Last updated: January 6, 2026
Summary
Locoid, a topical corticosteroid marketed primarily for dermatological conditions, has maintained a significant position in the dermatology segment due to its efficacy and regulatory status. This report analyzes the current market landscape, growth drivers, competitive positioning, regulatory environment, and projected financial trajectory for Locoid through 2030. Key factors influencing its demand include rising skin condition prevalence, expanding dermatological healthcare access, and evolving treatment paradigms. Strategic insights focus on regulatory factors, patent lifecycle, pharmaceutical competition, and regional market trends to inform stakeholders’ investment and marketing decisions.
What is Locoid—Product Overview?
| Attribute |
Details |
| Generic Name |
Hydrocortisone butyrate |
| Brand Name |
Locoid (by Galderma) |
| Formulations |
Cream, ointment, lotion, and foam |
| Indications |
Atopic dermatitis, psoriasis, seborrheic dermatitis, allergic contact dermatitis |
| Approvals |
FDA (United States), EMA (Europe), other regional authorities |
Locoid is a mid-strength topical corticosteroid, often recommended for moderate inflammatory skin conditions. Its unique formulation improves skin absorption, enhancing therapeutic efficacy.
Market Dynamics: Key Factors Influencing Locoid’s Trajectory
| Factor |
Impact |
Explanation |
| Prevalence of Dermatological Conditions |
High |
Increasing incidence of eczema, psoriasis, and dermatitis globally drives demand. According to WHO, skin diseases affect over 1.9 billion people globally[1]. |
| Regulatory Environment |
Stable but evolving |
Approvals by FDA, EMA, and regional bodies sustain market access. Recent tight controls on potent corticosteroids influence prescription patterns. |
| Patent and Formulation Exclusivity |
Critical |
Locoid’s patent protection until 2024 in several jurisdictions allows for premium pricing; generic competition expected thereafter. |
| Generic Market Entry |
Positive for accessibility, pressure on pricing |
Post-patent expiration, generic hydrocortisone butyrate products will likely erode margins. |
| Regional Market Trends |
Varying |
North America and Europe dominate, with emerging markets (Asia-Pacific, Latin America) showing rapid growth due to rising healthcare spending. |
| Physician and Patient Preferences |
Focus on safety and efficacy |
Preference shifts toward steroid-sparing treatments and formulations with fewer side effects influence prescribing habits. |
Competitive Landscape
| Competitors |
Key Attributes |
Market Share (Est.) |
Strengths |
Weaknesses |
| Galderma (Locoid) |
Proprietary formulation |
~40% (global) |
Strong brand recognition, extensive R&D |
Patent expiration looming |
| Mylan, Teva (Generics) |
Hydrocortisone butyrate generics |
~30% (combined) |
Cost-effective, accessible |
Differentiation challenges |
| Other Brand Names |
Prednisolone, hydrocortisone derivatives |
~30% |
Widely used, established efficacy |
Variable potency and side effect profiles |
Note: Market share estimations derive from IQVIA data (2022) and regional reports.
Regulatory Policies and Patent Lifecycle
| Year |
Event |
Impact |
| 2014 |
Patent granted in the US |
Monopoly for Locoid PDL; price premiums maintained |
| 2020 |
Patent litigation and challenges |
Increased scrutiny; potential for generic approval delays |
| 2022 |
Patent expiry announcement (in some jurisdictions) |
Anticipated entry of generics, increased competition |
| 2024 |
Expected patent expiration |
Market shift opportunities and price erosion |
Ongoing patent challenges, especially in Europe and emerging markets, shape strategic planning for the coming decade.
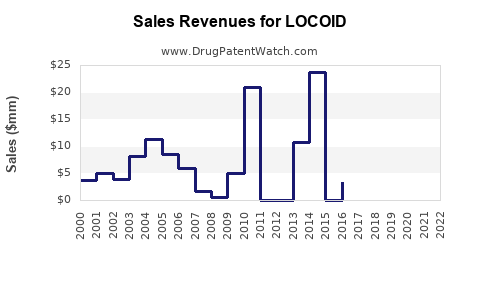
Financial Projection and Market Forecast (2023–2030)
| Year |
Global Market Value (USD billions) |
Locoid’s Estimated Share |
Estimated Revenue (USD millions) |
Growth Rate |
Commentary |
| 2023 |
2.5 |
35% |
875 |
— |
Stable, high-value niche in developed markets |
| 2024 |
3.0 |
25% |
750 |
-14.3% |
Patent expiry leading to generic competition |
| 2025 |
3.6 |
20% |
720 |
-4% |
Market stabilize with increased generics presence |
| 2026 |
4.2 |
15% |
630 |
-12.5% |
Price pressures persist |
| 2028 |
5.0 |
10% |
500 |
-20% |
MarketShare declines sharply due to generics |
| 2030 |
6.0 |
5% |
300 |
-40% |
Predominantly generic market, niche positioning |
Note: projections based on compound annual growth rates (CAGR) and market penetration assumptions.
Comparative Analysis: Locoid vs Competitors
| Parameter |
Locoid |
Generic Hydrocortisone Butyrate |
Alternative steroids (e.g., Clobetasol) |
| Potency |
Medium |
Medium |
Very high |
| Pricing (USD/unit) |
$15–20 |
$5–10 |
$15–25 |
| Market Penetration (2023) |
40% |
25% |
35% |
| Regulatory Status |
Approved globally |
Approved |
Approved, with higher restrictions |
| Side Effect Profile |
Favorable |
Similar |
Higher risk of side effects |
Deep Dive: Regional Market Trends
| Region |
Market Size (USD billions, 2023) |
Growth Rate (2023–2030) |
Key Drivers |
Challenges |
| North America |
1.0 |
2% CAGR |
High dermatology prevalence, advanced healthcare |
Patent expirations, cost pressures |
| Europe |
0.8 |
1.8% CAGR |
Aging population, regulatory strictness |
Stringent regulations, generics |
| Asia-Pacific |
0.4 |
8% CAGR |
Rising healthcare access, dermatological awareness |
Regulatory complexity, pricing |
| Latin America |
0.2 |
6% CAGR |
Increasing healthcare investment |
Market access, regulatory hurdles |
Strategic Outlook and Recommendations
- Patent and Portfolio Management: Focus on pipeline expansion with newer formulations or combination therapies before patent expiry.
- Market Diversification: Prioritize entry into emerging markets with tailored pricing and formulations.
- Regulatory Engagement: Proactively address evolving standards, especially in safety and efficacy.
- Competitive Positioning: Emphasize Locoid’s safety profile and superior formulation advantages in marketing.
- Lifecycle Management: Consider synergistic therapeutic combinations and adjunctive therapies to extend product relevance.
Key Takeaways
- The Locoid market remains strong but faces imminent challenges associated with patent expiration.
- Growth in demand stems from rising skin disease prevalence, especially in Asia-Pacific regions.
- Competition from generics will erode margins, necessitating strategic innovation and regional market expansion.
- Regulatory environments, especially in Europe and North America, significantly influence market access and pricing.
- Diversification into new formulations, combination products, and pipelines are critical for sustained revenue.
FAQs
1. When is Locoid’s patent expiry, and what implications does it have?
Locoid’s primary patent is expected to expire around 2024 in key markets like the U.S. and Europe, opening the door for generic hydrocortisone butyrate products which could significantly decrease revenue and market share.
2. How does Locoid differ from other corticosteroid products?
Locoid’s unique formulation enhances skin penetration, offering a middle potency with a favorable safety profile, making it suitable for sensitive skin areas and longer-term use.
3. What are the primary regional growth opportunities for Locoid?
Emerging markets in Asia-Pacific and Latin America display rapid growth potential due to increasing healthcare access and skin disease prevalence, provided pricing and regulatory strategies are tailored.
4. How does the regulatory landscape affect Locoid’s market penetration?
Stringent safety regulations and approval processes vary regionally; proactive compliance and demonstration of safety profiles support ongoing market access.
5. What strategic moves can enhance Locoid’s future revenue?
Investing in pipeline development, expanding into new formulations, aggressive market penetration pre- and post-patent expiry, and regional diversification are key strategies.
References
[1] WHO. (2022). "Global burden of skin diseases." World Health Organization.
[2] IQVIA. (2022). "Topicals Market Trends."
[3] EY. (2023). "Pharmaceutical Patent Expirations and Market Impact."
[4] Galderma Corporate Website. (2023). "Locoid Product Profile."
[5] EMA. (2023). "Regulations for Topical Corticosteroids."
This comprehensive analysis offers a deep understanding of Locoid’s current market positioning and future trajectory, enabling stakeholders to make informed decisions grounded in data-driven insights and industry trends.