JUNEL Drug Patent Profile
✉ Email this page to a colleague
When do Junel patents expire, and what generic alternatives are available?
Junel is a drug marketed by Barr and Barr Labs and is included in four NDAs.
The generic ingredient in JUNEL is ethinyl estradiol; norethindrone acetate. There are twenty-six drug master file entries for this compound. Twenty-five suppliers are listed for this compound. Additional details are available on the ethinyl estradiol; norethindrone acetate profile page.
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for JUNEL?
- What are the global sales for JUNEL?
- What is Average Wholesale Price for JUNEL?
Summary for JUNEL
| US Patents: | 0 |
| Applicants: | 2 |
| NDAs: | 4 |
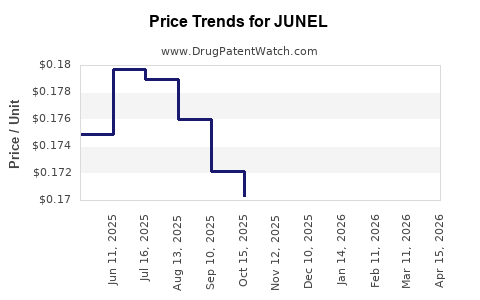
| Drug Prices: | Drug price information for JUNEL |
| Drug Sales Revenues: | Drug sales revenues for JUNEL |
| DailyMed Link: | JUNEL at DailyMed |
US Patents and Regulatory Information for JUNEL
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Barr | JUNEL 1.5/30 | ethinyl estradiol; norethindrone acetate | TABLET;ORAL-21 | 076381-001 | May 30, 2003 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Barr | JUNEL FE 1/20 | ethinyl estradiol; norethindrone acetate | TABLET;ORAL-28 | 076081-001 | Sep 18, 2003 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Barr Labs | JUNEL 1/20 | ethinyl estradiol; norethindrone acetate | TABLET;ORAL-21 | 076380-001 | May 30, 2003 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Barr | JUNEL FE 1.5/30 | ethinyl estradiol; norethindrone acetate | TABLET;ORAL-28 | 076064-001 | Sep 18, 2003 | AB | RX | No | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |