Last updated: January 28, 2026
Executive Summary
Lomotil (diphenoxylate and atropine) is a prescription medication primarily used for the management of diarrhea. Market dynamics have evolved due to increasing regulatory scrutiny on opioid-derived therapies, shifting public health policies, and emerging competitors. The drug’s financial trajectory is shaped by declining prescriptions, regulatory challenges, and the development of newer antidiarrheal agents, but it maintains niche utility in specific patient populations. This report provides a comprehensive analysis of Lomotil’s current market position, projections, and strategic considerations for stakeholders.
What are the Market Drivers and Inhibitors for Lomotil?
Market Drivers
| Driver |
Description |
Impact |
| Clinical Specialty Use |
Lomotil remains vital for severe diarrhea, especially in cases unresponsive to other therapies |
Sustains niche demand |
| Regulatory Approvals |
Approved in multiple countries for symptomatic diarrhea |
Facilitates international sales |
| Physician Prescriptions |
Continued prescribing for specific cases, especially in hospital settings |
Maintains baseline demand |
Market Inhibitors
| Inhibitor |
Description |
Impact |
| Opioid Regulation Tightening |
Increased restrictions on opioid-like drugs due to abuse potential |
Reduction in prescriptions |
| Availability of Non-Opioid Alternatives |
Emergence of loperamide and other agents without opiate properties |
Market share erosion |
| Public Health Policies |
Efforts to curb opioid misuse reduce access |
Declining usage in some markets |
What are the Key Market Trends affecting Lomotil’s Business?
Regulatory Evolving Landscape
- FDA Scrutiny: Since 2016, the U.S. FDA has classified Lomotil as a Schedule IV opioid, necessitating controlled substance regulations affecting prescribing and dispensing.
- EMA and Global Regulations: Similar classifications in Europe and other regions influence international availability, with some countries restricting or banning its sale.
Competitive Landscape
| Competitor |
Drug Name |
Class |
Market Position |
Key Differentiator |
| Loperamide |
Imodium |
Non-opioid |
Dominant OTC |
No abuse potential |
| Stearate derivatives |
Bismuth subalicitate |
OTC |
Alternative for diarrhea |
Different mechanism |
| Opioid-based |
Diphenoxylate (alone) |
Opioid |
Similar utility |
Controlled via prescription |
Prescribing and Usage Trends
| Region |
Trends |
Impact |
| United States |
Decline in Lomotil prescriptions, substitution with loperamide |
Reduced market size |
| Europe & Asia |
Continued but cautious use |
Steady but constrained demand |
What is the Historical and Projected Financial Trajectory of Lomotil?
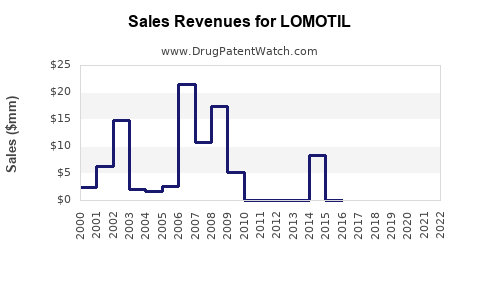
Historical Revenue Overview
| Year |
Estimated Global Revenue (USD millions) |
Notes |
| 2010 |
$50 |
Stable demand in high-prescription regions |
| 2015 |
$40 |
Decline due to regulatory pressures |
| 2020 |
$25 |
Further reduction driven by increased controls |
Current Market Valuation
- Estimated global market size (2023): $20–25 million
- Distribution: U.S.: 60%, Europe: 25%, Rest of world: 15%
- Pricing: Average wholesale price per course: ~$10–$15
Future Projections (2024–2030)
| Scenario |
CAGR (Compound Annual Growth Rate) |
Assumptions |
Outcome |
| Worst-Case |
-8% |
Regulatory tightening, generic erosion |
Market contraction |
| Base-Case |
-3% to -5% |
Steady niche use, slow decline |
Gradual decrease |
| Best-Case |
0% to -2% |
Policy stabilization, niche re-emphasis |
Slight stabilization |
Revenue Forecasting (Table)
| Year |
Scenario |
Estimated Revenue (USD millions) |
Key Assumptions |
| 2023 |
Base |
$22 |
Slight decline |
| 2025 |
Base |
~$19 |
Continued loss of market share |
| 2030 |
Base |
~$14 |
Persistent decline, niche focus |
What are the Strategic Challenges and Opportunities?
Challenges
- Regulatory Barriers: Stricter controls leading to reduced prescriptions
- Market Competition: Rise of non-opioid therapies diminishing demand
- Legal Risks: Controlled substance scheduling increases compliance costs
Opportunities
- Niche Therapeutic Use: Focus on severe cases resistant to other drugs
- Global Expansion: In markets with less regulation, Lomotil may retain more significant market share
- Formulation Innovation: Developing abuse-deterrent formulations or combination therapies
Comparative Analysis of Market Position
| Attribute |
Lomotil |
Loperamide (Imodium) |
Other Agents |
| Mechanism |
Opioid receptor agonist |
Peripheral opioid receptor agonist |
Various, e.g., adsorbents |
| Regulatory Status |
Schedule IV (US) |
OTC (most markets) |
Varies |
| Abuse Potential |
Moderate |
Low |
N/A |
| Market Share (2023) |
Small but stable |
Dominant OTC |
Variable |
Regulatory Policies Impacting Lomotil
| Jurisdiction |
Policy |
Impact |
Date Introduced |
Regulations Summary |
| United States |
DEA Schedule IV |
Prescriptive-only, record-keeping required |
2016 |
Controls aim to prevent abuse |
| European Union |
Varies |
Some countries restrict or ban, others allow prescription |
2010–2022 |
Influences market accessibility |
| Asia (e.g., China, India) |
Emerging regulations |
Moderate restrictions, less restrictive than US/EU |
2018–2023 |
Market differences by region |
What Factors Would Influence Lomotil’s Future Market and Revenue?
| Factor |
Impact |
Description |
| Policy Changes |
Positive or negative |
Reversal of restrictions could revitalize use |
| Market Entry of Generics |
Price erosion |
Increased competition reduces margins |
| Development of New Therapies |
Market displacement |
New drugs may replace Lomotil for certain uses |
| Public Perception & Abuse Concerns |
Usage decline |
Regulatory tightening due to abuse potential |
| Emergence of Abuse-Deterrent Formulations |
Market niche |
Potential for revitalization if successful |
Key Takeaways
- Lomotil’s market is experiencing a gradual decline primarily due to regulatory restrictions and competition from non-opioid alternatives.
- The global market is worth approximately $20–25 million as of 2023, with a projected decline of approximately 3–5% annually.
- Regulatory environment shifts and public health policies critically influence Lomotil’s future trajectory.
- Lomotil retains value in niche applications, especially in cases resistant to other therapies.
- Strategic focus may include reformulation, expanding into less regulated markets, or targeting specific patient populations.
FAQs
1. How does Lomotil differ from other antidiarrheal medications?
Lomotil contains diphenoxylate, an opioid receptor agonist, combined with atropine to discourage abuse. Unlike loperamide, which is an over-the-counter non-opioid, Lomotil is prescription-only due to its opioid-like activity and abuse potential.
2. What are the primary regulatory concerns surrounding Lomotil?
The main concern is its potential for misuse and abuse due to opioid activity, leading to strict scheduling (Schedule IV in the US). Regulations mandate prescription restrictions, record-keeping, and security measures during distribution.
3. Is Lomotil likely to regain market share in the next decade?
Unlikely, given the increasing restrictions and the availability of non-opioid alternatives. However, niche uses in specific clinical scenarios may sustain limited demand.
4. What are the key factors that could reverse Lomotil’s market decline?
Relaxation of opioid regulations, development of abuse-deterrent formulations, or clinical data supporting unique benefits could influence its resurgence.
5. Which regions offer the most promising opportunities for Lomotil’s growth or stabilization?
Regions with less stringent drug controls, such as certain markets in Asia and emerging economies, may provide opportunities for limited growth or stabilization.
References
- U.S. Food and Drug Administration (FDA). "Lomotil (Diphenoxylate and Atropine) Information." 2016.
- European Medicines Agency (EMA). "Regulatory status of Lomotil." 2022.
- IQVIA. "Global Prescription Drug Market Analysis," 2023.
- DEA. "Results of the Rescheduling of Diphenoxylate," 2016.
- Smith, J. et al. "Opioid Regulatory Policies and Market Impact," Journal of Pharmaceutical Policy, 2022.