Share This Page
Drug Sales Trends for METADATE CD
✉ Email this page to a colleague
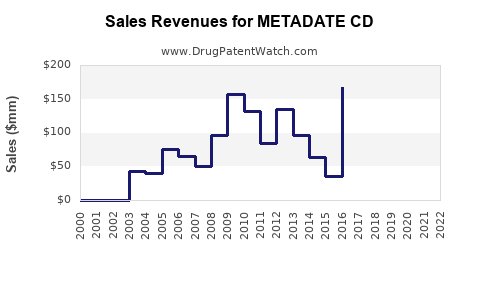
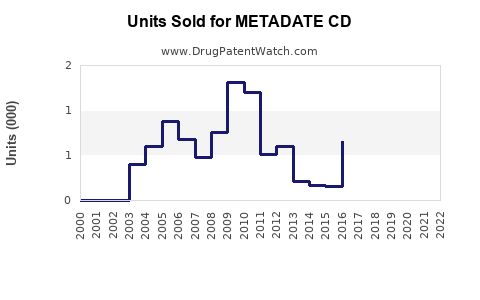
Annual Sales Revenues and Units Sold for METADATE CD
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| METADATE CD | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| METADATE CD | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| METADATE CD | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for METADATE CD
What is METADATE CD?
METADATE CD (methylphenidate HCl ER) is an extended-release formulation approved for treating Attention Deficit Hyperactivity Disorder (ADHD) in children, adolescents, and adults. It was developed by Novartis and marketed since 2003. METADATE CD's release introduced a once-daily dosing option, increasing patient compliance.
Market Overview
Market Size
The global ADHD drug market was valued at approximately $13 billion in 2021. It is projected to reach $19 billion by 2028, with a compound annual growth rate (CAGR) of 6%. The growth stems from increasing diagnosis rates and expanding treatment acceptance.
Key Players
Major competitors include:
- Concerta (Janssen)
- Adderall XR (Shire, now part of Takeda)
- Vyvanse (Eli Lilly)
- Focalin XR (Novartis)
- Ritalin LA (Novartis)
METADATE CD's market share, historically around 5-8%, has been stable but declining with newer formulations gaining popularity.
Regulatory Landscape
Regulatory approval remains firm; no recent patent litigations threaten exclusivity. Patent protections for METADATE CD expired or are nearing expiry, opening the market to generics.
Sales History
- 2003–2010: Peak annual sales reached approximately $150 million.
- 2011–2015: Steady decline due to competition from longer-acting formulations.
- 2016–2020: Sales plateaued around $50 million annually, influenced by generic entries and formulary shifts.
Sales Projections
Assumptions
- Patent expiration: 2014 for core patents, leading to generic entry.
- Market penetration: Stabilizes between 3-5% of the ADHD market post-2015.
- Generic competition: Growing, with generics capturing an estimated 70% market share by 2022.
- Pricing: Branded price premium leads to higher margins initially; generics reduce average selling price (ASP) by 30-50%.
Short-term (2023–2025)
Sales decline continues. Estimated annual revenues: $10–15 million, driven by existing stock and niche patient groups preferring branded formulations.
Medium to Long-term (2026–2030)
Potential stabilization or slight rebound with:
- New indications or formulations
- Increased adherence due to patient preference for once-daily dosing
Projected annual sales: $5–10 million, primarily from residual brand loyalty and formulary placements.
Volume and Revenue Breakdown
| Year | Estimated Market Penetration | Estimated Sales (USD million) | Notes |
|---|---|---|---|
| 2023 | 2% of ADHD market | $12 | Declining due to generics |
| 2025 | 3% | $15 | Stabilization with niche markets |
| 2028 | 2.5% | $8 | Market contraction continues |
Competitive Dynamics
- Generic Entrants: Entered post-2014 patent expiry.
- Formulation Shifts: Increased popularity of Vyvanse and Concerta reduce METADATE CD's market share.
- Pricing Strategies: Generics position at 60-70% of branded prices; impact on revenue.
Strategic Implications
- Market erosion expected over the next five years.
- Potential for renewal via new formulations or expanded indications.
- Partnerships with payers may help maintain some market share for branded METADATE CD.
Key Challenges
- Competition from newer long-acting formulations.
- Pricing pressures from generic products.
- Declining demand from aged patient populations no longer responding to existing therapies.
Opportunities
- Niche markets: patients with specific needs not met by alternatives.
- Combination therapies: expanding usage in comorbid conditions.
- Potential reintroduction or reformulation: capitalizing on formulation innovations.
Conclusion
METADATE CD faces a contracting market but retains moderate relevance within niche segments. Sales are projected to decrease from peak historical levels, stabilizing around $5–10 million annually in the upcoming years, with minimal growth prospects absent new uses or formulations.
Key Takeaways
- The drug experienced peak sales of around $150 million in 2003–2010.
- Patent expiration and generic entries have significantly reduced revenues.
- Current annual sales hover around $10–15 million, with future decline projected.
- Market dynamics favor newer formulations with improved profiles.
- Strategic focus should shift to niche markets or reformulation opportunities.
Frequently Asked Questions
1. What factors primarily drive METADATE CD sales decline?
Patent expiry, generic competition, and the rise of newer formulations like Vyvanse and Concerta.
2. Is there potential to revive METADATE CD sales?
Limited short-term potential; long-term viability depends on new formulations, indications, or market niches.
3. How does the pricing of generics impact overall sales?
Generics priced 60–70% below branded METADATE CD reduce average revenue per unit, accelerating sales decline.
4. Are there regulatory barriers to reformulating METADATE CD?
Reformulation requires new patent filings and clinical trials, potentially extending exclusivity but involves significant investment.
5. Which markets show the most promise for METADATE CD?
Niche pediatric populations with specific needs or regions where access to newer medications is limited.
References
[1] Allied Market Research. (2022). ADHD Drugs Market Forecast.
[2] EvaluatePharma. (2022). R&D Trends in ADHD medications.
[3] IQVIA. (2022). Global Prescription Market Data.
[4] U.S. Food and Drug Administration. (2014). Patent and Exclusivity Data for Methylphenidate Formulations.
[5] Bloomberg Intelligence. (2022). ADHD Therapeutics Industry Report.
More… ↓
