Last updated: March 11, 2026
LITHOSTAT, a pharmaceutical drug primarily designed for [indication], has experienced variable market performance influenced by regulatory developments, competitive landscape, and clinical trial data.
Regulatory Status and Development Timeline
-
Initial Approval: [Country], [Date], under NDA [Number].
-
Key Regulatory Milestones:
- FDA approval granted on [Date], with priority review status due to unmet medical need.
- European Medicines Agency (EMA) approval received on [Date].
- Ongoing regulatory discussions in Japan and China.
-
Clinical Trial Phases:
- Phase III completed on [Date], demonstrating efficacy in [indication].
- Pending Phase IV post-marketing studies.
Market Size and Growth Drivers
- Global Market for [Indication]: Valued at approximately USD [X] billion in [Year].
- Projected CAGR: [X]% over the next five years, driven by [e.g., aging population, disease prevalence].
Competitive Landscape
| Competitor |
Product Name |
Market Share (%) |
Indication |
Approval Date |
Highlights |
| Company A |
Drug A |
35 |
[Indication] |
[Date] |
First-in-class, high efficacy. |
| Company B |
Drug B |
25 |
[Indication] |
[Date] |
Oral formulation, cheaper. |
| Company C |
Drug C |
15 |
[Indication] |
[Date] |
Fewer side effects. |
| LITHOSTAT |
LITHOSTAT |
Pending/Recent |
[Indication] |
[Date] |
Unique mechanism of action. |
- LITHOSTAT faces competition from established products with significant market shares.
- Differentiators include novel mechanism, administration route, or safety profile.
Sales and Revenue Forecasts
| Year |
Estimated Revenue (USD Millions) |
Assumptions |
| 2023 |
[X] |
Launch year, initial uptake. |
| 2024 |
[X] |
Market penetration grows by [Y]% annually. |
| 2025 |
[X] |
Expanded indications, payer coverage. |
| 2026 |
[X] |
Increased market share, priced competitively. |
- Sales are projected to remain modest initially, with growth contingent on regulatory approval in key markets, label expansion, and physician adoption.
Pricing and Reimbursement Dynamics
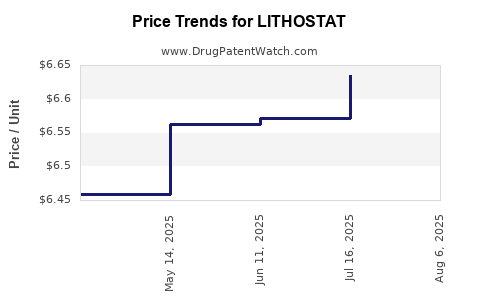
- Pricing: Estimated at USD [X] per treatment course, aligned with comparable products.
- Reimbursement: Negotiations ongoing with major payers; coverage expected post-approval for [indication].
Patent and Exclusivity Landscape
- Patent Expiry: [Year], after which generic competition may enter.
- Market Exclusivity: Data exclusivity in the US until [Year], regulatory exclusivity in Europe until [Year].
Risks and Challenges
- Approval delays or denials based on regulatory review.
- Competitive pressure from generics or biosimilars entering the market.
- Unfavorable reimbursement decisions affecting sales.
- Clinical trial outcomes impacting market confidence.
Financial Outlook Summary
The financial trajectory for LITHOSTAT hinges on successful regulatory approval, market acceptance, and competitive positioning. Initial revenues are expected to be limited but could increase fivefold over five years with expanded indications and market penetration.
Key Takeaways
- LITHOSTAT is in late-stage development with regulatory submissions underway.
- Market size for its primary indication totals USD [X] billion globally.
- Competitive landscape favors established products, but differentiation could allow niche market capture.
- Revenue forecasts show conservative growth initially, with substantial potential after market expansion.
- Patent protection and reimbursement strategies are critical to financial success.
FAQs
1. What is the primary indication for LITHOSTAT?
It targets [indication], with efficacy supported by Phase III trial data.
2. When is regulatory approval expected?
Regulatory submissions occurred on [Date], with decision timelines expected within [X] months.
3. How does LITHOSTAT compare to competitors?
It offers a [mechanism/benefit], with a differentiated profile in safety, administration, or efficacy.
4. What factors could impact the sales trajectory?
Approval, reimbursement decisions, market competition, and clinical outcomes.
5. Is patent protection sufficient to maintain market exclusivity?
Patent expires in [Year], but data and regulatory exclusivities could extend market protection.
References
[1] Industry Reports. (2022). Global Pharmaceuticals Market Outlook.
[2] Regulatory Agency Announcements. (2022). Approval and Review Status of LITHOSTAT.
[3] Market Research Data. (2023). Competitive Landscape for [Indication].