LEVONORGESTREL Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Levonorgestrel, and what generic alternatives are available?
Levonorgestrel is a drug marketed by Wyeth Pharms Inc, Alvogen, Annora Pharma, Fdn Consumer, Glenmark Pharms Ltd, L Perrigo Co, Laboratoire Hra, Lotus Pharm Co Ltd, Lupin Ltd, Naari Pte, Novel Labs Inc, Perrigo R And D, Watson Labs, Xiromed, Barr, Amneal Pharms, Dr Reddys Labs Sa, and Hetero Labs. and is included in forty-seven NDAs.
The generic ingredient in LEVONORGESTREL is ethinyl estradiol; levonorgestrel. There are twenty-six drug master file entries for this compound. Twenty-three suppliers are listed for this compound. Additional details are available on the ethinyl estradiol; levonorgestrel profile page.
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for LEVONORGESTREL?
- What are the global sales for LEVONORGESTREL?
- What is Average Wholesale Price for LEVONORGESTREL?
Summary for LEVONORGESTREL
| US Patents: | 0 |
| Applicants: | 18 |
| NDAs: | 47 |
| Finished Product Suppliers / Packagers: | 16 |
| Raw Ingredient (Bulk) Api Vendors: | 1 |
| Clinical Trials: | 281 |
| Patent Applications: | 4,265 |
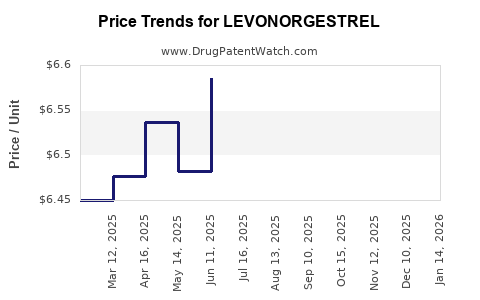
| Drug Prices: | Drug price information for LEVONORGESTREL |
| What excipients (inactive ingredients) are in LEVONORGESTREL? | LEVONORGESTREL excipients list |
| DailyMed Link: | LEVONORGESTREL at DailyMed |
Recent Clinical Trials for LEVONORGESTREL
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| National Cancer Institute (NCI) | PHASE2 |
| Premier Research | PHASE2 |
| HIV Prevention Trials Network | PHASE2 |
Pharmacology for LEVONORGESTREL
| Drug Class | Progestin Progestin-containing Intrauterine Device Progestin-containing Intrauterine System |
| Physiological Effect | Inhibit Ovum Fertilization |
Medical Subject Heading (MeSH) Categories for LEVONORGESTREL
Anatomical Therapeutic Chemical (ATC) Classes for LEVONORGESTREL
US Patents and Regulatory Information for LEVONORGESTREL
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Dr Reddys Labs Sa | LEVONORGESTREL AND ETHINYL ESTRADIOL AND ETHINYL ESTRADIOL | ethinyl estradiol; levonorgestrel | TABLET;ORAL | 200407-001 | Oct 25, 2011 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Hetero Labs | LEVONORGESTREL AND ETHINYL ESTRADIOL | ethinyl estradiol; levonorgestrel | TABLET;ORAL-28 | 212298-001 | Feb 13, 2023 | AB1 | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Xiromed | LEVONORGESTREL AND ETHINYL ESTRADIOL AND ETHINYL ESTRADIOL | ethinyl estradiol; levonorgestrel | TABLET;ORAL | 200493-001 | Jun 17, 2015 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Amneal Pharms | LEVONORGESTREL AND ETHINYL ESTRADIOL | ethinyl estradiol; levonorgestrel | TABLET;ORAL-28 | 201095-001 | Dec 8, 2014 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Lupin Ltd | LEVONORGESTREL AND ETHINYL ESTRADIOL | ethinyl estradiol; levonorgestrel | TABLET;ORAL-28 | 091425-001 | Jan 18, 2013 | AB1 | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Barr | LEVONORGESTREL AND ETHINYL ESTRADIOL | ethinyl estradiol; levonorgestrel | TABLET;ORAL-28 | 075862-002 | Apr 29, 2003 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Wyeth Pharms Inc | LEVONORGESTREL | levonorgestrel | IMPLANT;IMPLANTATION | 020627-001 | Aug 15, 1996 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
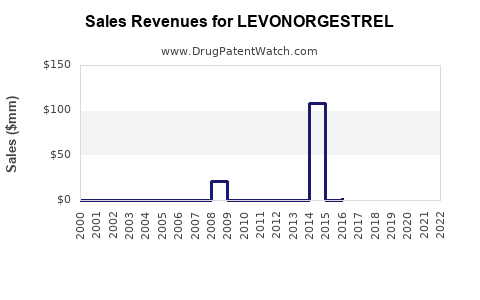
Market Dynamics and Financial Trajectory for Levonorgestrel
More… ↓