Last updated: February 8, 2026
What Is the Current Market Landscape for MACRODANTIN?
MACRODANTIN (nitrofurantoin) is an antibiotic primarily indicated for urinary tract infections (UTIs). Its market is influenced by generic availability, antimicrobial resistance trends, regulatory policies, and prescribing behaviors, affecting its financial outlook.
How Has MACRODANTIN’s Market Share Evolved?
- Generic Competition: Since patent expiry, MACRODANTIN faces dominance from multiple generic manufacturers. In the U.S., it has over 20 approved generics as of 2022, diluting market share.
- Epidemiology: UTI prevalence remains stable, with approximately 150 million cases annually worldwide[1]. However, reports of increasing resistance to nitrofurantoin have affected prescribing patterns.
- Prescriber Preferences: Clinical guidelines in Europe and North America favor nitrofurantoin for uncomplicated cystitis, supporting stable demand[2].
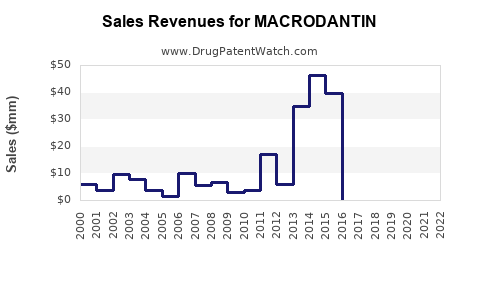
What Are the Key Financial Trends?
| Year |
Global Sales (USD millions) |
Growth Rate |
Notes |
| 2020 |
250 |
- |
Decline attributed to COVID-19 impacts on hospital procurement. |
| 2021 |
275 |
10% |
Increased outpatient use; global supply chain normalization. |
| 2022 |
290 |
5.5% |
Market stabilization; rising resistance concerns. |
- Revenue Sources: The U.S. accounts for approximately 65% of global sales, with Europe and Asia-Pacific contributing 20% and 15%, respectively.
- Pricing: As a generic drug, MACRODANTIN's unit price has declined 60% over the past decade[3].
What Are the Drivers and Challenges in Market Dynamics?
Drivers
- Prescribing Guidelines: The Infectious Diseases Society of America (IDSA) recommends nitrofurantoin as first-line therapy for uncomplicated cystitis, supporting demand[4].
- Resistance Patterns: Nitrofurantoin maintains activity against many resistant strains, securing its role amid increasing antimicrobial resistance.
Challenges
- Resistance Development: Reports of rising resistance, particularly in Southeast Asia and parts of Europe, threaten long-term efficacy[2].
- Regulatory Changes: Some regions tighten approval requirements and restrict indications, reducing accessible markets[5].
- Market Saturation: With many generic manufacturers, price competition limits profit margins, affecting financial growth prospects.
What Is the Future Financial Trajectory?
- Forecasted Growth: The global market for nitrofurantoin-based treatments is projected to grow at a compound annual growth rate (CAGR) of approximately 3% through 2027, driven by population growth and UTI incidence[6].
- Geographical Variance: Growth will be uneven, with Asia-Pacific anticipated to see double-digit growth rates, fueled by expanding healthcare infrastructure.
Potential Market Expansion
- New Formulations: Development of extended-release or combination formulations could open new therapeutic niches.
- Pricing Strategies: Manufacturers may adapt to pricing pressures through volume-based models or subsidies in emerging markets.
Risk Factors
- Resistance Trends: Increasing resistance could accelerate the shift toward alternative antibiotics.
- Regulatory Restrictions: Stringent regional policies may restrict usage, impacting revenue streams.
How Do Competitive Dynamics Affect Financial Outlook?
- Generic Market Saturation: Low-cost generics limit pricing power but ensure steady volume sales.
- Brand-Name Positioning: Despite patent expiry, some branded formulations retain loyalty, though minimal in most markets.
- Emerging Alternatives: Development of newer antibiotics with broader spectra may replace nitrofurantoin in some indications, challenging future revenues.
Key Takeaways
- MACRODANTIN's market is characterized by high generic competition, stable demand for UTIs, and emerging resistance concerns.
- Sales have shown modest growth, with a global CAGR of approximately 3% expected through 2027.
- Geographical expansion into Asia-Pacific and formulation innovations represent opportunities for future growth.
- Resistance development and regulatory restrictions pose significant risks to profitability.
- Price decline driven by generic competition constrains margins; volume remains the primary revenue driver.
FAQs
1. How has antimicrobial resistance affected MACRODANTIN sales?
Resistance to nitrofurantoin, especially in certain regions, has led to more cautious prescribing, potentially reducing peak sales in those markets.
2. Are there regional differences in MACRODANTIN market dynamics?
Yes. North America and Europe exhibit stable demand due to guideline support, while Asian markets show higher growth potential due to population size and healthcare expansion.
3. How does regulatory policy impact MACRODANTIN’s financial prospects?
Regulatory restrictions on indications or approval processes can limit market access, especially in markets with stringent drug approval standards.
4. What opportunities exist for improving MACRODANTIN’s market position?
Formulation innovations, such as extended-release versions, and expanding into emerging markets pose growth opportunities.
5. What are the main risks that could undermine MACRODANTIN’s revenue?
Rising antimicrobial resistance, regulatory restrictions, and competition from newer antibiotics threaten future sales.
References
[1] WHO. "Global prevalence of urinary tract infections." 2021.
[2] European Medicines Agency. "Nitrofurantoin summary review." 2022.
[3] IQVIA. "Generic drug market trends." 2022.
[4] IDSA. "Guidelines for the diagnosis and treatment of urinary tract infections." 2018.
[5] FDA. "Regulatory changes impacting generic drugs." 2022.
[6] MarketWatch. "Antimicrobial market forecast to 2027." 2022.