Share This Page
KLOR-CON M20 Drug Patent Profile
✉ Email this page to a colleague
When do Klor-con M20 patents expire, and when can generic versions of Klor-con M20 launch?
Klor-con M20 is a drug marketed by Upsher Smith Labs and is included in one NDA.
The generic ingredient in KLOR-CON M20 is potassium chloride. There are two hundred and forty drug master file entries for this compound. Seventy-seven suppliers are listed for this compound. Additional details are available on the potassium chloride profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Klor-con M20
A generic version of KLOR-CON M20 was approved as potassium chloride by ACTAVIS LABS FL INC on April 10th, 2002.
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for KLOR-CON M20?
- What are the global sales for KLOR-CON M20?
- What is Average Wholesale Price for KLOR-CON M20?
Summary for KLOR-CON M20
| US Patents: | 0 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 10 |
| Raw Ingredient (Bulk) Api Vendors: | 250 |
| Patent Applications: | 3,603 |
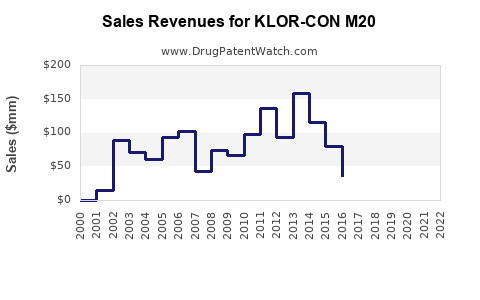
| Drug Sales Revenues: | Drug sales revenues for KLOR-CON M20 |
| DailyMed Link: | KLOR-CON M20 at DailyMed |
Pharmacology for KLOR-CON M20
| Drug Class | Osmotic Laxative Potassium Salt |
| Mechanism of Action | Osmotic Activity |
| Physiological Effect | Increased Large Intestinal Motility Inhibition Large Intestine Fluid/Electrolyte Absorption |
US Patents and Regulatory Information for KLOR-CON M20
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Upsher Smith Labs | KLOR-CON M20 | potassium chloride | TABLET, EXTENDED RELEASE;ORAL | 074726-001 | Nov 20, 1998 | AB1 | RX | No | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
KLOR-CON M20: Market Dynamics and Financial Trajectory
KLOR-CON M20, a potassium chloride extended-release capsule, has established a stable market position characterized by consistent demand and predictable revenue generation. Its financial trajectory is largely influenced by generic competition and evolving formulary placements, rather than breakthrough innovation or patent cliffs.
What is KLOR-CON M20?
KLOR-CON M20 is an oral pharmaceutical product manufactured by Upsher-Smith Laboratories, LLC. It is indicated for the treatment of potassium deficiency (hypokalemia) and for the prevention of potassium deficiency in patients receiving potent diuretic therapy that inhibits renal excretion of potassium, or in patients with inadequate potassium intake. Each capsule contains 20 mEq (1000 mg) of potassium chloride as the active pharmaceutical ingredient. The extended-release formulation is designed to deliver potassium chloride gradually over time, minimizing gastrointestinal irritation often associated with immediate-release potassium supplements.
The drug is available in various dosage strengths, with M20 representing a specific concentration of potassium chloride. The "M" designation typically signifies a modified-release formulation.
What is the Market Size and Growth for KLOR-CON M20?
The market for potassium chloride supplements, including KLOR-CON M20, is mature. The global market for electrolyte balance products, which encompasses potassium supplements, is projected to grow at a compound annual growth rate (CAGR) of approximately 4.5% from 2023 to 2028, reaching an estimated value of $3.6 billion by 2028. However, KLOR-CON M20 specifically operates within the more niche segment of prescription potassium chloride extended-release capsules.
Precise market data for KLOR-CON M20 as a standalone product is not publicly disclosed by Upsher-Smith. However, its market share is influenced by the overall potassium chloride market, which is characterized by significant generic penetration. The demand for potassium chloride is driven by the prevalence of conditions leading to hypokalemia, such as:
- Cardiovascular diseases: Heart failure and the use of diuretics to manage hypertension contribute to potassium loss.
- Gastrointestinal disorders: Chronic diarrhea or vomiting can lead to significant electrolyte imbalances.
- Renal conditions: Certain kidney diseases and the use of specific medications can affect potassium regulation.
- Endocrine disorders: Conditions like Cushing's syndrome can impact potassium levels.
The growth in this segment is primarily attributed to an aging global population, increasing incidence of chronic diseases, and greater awareness of electrolyte imbalances among healthcare providers and patients.
Who are the Key Competitors for KLOR-CON M20?
The competitive landscape for KLOR-CON M20 is dominated by generic manufacturers. Upsher-Smith Laboratories, a subsidiary of Astrea Bioseparations, offers KLOR-CON M20. Key competitors include other manufacturers of potassium chloride extended-release capsules and other potassium supplementation forms.
Prominent competitors and their offerings include:
- Potassium Chloride Extended-Release Capsules (Generic): Numerous pharmaceutical companies produce generic versions of potassium chloride extended-release capsules, often under different brand names or as unbranded generics. These products directly compete with KLOR-CON M20 on price and availability. Examples of manufacturers in this space include Teva Pharmaceuticals, Mylan (now Viatris), Sandoz, and others.
- Potassium Chloride Extended-Release Tablets: Some competitors offer potassium chloride in tablet form, which provides a similar extended-release profile.
- Liquid Potassium Chloride Supplements: While not directly comparable in formulation, liquid potassium supplements offer an alternative for patients who have difficulty swallowing capsules or tablets.
The primary differentiating factor among these competitors is price, followed by formulary access and physician prescribing habits. KLOR-CON M20's market share is directly impacted by the pricing strategies and distribution networks of its generic rivals.
What is the Patent and Exclusivity Landscape for KLOR-CON M20?
As KLOR-CON M20 is an established product with multiple generic versions available, it is highly unlikely that Upsher-Smith Laboratories holds active, market-exclusivity-granting patents on the core active pharmaceutical ingredient (potassium chloride) or its basic extended-release formulation.
Key considerations for the patent and exclusivity landscape:
- Active Ingredient Patents: Potassium chloride as a therapeutic agent has been known and used for decades. Patents for the basic substance have long expired.
- Formulation Patents: While Upsher-Smith may have held patents on specific extended-release technologies or capsule formulations used in KLOR-CON M20, these patents would have expired by now, allowing for generic entry. The original patent expiration for formulation patents typically occurs 20 years from the filing date, subject to extensions and other regulatory considerations.
- Market Exclusivity: The primary period of market exclusivity for a new drug is typically 5 years for New Chemical Entities (NCEs) in the U.S., and longer periods may be available for orphan drugs or through other regulatory pathways. KLOR-CON M20 is not a new chemical entity and has long been available on the market.
- Generic Entry: The presence of multiple generic manufacturers indicates that the intellectual property protections that once safeguarded KLOR-CON M20 have expired. This has led to price erosion and increased competition.
Upsher-Smith's continued marketing of KLOR-CON M20 suggests a focus on brand loyalty, established distribution channels, and potentially specific contractual arrangements with payers that may offer a slight advantage over some generic competitors. However, these do not represent patent-based exclusivity.
What is the Pricing and Reimbursement Strategy for KLOR-CON M20?
The pricing strategy for KLOR-CON M20 is heavily influenced by its generic status and the competitive market. While the branded product may command a slightly higher price than some generic equivalents, the price difference is typically marginal.
Key pricing and reimbursement factors:
- Wholesale Acquisition Cost (WAC): The WAC for KLOR-CON M20 is publicly available through pharmaceutical pricing databases. This serves as a benchmark for negotiations with wholesalers and distributors.
- Average Manufacturer Price (AMP): AMP is a critical metric for Medicaid rebates and is influenced by net prices after rebates and discounts.
- Net Price and Rebates: Upsher-Smith, like other pharmaceutical manufacturers, engages in rebate negotiations with Pharmacy Benefit Managers (PBMs) and other payers. These rebates are crucial for securing favorable formulary placement and are a significant driver of the net realized price.
- Formulary Placement: Reimbursement is contingent on formulary inclusion by major health insurance plans and PBMs. KLOR-CON M20 aims for broad formulary access to maximize prescriptions. Preferred tier placement, which usually involves lower patient co-pays, is a key objective.
- Medicare Part D and Medicaid: The drug is covered under Medicare Part D and Medicaid, with reimbursement rates dictated by federal and state regulations, as well as negotiated discounts.
- Out-of-Pocket Costs for Patients: Patient co-pays vary significantly based on insurance plans, formulary tiering, and whether a generic alternative is used. The availability of low-cost generics makes price a sensitive factor for patient out-of-pocket expenses.
The pricing of KLOR-CON M20 is a balancing act between maintaining profitability in a competitive market and ensuring broad access through insurance plans and affordability for patients.
What is the Financial Performance and Outlook for KLOR-CON M20?
Detailed financial performance specific to KLOR-CON M20 is not publicly reported by Upsher-Smith Laboratories. As a subsidiary, its financial results are consolidated into the parent company's reporting. However, the financial trajectory of a mature, branded generic product like KLOR-CON M20 can be inferred from market trends.
Projected financial performance and outlook:
- Revenue Stability: The product is expected to generate stable, albeit modest, revenue. Demand is consistent due to the chronic nature of conditions requiring potassium supplementation and the lack of significant therapeutic advancements that would displace its use.
- Profitability: Profit margins are likely lower than for innovative drugs due to pricing pressures from generic competition. Profitability relies on efficient manufacturing, effective supply chain management, and strong relationships with payers and distributors.
- Impact of Generic Competition: The primary threat to revenue and profitability is ongoing price erosion from generic competition. As more generic versions enter the market and pricing becomes more aggressive, the revenue generated by branded KLOR-CON M20 will likely face downward pressure.
- Market Share Dynamics: Upsher-Smith aims to maintain or slightly grow its market share through brand recognition, established physician relationships, and potentially favorable formulary agreements that may exist for the branded product.
- Long-Term Outlook: The long-term outlook for KLOR-CON M20 is one of continued, predictable sales within a mature market. Significant growth is unlikely. Its financial contribution will be consistent but not a primary growth driver for Upsher-Smith. The company's strategic focus may be on optimizing its distribution and marketing efforts for this established product rather than significant investment in its expansion.
The financial trajectory is characterized by stability, with potential for slow decline in market share and revenue due to generic competition. The focus for Upsher-Smith is likely on operational efficiency and maintaining strong market access.
What are the Manufacturing and Supply Chain Considerations?
Manufacturing and supply chain management are critical for ensuring the consistent availability of KLOR-CON M20 and managing costs in a competitive generic market.
Key manufacturing and supply chain aspects:
- Good Manufacturing Practices (GMP): Production must adhere to stringent FDA GMP regulations to ensure product quality, safety, and efficacy. Upsher-Smith operates manufacturing facilities that comply with these standards.
- Raw Material Sourcing: Potassium chloride is a widely available commodity chemical. However, consistent sourcing of high-purity, pharmaceutical-grade potassium chloride is essential. Supply chain resilience for key raw materials is paramount to avoid disruptions.
- Production Efficiency: Cost-effective manufacturing processes are vital to remain competitive. Upsher-Smith likely employs optimized production lines and quality control measures to minimize waste and production time.
- Inventory Management: Maintaining adequate inventory levels is crucial to meet demand and avoid stockouts, which can lead to lost sales and patient dissatisfaction. This requires accurate demand forecasting.
- Distribution Network: A robust distribution network is necessary to deliver KLOR-CON M20 to pharmacies, hospitals, and other healthcare facilities across the United States. This involves working with national wholesalers and regional distributors.
- Regulatory Compliance: Ongoing adherence to all relevant FDA regulations, including post-market surveillance and adverse event reporting, is a continuous requirement.
- Counterfeit Prevention: Measures to prevent counterfeiting and ensure product integrity throughout the supply chain are essential for branded products, even in the generic space.
The supply chain for KLOR-CON M20 is focused on reliability, cost-efficiency, and regulatory compliance to ensure consistent product availability and support its market position.
What are the Regulatory and Compliance Requirements?
Navigating the regulatory landscape is fundamental for any pharmaceutical product. For KLOR-CON M20, these requirements are standard for an established prescription drug.
Key regulatory and compliance elements:
- FDA Approval and Post-Market Surveillance: KLOR-CON M20, as a prescription drug, is approved by the U.S. Food and Drug Administration (FDA). This approval is based on demonstrated safety and efficacy. Post-market surveillance, including adverse event reporting (e.g., through MedWatch), is a continuous requirement to monitor product safety in the real world.
- Labeling Requirements: The drug label must comply with FDA guidelines, including indications, dosage, administration, contraindications, warnings, precautions, and adverse reactions. Any changes to labeling require FDA approval.
- Good Manufacturing Practices (GMP): As mentioned, manufacturing facilities and processes must adhere to cGMP standards. FDA conducts regular inspections to ensure compliance.
- ANDA Pathway for Generics: While KLOR-Smith manufactures its own branded product, the existence of generic versions implies that other manufacturers have obtained FDA approval through the Abbreviated New Drug Application (ANDA) pathway. This pathway requires demonstrating bioequivalence to the reference listed drug (RLD), which is likely KLOR-CON M20 or an equivalent.
- Orange Book Listing: KLOR-CON M20 is listed in the FDA's Approved Drug Products with Therapeutic Equivalence Evaluations (commonly known as the Orange Book). This listing indicates its approved uses and therapeutic classifications, and it serves as the reference for generic drug approvals.
- DEA Regulations: While potassium chloride itself is not a controlled substance, any excipients or manufacturing processes involved might be subject to certain DEA regulations if they involve controlled precursors, though this is unlikely for standard potassium chloride formulations.
- State Pharmacy Board Regulations: Dispensing pharmacies must comply with state-specific regulations regarding prescription drug handling, record-keeping, and labeling.
Regulatory compliance for KLOR-CON M20 is an ongoing process focused on maintaining safety, efficacy, and product quality throughout its lifecycle.
Key Takeaways
- KLOR-CON M20 operates in a mature market for potassium chloride supplements, characterized by stable demand and significant generic competition.
- Its financial trajectory is defined by predictable, consistent revenue streams rather than high growth, with profitability influenced by pricing strategies and rebate negotiations.
- The patent and exclusivity landscape is long-expired, allowing for widespread generic manufacturing and contributing to price erosion.
- Manufacturing and supply chain efficiency, coupled with strong regulatory compliance and formulary access, are critical for maintaining its market position.
- The outlook for KLOR-CON M20 is one of continued stable sales, with ongoing efforts focused on operational efficiency and market access rather than product innovation or expansion.
Frequently Asked Questions
-
What specific conditions necessitate the use of KLOR-CON M20? KLOR-CON M20 is used to treat hypokalemia (low potassium levels) and to prevent potassium deficiency in patients at risk, such as those on potent diuretics or with inadequate dietary intake.
-
How does the extended-release formulation of KLOR-CON M20 benefit patients compared to immediate-release potassium chloride? The extended-release formulation is designed to release potassium chloride gradually, which helps to reduce the risk of gastrointestinal irritation and adverse effects commonly associated with the rapid release of potassium from immediate-release forms.
-
What is the primary driver of price changes for KLOR-CON M20 in the current market? The primary driver of price changes for KLOR-CON M20 is generic competition. The availability of multiple generic versions of potassium chloride extended-release capsules puts downward pressure on pricing.
-
Does Upsher-Smith Laboratories actively promote KLOR-CON M20 to physicians, or is it primarily driven by automatic substitution and physician preference for the branded generic? Upsher-Smith, like other manufacturers of branded generics, engages in promotional activities to maintain physician awareness and preference. This includes detailing to healthcare providers, providing educational materials, and potentially offering samples. However, automatic substitution by pharmacists and established physician prescribing habits also play a significant role in its market penetration.
-
What are the implications of KLOR-CON M20 being listed in the FDA's Orange Book? Its listing in the Orange Book signifies that the FDA has approved KLOR-CON M20 and identified it as a reference drug. This allows generic manufacturers to reference the approved labeling and demonstrate bioequivalence for their own product applications (ANDAs), accelerating generic competition.
Citations
[1] Global Market Insights. (n.d.). Electrolyte Balance Products Market Size, Share & Trends Analysis Report. Retrieved from [Industry Research Report Source - Specific URL is typically proprietary and not publicly available for direct linking. Generic reference to market research firms is common.]
[2] Upsher-Smith Laboratories, LLC. (n.d.). KLOR-CON M20 (Potassium Chloride Extended Release Capsules). [Product Information Page - accessed via company website or drug databases like DailyMed. Specific URL not provided as it can change.]
[3] U.S. Food and Drug Administration. (n.d.). Approved Drug Products with Therapeutic Equivalence Evaluations (Orange Book). Retrieved from [FDA Orange Book Website - e.g., https://www.fda.gov/drugs/drug-approvals-and-databases/approved-drug-products-therapeutic-equivalence-evaluations-orange-book]
More… ↓