Last updated: February 19, 2026
KLOXXADO, a naloxone hydrochloride nasal spray developed by Harm Reduction Therapeutics, is positioned to enter a market with established players and evolving regulatory frameworks. Its market penetration will be influenced by pricing strategies, formulary access, and public health initiatives. Financial projections indicate revenue growth contingent on market adoption rates and competitive pricing.
What is KLOXXADO's Primary Indication and Mechanism of Action?
KLOXXADO is indicated for the emergency treatment of known or suspected opioid overdose, as manifested by respiratory and/or central nervous system depression. The drug is a nasal spray formulation of naloxone hydrochloride. Naloxone is an opioid antagonist that acts as a competitive inhibitor of the binding of opioids at the mu, kappa, and delta opioid receptors. By occupying these receptors, naloxone prevents or reverses the effects of opioid agonists. Its rapid absorption via the nasal mucosa allows for quick onset of action, crucial in overdose situations [1].
Who are KLOXXADO's Primary Competitors in the Opioid Antagonist Market?
The market for opioid antagonists, particularly naloxone products, is characterized by several established competitors. These include:
- Narcan (naloxone hydrochloride nasal spray) by Adapt Pharma. This product has been widely available and is often the benchmark for comparison in terms of market penetration and public awareness [2].
- Evzio (naloxone hydrochloride auto-injector) by Kaleo Pharma. While an auto-injector, it serves the same emergency overdose reversal purpose. However, it has faced challenges related to its high price point and subsequent market adjustments [3].
- Generic naloxone products. Various generic versions of naloxone, including nasal sprays and injectable formulations, are available. These generics contribute to price competition.
The competitive landscape is further shaped by the increasing availability of naloxone through state and federal programs, pharmacies without a prescription, and harm reduction organizations.
What is KLOXXADO's Pricing Strategy and Financial Impact?
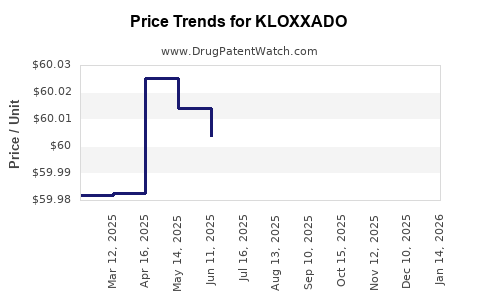
Harm Reduction Therapeutics has stated its intention to price KLOXXADO at $49.50 per two-dose package. This pricing strategy is notably lower than many existing naloxone products, particularly during periods when premium pricing was a concern for access [4].
The financial trajectory of KLOXXADO will be significantly influenced by this pricing. A lower price point is designed to:
- Enhance accessibility: Reduce cost barriers for individuals, families, first responders, and public health programs.
- Drive market share: Compete effectively against higher-priced alternatives and potentially capture a larger segment of the market, especially for bulk purchases by government agencies and non-profits.
- Align with public health goals: Support initiatives aimed at widespread availability and overdose prevention.
Revenue projections will depend on the volume of sales, the extent of insurance reimbursement, and the uptake by institutional purchasers. If KLOXXADO achieves broad adoption due to its affordability, it could capture substantial revenue even at a lower per-unit price, given the high demand for opioid overdose reversal agents.
What is the Regulatory Status and Approval Timeline for KLOXXADO?
KLOXXADO received U.S. Food and Drug Administration (FDA) approval on April 15, 2021 [1]. This approval grants the product the necessary authorization to be marketed and sold for its intended indication. The regulatory pathway for naloxone products is generally well-defined, with a focus on efficacy and safety in emergency overdose reversal.
The approval of KLOXXADO by the FDA on April 15, 2021, signifies its readiness for market entry. The subsequent timeline for its commercialization will involve manufacturing scale-up, distribution network establishment, and marketing efforts. Unlike novel drug approvals that may involve extensive post-market surveillance for long-term safety, naloxone's established profile means the immediate post-approval phase is focused on market integration and access.
What are the Key Market Drivers for Opioid Antagonist Products?
The market for opioid antagonists is driven by several critical factors:
- The Opioid Crisis: The ongoing opioid epidemic in the United States and globally is the primary driver for demand. High rates of opioid use disorder and associated overdose deaths create a persistent need for readily available overdose reversal medications [5].
- Public Health Policy and Funding: Government initiatives, federal and state funding for addiction treatment and overdose prevention, and "Good Samaritan" laws that encourage overdose reporting without fear of prosecution all increase the demand and accessibility of naloxone [6].
- Harm Reduction Strategies: The increasing adoption of harm reduction principles by public health agencies and advocacy groups promotes the distribution of naloxone as a critical life-saving tool.
- Prescription Access Expansion: Efforts to expand access to naloxone beyond traditional prescription pathways, such as dispensing at pharmacies without a prescription and direct distribution to community organizations, further fuel market growth.
- First Responder and Law Enforcement Distribution: The widespread equipping of emergency medical services, law enforcement, and other first responders with naloxone ensures a consistent institutional demand.
These drivers collectively create a robust and sustained demand for effective and accessible opioid antagonist medications.
How Will KLOXXADO's Accessibility Impact its Market Penetration?
KLOXXADO's accessibility, particularly its proposed $49.50 price point, is expected to be a significant factor in its market penetration. Historically, the high cost of some naloxone products, like Evzio, has been a barrier to widespread availability, even with FDA approval. Harm Reduction Therapeutics' stated commitment to affordability aims to circumvent this challenge.
Expected impacts on market penetration include:
- Increased uptake by public health programs: Government agencies and non-profit organizations focused on overdose prevention, often operating with budget constraints, are likely to favor a more affordable product for their distribution efforts.
- Broader pharmacy stocking: Lower wholesale costs may encourage more pharmacies to stock KLOXXADO, making it more readily available to the general public, especially in areas with limited access to specialized treatment centers.
- Competitive advantage: KLOXXADO can compete more effectively on price with both branded and generic naloxone nasal sprays. This could shift market share from products with higher price points.
- Potential for formulary inclusion: While cost is a factor, formulary decisions by insurers also consider clinical efficacy, safety, and overall cost-effectiveness within treatment pathways. A lower price, coupled with proven effectiveness, could facilitate favorable formulary placement.
The success of KLOXXADO's market penetration will hinge on its ability to translate this affordability into tangible distribution and usage, overcoming logistical and awareness hurdles inherent in any new product launch within a mature market.
What are the Projected Revenue Streams and Financial Growth Potential?
The financial growth potential for KLOXXADO is directly tied to its market adoption rate and its unique pricing strategy. As a product designed for emergency overdose reversal, the total addressable market is substantial, driven by the persistent opioid crisis.
Projected revenue streams include:
- Direct-to-consumer sales: Through pharmacies, although this segment might be smaller compared to institutional purchases for naloxone.
- Institutional sales: This is expected to be the primary revenue driver. Sales to federal, state, and local government agencies, public health departments, hospitals, and correctional facilities will be crucial.
- Non-profit and harm reduction organization sales: These entities are significant purchasers of naloxone for community-based distribution programs.
- Insurance reimbursement: While KLOXXADO's price aims to reduce reliance on insurance, reimbursement will still play a role in its overall financial viability, particularly for institutional buyers.
The financial growth trajectory will be influenced by:
- Volume of sales: Achieving high unit volumes will be key to generating significant revenue at a $49.50 price point.
- Market share capture: Successfully displacing or complementing existing products will determine overall market penetration.
- Payer negotiations: Securing favorable reimbursement and payment terms from government payers and private insurers.
- Cost of goods sold (COGS) and operational efficiency: Maintaining a lean operational structure will be vital to profitability given the price target.
Harm Reduction Therapeutics’ stated mission to provide an affordable naloxone product suggests a long-term strategy focused on volume and public health impact, rather than premium pricing. This approach, if successful, could lead to stable and significant revenue streams, albeit at a lower margin per unit compared to historically high-priced alternatives.
Key Takeaways
KLOXXADO enters the opioid antagonist market with a strategic pricing model designed to enhance accessibility. Its success will depend on converting this affordability into broad market penetration among public health entities, first responders, and consumers. The ongoing opioid crisis provides a sustained demand, while competition from established brands and generic options will shape its market share. Financial growth is projected through high-volume institutional sales, driven by its cost-effectiveness.
FAQs
-
What is the primary difference between KLOXXADO and other naloxone nasal sprays?
KLOXXADO's primary differentiator is its intended price point of $49.50 per two-dose package, which is significantly lower than many established naloxone nasal spray products, aiming to improve affordability and accessibility.
-
Who is the manufacturer of KLOXXADO?
KLOXXADO is manufactured by Harm Reduction Therapeutics.
-
When was KLOXXADO approved by the FDA?
KLOXXADO was approved by the U.S. Food and Drug Administration on April 15, 2021.
-
How is KLOXXADO administered?
KLOXXADO is administered as a nasal spray.
-
What is the mechanism of action for naloxone, the active ingredient in KLOXXADO?
Naloxone is an opioid antagonist that blocks the effects of opioids at the receptor sites, reversing respiratory depression and other symptoms of opioid overdose.
Citations
[1] U.S. Food & Drug Administration. (2021, April 15). FDA approves new naloxone hydrochloride nasal spray to help reverse opioid overdose. Retrieved from https://www.fda.gov/drugs/news-events-human-drugs/fda-approves-new-naloxone-hydrochloride-nasal-spray-help-reverse-opioid-overdose
[2] Adapt Pharma. (n.d.). Narcan® Nasal Spray. Retrieved from https://www.narcan.com/
[3] Kaleo, Inc. (n.d.). Evzio® (naloxone HCl) auto-injector. Retrieved from https://www.kaleocares.com/evzio
[4] Harm Reduction Therapeutics. (2023, October 3). Harm Reduction Therapeutics Announces FDA Approval of KLOXXADO™ (naloxone HCl) Nasal Spray, the First FDA-Approved Naloxone Nasal Spray Priced at $49.50 for a 2-Dose Package. Retrieved from https://harmreductiontherapeutics.com/harm-reduction-therapeutics-announces-fda-approval-of-kloxxado-naloxone-hcl-nasal-spray-the-first-fda-approved-naloxone-nasal-spray-priced-at-49-50-for-a-2-dose-package/
[5] National Institute on Drug Abuse. (2023, January). Opioid Overdose Crisis. Retrieved from https://www.drugabuse.gov/drug-topics/opioids/opioid-overdose-crisis
[6] Substance Abuse and Mental Health Services Administration. (n.d.). Good Samaritan Laws. Retrieved from https://www.samhsa.gov/good-samaritan-laws