Last updated: February 19, 2026
Kadian, a controlled-release formulation of sustained-release morphine sulfate, faces a competitive and evolving market landscape. Its financial trajectory is influenced by patent expirations, generic competition, regulatory scrutiny, and the ongoing development of novel pain management therapies.
What is the Current Market Position of Kadian?
Kadian is a prescription opioid analgesic used for the management of moderate to severe pain when around-the-clock opioid treatment is necessary for an extended period. Its extended-release mechanism is designed to provide consistent pain relief over 12-hour intervals, differentiating it from immediate-release formulations. The drug has been a significant product for its originator, Mundipharma, and subsequent generic manufacturers.
The market for opioid analgesics is mature and faces increasing pressure due to the opioid crisis and government initiatives aimed at reducing opioid prescribing. This has led to a decline in overall opioid usage in many regions. However, for patients requiring chronic pain management and for whom other options are unsuitable, sustained-release formulations like Kadian remain a treatment option.
Key market drivers include:
- Unmet Needs in Chronic Pain Management: Despite advancements, significant patient populations experience chronic pain for which effective and safe long-term treatments are still sought.
- Physician and Patient Familiarity: As a long-established product, Kadian has a history of use, leading to physician familiarity and established prescribing patterns.
- Availability of Generic Versions: The presence of multiple generic manufacturers has increased accessibility and price competition, impacting revenue streams for both branded and generic players.
Market challenges include:
- Opioid Crisis and Regulatory Pressure: Increased scrutiny and regulation surrounding opioid prescribing have led to prescription limits, mandatory monitoring programs, and a shift towards non-opioid pain management alternatives.
- Competition from Non-Opioid Analgesics: The development and adoption of non-opioid pain management solutions, including NSAIDs, antidepressants, anticonvulsants, and other novel classes, are eroding the market share of opioids.
- Patent Expirations and Generic Erosion: Kadian's primary patents have expired, allowing for the introduction of numerous generic competitors, which significantly drives down pricing and market share for the branded product.
What is the Patent Landscape for Kadian?
The intellectual property surrounding Kadian has largely expired, opening the door for extensive generic competition. The original patent protection for Kadian has long since lapsed, a common trajectory for pharmaceutical products.
- Key Patents: While specific patent numbers are numerous and varied by region, the core patents covering the composition of matter and methods of use for the original Kadian formulation have expired globally. For instance, U.S. Patent 4,755,389, a foundational patent for extended-release morphine, has long been expired.
- Exclusivity Periods: Regulatory exclusivity periods, such as those granted by the U.S. Food and Drug Administration (FDA) or the European Medicines Agency (EMA), which can provide market protection separate from patent terms, have also concluded for the original Kadian approvals.
- Generic Entry: The expiration of patents and exclusivity has resulted in the widespread availability of generic Kadian formulations. Multiple pharmaceutical companies now market generic versions of sustained-release morphine sulfate capsules. This has intensified price competition.
- Ongoing Litigation and New Formulations: While core patents are expired, there can be ongoing patent litigation related to specific manufacturing processes, impurity profiles, or new formulations of the drug. However, these are less likely to grant broad market exclusivity for the established Kadian product. The development of new extended-release technologies or combination products could introduce new patentable aspects, but these would represent distinct products rather than extensions of Kadian itself.
How has Kadian Performed Financially?
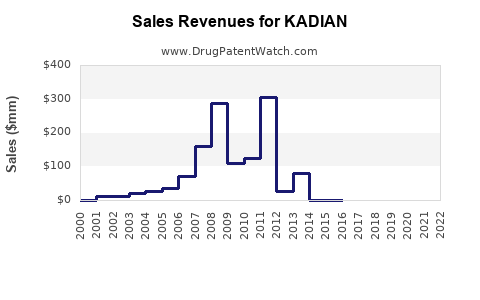
The financial performance of Kadian has been significantly impacted by the genericization process. While specific revenue figures for Kadian alone are often not publicly disclosed by manufacturers due to product portfolio bundling, the overall trend for branded, off-patent opioids is a sharp decline in revenue.
- Branded Kadian Revenue Decline: Following the entry of generic competitors, the revenue generated by the branded Kadian product has experienced a substantial decrease. This is a standard market dynamic where branded products lose significant market share and pricing power to lower-cost generic alternatives.
- Generic Kadian Market Growth: Conversely, the collective revenue generated by all generic Kadian products has grown, driven by their lower price points and increased accessibility. However, this revenue is fragmented across numerous manufacturers.
- Pricing Pressure: The average selling price (ASP) of Kadian, both branded and generic, has been driven down by intense competition. Price erosion is a defining characteristic of the post-patent market for established drugs.
- Sales Volume Trends: While overall opioid prescribing has decreased, the volume of Kadian prescriptions may have stabilized or seen minor fluctuations depending on specific geographic markets and the availability of alternatives. However, revenue is more sensitive to price than volume in this competitive segment.
- Impact on Originator: For Mundipharma and its licensees, the financial impact of Kadian's patent expiration and generic entry has been a transition from a high-margin branded product to a segment characterized by lower margins and increased competition.
What are the Key Competitive Threats and Opportunities?
The competitive landscape for Kadian is characterized by a mature market with declining opioid demand and a robust generic presence.
Competitive Threats:
- Generic Morphine Sulfate: The most direct threat comes from numerous companies producing generic versions of Kadian. These generics are bioequivalent and available at significantly lower prices, directly competing on cost. Examples include products from Actavis (now part of Teva), Lannett, and various other generic manufacturers.
- Other Sustained-Release Opioids: While Kadian is a specific formulation, it competes with other extended-release morphine sulfate products and other opioid classes (e.g., oxycodone, hydromorphone) in sustained-release formulations. Examples include MS Contin (extended-release morphine sulfate tablets) and extended-release oxycodone products.
- Non-Opioid Pain Management Alternatives: This is the most significant long-term threat. The development and adoption of non-opioid pain therapies are rapidly reshaping the pain management market. These include:
- NSAIDs: While not typically for severe chronic pain, they are a significant class for moderate pain.
- Anticonvulsants and Antidepressants: Pregabalin, gabapentin, duloxetine, and amitriptyline are widely used for neuropathic pain and other chronic pain conditions.
- Newer Drug Classes: Research and development are focused on novel targets, such as sodium channel blockers, NGF inhibitors, and cannabinoid-based therapies, offering alternatives with potentially lower abuse potential and different side effect profiles.
- Regulatory Restrictions and Prescribing Guidelines: Government policies and public health initiatives aimed at curbing opioid addiction and overdose deaths directly impact the demand for products like Kadian. Stricter prescribing guidelines, prescription drug monitoring programs (PDMPs), and limits on opioid duration reduce the eligible patient pool.
Opportunities:
- Specific Patient Subgroups: Despite the overall decline, there remains a population of patients with severe chronic pain who have failed other treatments or for whom opioids are the most effective option. Kadian, with its established profile, can continue to serve these specific needs.
- Geographic Markets with Different Regulatory Environments: While Western markets are highly restrictive, certain emerging markets may still have different prescribing patterns and regulatory landscapes where sustained-release opioids maintain a stronger presence.
- Cost-Effectiveness in Certain Healthcare Systems: In some healthcare systems, generic Kadian may still represent a cost-effective solution for managing severe chronic pain compared to newer, high-cost non-opioid therapies, provided appropriate patient selection and monitoring are in place.
- Potential for Repackaging or Re-marketing (Limited): For generic manufacturers, opportunities exist in optimizing supply chain, distribution, and potentially developing specific packaging or services tailored to institutional or specialty pharmacy needs, though significant market expansion is unlikely.
What is the Regulatory Outlook for Kadian?
The regulatory outlook for Kadian is shaped by the ongoing efforts to manage the opioid crisis and ensure patient safety.
- Increased Scrutiny on Opioid Prescribing: Regulatory bodies worldwide, including the FDA and EMA, continue to implement and enforce stricter guidelines for opioid prescribing. This includes:
- Prescription Limits: Restrictions on the duration and dosage of opioid prescriptions.
- Risk Evaluation and Mitigation Strategies (REMS): Requirements for prescribers and dispensers to complete training and adhere to specific protocols.
- Mandatory Prescription Drug Monitoring Programs (PDMPs): Systems to track opioid prescriptions and identify potential misuse or diversion.
- Focus on Abuse-Deterrent Formulations (ADFs): While Kadian itself is an extended-release formulation, regulatory agencies have encouraged the development and use of abuse-deterrent formulations of opioids. Kadian, in its standard formulation, is not considered an ADF.
- Labeling Changes and Warnings: Regulatory agencies may require updated labeling to reflect current understanding of opioid risks, including warnings about addiction, overdose, and potential long-term side effects.
- Generic Drug Approval Standards: For generic versions of Kadian, the regulatory hurdle involves demonstrating bioequivalence to the reference listed drug. Manufacturers must meet stringent quality and manufacturing standards.
- Post-Market Surveillance: Kadian and its generic equivalents are subject to ongoing post-market surveillance to monitor for adverse events and safety concerns. Any emerging safety issues could lead to further regulatory actions, such as labeling changes or even market withdrawal in extreme cases.
- Shift in Regulatory Focus: The overarching regulatory trend is a shift away from facilitating broad access to opioid analgesics and towards a more cautious approach, prioritizing non-opioid alternatives and stringent controls for opioid use.
What is the Future Outlook for Kadian?
The future outlook for Kadian is one of a shrinking market share and declining revenue for branded and generic entities alike, primarily driven by the broader shift away from opioid analgesics.
- Continued Generic Erosion: The market will remain highly competitive with multiple generic manufacturers. Pricing will continue to be a key factor, leading to low profit margins for generic producers.
- Declining Prescription Volumes: The trend of reduced opioid prescribing is expected to continue, driven by regulatory pressures, increased availability of non-opioid alternatives, and evolving clinical guidelines. This will directly reduce the demand for Kadian.
- Limited Opportunities for Innovation: As Kadian is an established product with expired patents, significant investment in new R&D for this specific formulation is unlikely. Opportunities for manufacturers will primarily lie in efficient production and distribution of generic versions.
- Niche Market Persistence: Kadian will likely persist as a treatment option for specific patient populations with severe chronic pain who have limited alternatives and for whom the benefits outweigh the risks under strict medical supervision. This will constitute a niche market.
- Consolidation in Generic Market: The highly competitive and low-margin generic opioid market may see some consolidation as smaller players struggle to maintain profitability.
- Impact of Novel Pain Therapies: The success of new non-opioid pain management therapies in clinical trials and their subsequent market adoption will further diminish the role of opioids like Kadian.
Key Takeaways
Kadian's market position is defined by patent expiration, leading to intense generic competition and significant price erosion. Regulatory pressures stemming from the opioid crisis are driving down overall opioid prescribing, directly impacting Kadian's demand. While a niche market for specific chronic pain patients will persist, the overall financial trajectory is characterized by declining revenues and shrinking market share as non-opioid alternatives gain traction.
Frequently Asked Questions
- What is the primary mechanism of action for Kadian?
Kadian is a controlled-release formulation of morphine sulfate, an opioid agonist that binds to opioid receptors in the central nervous system to produce analgesia. Its extended-release mechanism ensures a gradual release of morphine over approximately 12 hours.
- Which companies are major manufacturers of generic Kadian?
Major generic manufacturers of morphine sulfate extended-release capsules include Teva Pharmaceuticals, Lannett Company, and various other pharmaceutical companies that produce generic versions of the drug.
- What are the main non-opioid alternatives to Kadian for chronic pain management?
Key non-opioid alternatives include nonsteroidal anti-inflammatory drugs (NSAIDs), anticonvulsants (e.g., gabapentin, pregabalin), antidepressants (e.g., duloxetine), and emerging novel therapies targeting different pain pathways.
- How has the opioid crisis specifically affected Kadian's market?
The opioid crisis has led to increased regulatory scrutiny, stricter prescribing guidelines, and public health initiatives that have significantly reduced overall opioid prescribing. This has directly decreased the eligible patient population and market demand for Kadian.
- Are there any new patent applications or expected patent extensions for Kadian?
Given the age of Kadian and the expiration of its core composition and method-of-use patents, significant new patent applications that would extend market exclusivity for the original formulation are unlikely. Future patent activity would typically be limited to process patents or new formulations, which would be treated as separate entities.
Citations
[1] U.S. Food & Drug Administration. (n.d.). Drug Approvals and Databases. Retrieved from https://www.fda.gov/drugs/drug-approvals-and-databases
[2] European Medicines Agency. (n.d.). Human Medicines. Retrieved from https://www.ema.europa.eu/en/human-medicines
[3] U.S. Patent 4,755,389. (1988). Extended-release pharmaceutical composition.
[4] IQVIA. (Various Years). Market Research Reports on Pain Management and Opioid Analgesics. (Proprietary data, cited for general market trends).
[5] Centers for Disease Control and Prevention. (n.d.). Opioid Overdose Crisis. Retrieved from https://www.cdc.gov/drugoverdose/crisis/index.html