Last updated: February 19, 2026
Metadate CD, an extended-release formulation of methylphenidate, is positioned within the growing market for Attention Deficit Hyperactivity Disorder (ADHD) treatments. Its market penetration and financial performance are influenced by patent exclusivity, generic competition, evolving treatment guidelines, and the competitive landscape of stimulant and non-stimulant medications.
What is the patent status of Metadate CD?
Metadate CD's primary patents have expired, opening the door for generic competition. The original patent for the extended-release technology used in Metadate CD likely covered specific formulations and delivery mechanisms. However, as these patents lapse, the market dynamics shift significantly.
- Original Exclusivity: Metadate CD was developed by Celltech Pharmaceuticals (later acquired by UCB Pharma). The initial patent protection provided a period of market exclusivity, allowing for premium pricing and recoupment of R&D investments.
- Patent Expiry Timeline: Specific patent expiration dates are crucial for market analysis. While precise dates for all patents related to Metadate CD are proprietary and vary by region, the general trend for older, established drug formulations like this is that core patents have expired or are nearing expiration in major markets like the United States and Europe.
- Orphan Drug Exclusivity: Metadate CD is not designated as an orphan drug, meaning it did not benefit from the extended market exclusivity periods granted for rare disease treatments.
- Pediatric Exclusivity: In the United States, the U.S. Food and Drug Administration (FDA) may grant an additional six months of market exclusivity for new pediatric studies. The status of whether this was sought or granted for Metadate CD, and its expiration, would impact the timeline of generic entry.
- Ongoing Litigation: Pharmaceutical companies often engage in patent litigation to defend their market position. Litigation surrounding Metadate CD's patents or those of its generic competitors could impact market entry timelines and pricing. Such disputes are typically complex and involve arguments about patent validity, infringement, and potential invalidity defenses.
How has generic competition impacted Metadate CD's market share and pricing?
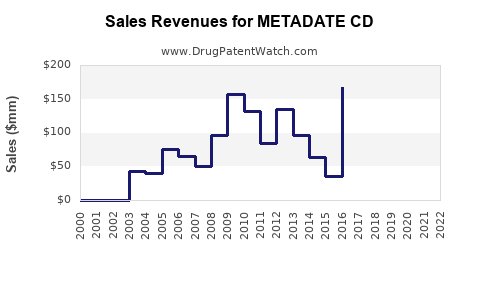
The expiration of Metadate CD's patents has led to the introduction of generic methylphenidate extended-release products, significantly altering its market dynamics.
- Price Erosion: Generic entry typically results in substantial price reductions. The average selling price (ASP) for Metadate CD has declined following the availability of generic alternatives. This price erosion is a standard phenomenon in the pharmaceutical market once patent protection ends.
- Market Share Diversion: Prescriptions for Metadate CD have shifted to lower-cost generic versions. This diversion directly impacts the brand-name drug's market share. While Metadate CD might retain some market share due to physician familiarity or specific patient needs, the majority of volume is likely to transition to generics.
- Reimbursement Landscape: Payer formularies often favor lower-cost generics, further incentivizing their use over branded products. This influence on reimbursement policies accelerates the shift away from branded Metadate CD.
- Market Volume: The overall market volume for extended-release methylphenidate has likely grown due to increased ADHD diagnosis and treatment, and the availability of multiple generic options at lower price points. However, the revenue generated by the branded Metadate CD within this growing market has proportionally decreased.
- Manufacturer Response: The manufacturer of branded Metadate CD may attempt to mitigate losses through strategies such as reformulating the drug, focusing on specific market segments, or engaging in authorized generic partnerships. However, the fundamental impact of generic competition remains significant.
What is the current competitive landscape for ADHD medications?
Metadate CD operates within a highly competitive therapeutic class, facing competition from both stimulant and non-stimulant medications, as well as other extended-release methylphenidate formulations.
- Stimulant Medications:
- Methylphenidate-based: This class includes immediate-release and extended-release formulations.
- Immediate-Release: Ritalin, Methylin.
- Extended-Release (similar to Metadate CD): Concerta (orally disintegrating tablet), Ritalin LA (longer-acting), Quillivant XR (liquid), Adzenys XR-ODT (orally disintegrating tablet). These competitors offer different dosing mechanisms, durations of action, and delivery formats, catering to varied patient and physician preferences.
- Amphetamine-based: Adderall (mixed amphetamine salts), Vyvanse (lisdexamfetamine dimesylate), Dexedrine. These offer an alternative stimulant mechanism and are widely prescribed.
- Non-Stimulant Medications: These are often used as second-line treatments or when stimulant side effects are problematic.
- Atomoxetine: Strattera.
- Alpha-2 Adrenergic Agonists: Intuniv (guanfacine extended-release), Kapvay (clonidine extended-release).
- Antidepressants (off-label use): Bupropion (Wellbutrin).
- Market Trends:
- Increased Diagnosis Rates: The prevalence of ADHD diagnosis continues to rise globally, fueling overall market growth.
- Formulation Innovation: There is a continuous development of novel delivery systems and formulations aiming to improve efficacy, reduce side effects, and enhance patient adherence (e.g., long-acting injectables, ODTs, liquids).
- Focus on Co-morbidities: Treatment strategies increasingly consider co-occurring conditions such as anxiety and depression, influencing medication choice.
- Generic Penetration: The availability of generics across multiple ADHD drug classes exerts downward pressure on pricing for both branded and generic products.
What is the projected financial trajectory for Metadate CD?
The financial trajectory for branded Metadate CD is characterized by a decline in revenue due to generic competition, offset by the overall growth in the ADHD market and potential residual sales.
- Revenue Decline: Post-patent expiry, branded Metadate CD revenue is projected to decrease significantly year-over-year. This decline is a direct consequence of volume and price erosion caused by generic methylphenidate extended-release products.
- Market Share Erosion: The market share captured by branded Metadate CD will likely continue to shrink as healthcare providers and payers opt for more cost-effective generic alternatives.
- Niche Market: Branded Metadate CD may retain a small but stable revenue stream from patients who specifically require or are accustomed to this particular formulation, or where payers have specific tiering that limits generic access.
- Generic Market Contribution: The broader market for extended-release methylphenidate, which includes generic versions of Metadate CD and other formulations, is expected to continue growing. While branded Metadate CD will not capture this growth directly, the underlying therapeutic category's expansion provides a backdrop.
- Manufacturing and Distribution Costs: As sales volumes for the branded product decrease, the per-unit cost of manufacturing and distribution may increase, impacting profit margins.
- Company Strategy: The pharmaceutical company responsible for Metadate CD may shift its focus towards newer, patent-protected ADHD treatments or other therapeutic areas, potentially reducing investment in marketing and sales support for the declining branded product.
- Long-Term Outlook: The long-term financial trajectory for branded Metadate CD will likely see it become a niche product with minimal revenue contribution compared to its peak sales period. Its primary financial impact will be historical, as a predecessor to newer generics and formulations.
What are the key considerations for R&D and investment related to Metadate CD?
For R&D and investment decisions concerning Metadate CD, the focus must shift from the branded product's lifecycle to understanding the broader landscape of extended-release methylphenidate and ADHD therapeutics.
- Generic Opportunities: For generic manufacturers, the expired patents on Metadate CD represent an opportunity for market entry. The key considerations include:
- Bioequivalence Studies: Successfully demonstrating bioequivalence to the reference listed drug (RLD) is a prerequisite for FDA approval.
- Manufacturing Scale and Cost: Achieving efficient, large-scale manufacturing at competitive costs is critical for market success.
- Distribution and Market Access: Securing contracts with wholesalers, pharmacies, and payers is essential for broad market penetration.
- Next-Generation Formulations: For R&D, the focus is on developing novel formulations that offer distinct advantages over existing products, including Metadate CD generics. This could involve:
- Improved Efficacy/Duration: Formulations with longer or more consistent plasma profiles.
- Reduced Side Effects: Delivery systems designed to minimize peak-and-trough effects or specific side effects like appetite suppression or sleep disturbances.
- Enhanced Adherence: Novel dosing forms (e.g., patches, injectables, dissolvable films) or combination therapies.
- Addressing Co-morbidities: Developing treatments that also target common co-occurring conditions.
- Investment in ADHD Market Growth: Investment in the overall ADHD market remains attractive due to increasing diagnosis rates and the ongoing need for effective treatments. Potential investment areas include:
- Emerging Therapeutics: Companies developing novel mechanisms of action or non-stimulant treatments.
- Specialty Pharmacies: Businesses focused on dispensing complex ADHD medications and offering patient support services.
- Diagnostic Technologies: Advancements in ADHD diagnosis and monitoring.
- Generic Manufacturers: Companies with strong capabilities in developing and marketing generic versions of established ADHD drugs, including methylphenidate ER.
- Intellectual Property Landscape: While Metadate CD's primary patents are expired, new intellectual property can be generated around novel formulations, manufacturing processes, or combination therapies within the methylphenidate class. Monitoring this evolving IP landscape is crucial.
- Regulatory Environment: Understanding current FDA guidance on ANDA (Abbreviated New Drug Application) filings, bioequivalence requirements, and labeling is essential for generic development. Similarly, awareness of evolving prescribing guidelines and payer policies influences market access.
Key Takeaways
- Branded Metadate CD's patent exclusivity has expired, leading to significant price erosion and market share loss to generic methylphenidate extended-release products.
- The ADHD treatment market is competitive, with numerous stimulant and non-stimulant options, driving innovation in formulations and delivery systems.
- The financial trajectory for branded Metadate CD is one of decline, with residual revenue from a niche market.
- Opportunities for generic manufacturers exist, while R&D investment should focus on next-generation ADHD therapeutics and novel formulations.
FAQs
-
What are the primary differences in efficacy between branded Metadate CD and its generic equivalents?
Generic formulations are required to demonstrate bioequivalence to the reference product, meaning they deliver the same amount of active ingredient into the bloodstream over the same period. Therefore, efficacy is generally considered comparable.
-
Will Metadate CD be discontinued by its manufacturer?
The discontinuation of a branded product is a business decision based on sales volume and profitability. While sales of branded Metadate CD have declined, it may continue to be manufactured as long as there is sufficient market demand from a specific patient or payer segment.
-
What are the advantages of extended-release formulations like Metadate CD over immediate-release methylphenidate?
Extended-release formulations provide a smoother, more sustained therapeutic effect throughout the day, reducing the need for multiple daily doses and potentially minimizing the "rebound" effect often seen with immediate-release versions.
-
How do payer formularies typically cover branded Metadate CD versus its generic versions?
Most payer formularies prioritize generic medications due to their lower cost. Branded Metadate CD would likely be placed on a higher cost-sharing tier or require prior authorization, while generic methylphenidate extended-release would be preferred and covered at a lower co-payment.
-
Are there any specific patient populations for whom branded Metadate CD might still be preferred over generics?
While rare, some patients may experience subtle differences in tolerability or response with specific inactive ingredients or manufacturing processes unique to the branded formulation. If a patient has a documented history of adverse events with generic formulations or has achieved optimal therapeutic benefit with the branded product, a physician may request its continued use, though this is increasingly challenging due to cost pressures.
Citations
[1] U.S. Food & Drug Administration. (n.d.). Approved Drug Products with Therapeutic Equivalence Evaluations (Orange Book). Retrieved from https://www.fda.gov/drugs/drug-approvals-and-databases/approved-drug-products-therapeutic-equivalence-evaluations-orange-book
[2] UCB Pharma. (n.d.). UCB Pharmaceutical Products. (Company website information regarding product portfolios and history).
[3] National Institute of Mental Health. (n.d.). Attention-Deficit/Hyperactivity Disorder (ADHD). Retrieved from https://www.nimh.nih.gov/health/topics/attention-deficit-hyperactivity-disorder-adhd