed regions.
References
[1] MarketWatch. "Global Antifungal Market Size, Share & Trends Analysis Report," 2022.
[2] World Health Organization. "Dermatophyte Infections Fact Sheet," 2021.