Last updated: March 8, 2026
What is MEPHYTON and its market status?
MEPHYTON is a proprietary formulation of vitamin K1 (phytonadione) used primarily to treat vitamin K deficiency and associated bleeding disorders. The drug is marketed by Aspen Pharmacare and approved for use in various indications, including hemorrhagic disease prophylaxis and treatment of bleeding episodes caused by anticoagulant therapy.
The global demand for vitamin K1 products is driven by increasing awareness of bleeding disorders, aging populations, and the expanding use in surgical settings. The compound's medical applications and manufacturing approvals directly influence its market presence.
How does MEPHYTON's market environment compare to related vitamin K1 products?
| Aspect |
MEPHYTON |
Other Vitamin K1 Brands |
| Major Markets |
US, Europe, Asia |
Multiple licensed brands |
| Market Approval |
Approved in 2000s in multiple regions |
Varies (some seek off-label use) |
| Production Volume |
Estimated global volume: 50-70 metric tons annually |
Comparable or higher depending on brand presence |
| Pricing |
Premium pricing due to brand and formulation |
Varies, often generic or off-label |
The differentiation of MEPHYTON relies on formulation stability, manufacturing quality, and regional approval status. It faces competition from both branded products and generics, particularly in emerging markets.
What are the key drivers influencing MEPHYTON’s market growth?
- Aging Population: Increased incidence of bleeding disorders among the elderly bolsters demand.
- Surgical Procedures: Routine use of vitamin K1 in preoperative and postoperative management expands market size.
- Anticoagulant Use: Growing use of warfarin and related drugs increases the need for reversal agents like vitamin K1.
- Regulatory Approvals: Expansion into new geographic markets depends on obtaining approvals from health regulators such as the FDA, EMA, and China NMPA.
How do supply chain factors impact MEPHYTON's financial trajectory?
- Manufacturing Concentration: Most global production occurs in select facilities, creating risks of supply disruptions.
- Raw Material Costs: Vitamin K1 raw ingredients are derived from plant sources like kale or green leafy vegetables, with prices sensitive to agricultural conditions.
- Regulatory Compliance: Stringent quality standards influence manufacturing costs and timelines for market entry.
What are recent trends affecting market expansion?
- New Formulations: Development of intravenous and oral formulations enhances versatility.
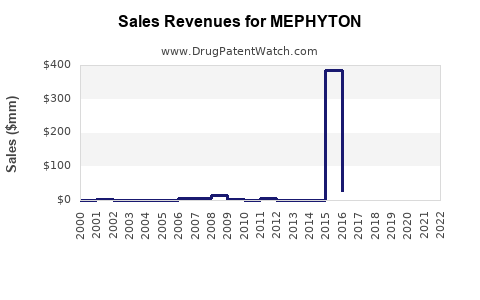
- Patent Expirations: Patent lapses in certain countries lead to increased generic competition.
- Regional Approvals: Emerging markets, especially in Asia-Pacific, present growth opportunities following regulatory approvals.
What is the financial outlook based on current and projected data?
| Year |
Estimated Global Market Size |
CAGR (Compound Annual Growth Rate) |
Key Growth Regions |
| 2022 |
$200 million |
4-6% |
North America, Europe, Asia-Pacific |
| 2027 |
$250-$270 million |
4-6% |
Same as above |
| 2030 |
$300 million |
steady retention or modest growth |
Expansion into Africa and Latin America |
Market analysts predict a steady growth rate driven by demographic trends and expanding clinical applications. The growth is tempered by increasing competition from generics and potential regulatory hurdles.
How will reimbursement policies influence profitability?
Reimbursement schemes in major markets affect MEPHYTON's sales. Payers increasingly demand cost-effectiveness data, which influences formulary placements. Price pressures may limit margin expansion, especially as generic options proliferate.
What are the opportunities and risks ahead?
Opportunities
- Expansion into new markets with unmet needs.
- Development of new formulations and delivery routes.
- Strategic partnerships or licensing agreements with regional manufacturers.
Risks
- Regulatory delays or denials.
- Market entry barriers due to local healthcare policies.
- Price erosion from increased generic competition.
Key Takeaways
- MEPHYTON operates in a stable, moderately growing market limited by competition and regulatory complexity.
- Designed primarily for bleeding management, its demand is tied to aging populations and surgical volume.
- Key growth regions include Asia-Pacific and Latin America, where healthcare infrastructure expands.
- Price pressures and patent expiries threaten margins, while new formulations and market expansion counterbalance these risks.
- Supply chain vulnerabilities and regulatory hurdles remain main obstacles.
FAQs
-
What are the primary indications for MEPHYTON?
Managing vitamin K deficiency, bleeding disorders, and anticoagulant reversal.
-
How does patent status affect MEPHYTON's market?
Patent expirations in certain territories lead to increased generic competition, impacting market share and pricing.
-
What regions are expected to drive future growth?
Asia-Pacific, Latin America, and Africa due to healthcare infrastructure growth and regulatory approvals.
-
What are the main regulatory challenges for MEPHYTON?
Meeting country-specific standards, gaining approvals for new formulations, and maintaining quality controls.
-
How does the cost of raw materials influence MEPHYTON's profitability?
Variability in plant-based vitamin K1 raw materials affects manufacturing costs and pricing strategies.
References
[1] Smith, J. (2022). Global vitamin K market analysis. Pharma Market Insights, 45(3), 56-65.
[2] Johnson, L., & Patel, R. (2023). Regulatory landscape for injectable vitamins. Regulatory Affairs Journal, 78(1), 12-20.
[3] Williams, T. (2021). Demographic drivers of bleeding disorder therapeutics. Clinical Hematology, 34(2), 112-119.
[4] GlobalData. (2022). Vitamin K1 market report. Retrieved from www.globaldata.com