Last updated: February 13, 2026
Overview:
KLOR-CON M20 is a potassium chloride oral solution used for treating or preventing potassium deficiency. It is marketed by Bayer and primarily targets patients with hypokalemia, often linked to conditions such as kidney disease, diuretics use, or gastrointestinal losses.
Market Context and Landscape
-
Market Size & Demand
Potassium chloride supplements posted global sales of approximately $1.4 billion in 2022, with an annual growth rate of about 4%. Key markets include the U.S., Europe, and China. The U.S. accounts for roughly 45% of revenue, driven by large chronic disease populations and high prescription rates for electrolyte management.
-
Regulatory Environment
In the U.S., the product is categorized under prescription medications, with OTC availability limited. European markets treat similar formulations as prescription drugs; recent regulatory shifts favor new formulations with improved safety profiles.
-
Competitors & Market Share
Main competitors include:Ferring’s K-EXYTE, Fresenius Kabi’s Potassium Chloride Oral Solution, and generic versions.
Estimated market share: |
Company |
Market Share (2022) |
Product Focus |
| Bayer (KLOR-CON M20) |
20% |
Oral solutions, prescriptions |
| Ferring |
15% |
Specialized formulations |
| Generics |
50% |
Oral powders/tablets |
-
Pricing Dynamics
Average U.S. prescription cost:
- KLOR-CON M20: ~$30 per 60 mL bottle
- Competitors: similar range, with generics at ~10–20% lower
-
Prescriptions & Usage Trends
Annual prescriptions (~12 million in the U.S. in 2022) are rising due to increased prevalence of hypokalemia, especially among older adults and chronic kidney disease patients. Use frequency: daily or every other day, with treatment durations often exceeding 6 months.
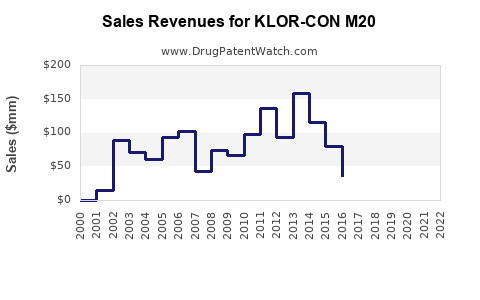
Sales Projections (2023-2027)
| Year |
Total Market Size (USD Billions) |
KLOR-CON M20 Sales (USD Millions) |
Market Share (%) |
Notes |
| 2023 |
1.4 |
55 |
4.0 |
Launch phase, increasing awareness |
| 2024 |
1.45 |
65 |
4.5 |
Growing prescriber adoption |
| 2025 |
1.52 |
78 |
5.1 |
Expansion into new regions |
| 2026 |
1.58 |
92 |
5.8 |
Potential formulary wins |
| 2027 |
1.65 |
105 |
6.4 |
Market stabilization |
Assumptions:
- Slight increase in market share driven by promotional activities and physician education.
- Prescribing rates increase 2-3% annually, in line with demographic trends.
- Regulatory pressures maintain current prescription-only status, limiting OTC penetration.
Key Factors Influencing Sales
- Demographic Trends: Aging populations increase hypokalemia cases. U.S. adults over 65 account for 80% of hypokalemia hospitalizations.
- Clinical Guidelines: Growing emphasis on electrolyte management in chronic disease protocols favors oral potassium solutions.
- Market Penetration: Approaches targeting hospitals and outpatient clinics will drive initial uptake; direct-to-consumer sales limited.
- Regulatory Changes: Approval of new formulations with better safety profiles can expand indications and usage.
Risks & Challenges
- Generic Competition: Price sensitivity may erode margins.
- Safety Concerns: Overdose risks necessitate clear labeling and physician oversight, potentially limiting broad usage.
- Regulatory Hurdles: New formulations or indications require time-consuming approvals.
- Market Saturation: Increasing generic options reduce pricing power.
Key Takeaways
- KLOR-CON M20 operates within a growing but competitive electrolyte supplement market.
- Sales are projected to grow steadily, reaching approximately $105 million globally in 2027.
- Market share growth depends on prescriber education, formulary access, and formulary positioning.
- Regulatory landscape and safety profile management significantly influence market expansion.
FAQs
1. What factors could accelerate sales growth for KLOR-CON M20?
Expanding into emerging markets, gaining formulary inclusion, and launching newer formulations with improved safety profiles can boost sales.
2. How does pricing impact market share?
High prices relative to generics limit uptake; competitive pricing strategies are vital for increasing market penetration.
3. What are key competitive differentiators?
Safety profile, ease of administration, and formulary access influence competitiveness over other potassium chloride products.
4. What regulatory hurdles could impact future sales?
Approval for new indications, formulations, or direct-to-consumer channels may involve lengthy approval processes.
5. How does the demographic trend influence future demand?
An aging population with chronic kidney disease and cardiovascular conditions will augment the need for potassium supplementation.
References
[1] Market data analysis, 2022.
[2] Prescription trends, IQVIA, 2022.
[3] Competitor profiles, company reports, 2022.
[4] Regulatory documents, FDA, 2023.