Last updated: February 19, 2026
JATENZO (testosterone undecanoate) is an orally administered testosterone replacement therapy approved for men with hypogonadism. Its market trajectory is influenced by the prevalence of hypogonadism, competitive landscape, patent protection, and pricing strategies.
What is the Current Market Size and Growth Potential for JATENZO?
The global testosterone replacement therapy (TRT) market is substantial and projected to grow. Hypogonadism, or low testosterone, affects an estimated 15% to 40% of aging men [1]. This demographic trend, coupled with increasing awareness and diagnosis of the condition, drives market expansion. JATENZO competes within this market, which also includes injectable, transdermal, and other oral formulations.
The market for TRT was valued at approximately USD 2.5 billion in 2022 and is anticipated to expand at a compound annual growth rate (CAGR) of 3.5% to 5.0% through 2030 [2]. Growth drivers include an aging male population, rising incidence of obesity and diabetes (conditions linked to low testosterone), and increased physician and patient acceptance of TRT. JATENZO's specific market share within this broader category is dependent on its ability to capture patients seeking an oral administration route, offering convenience over injections or daily transdermal applications.
What is the Competitive Landscape for Oral Testosterone Undecanoate?
JATENZO’s primary competition in the oral TRT segment includes other formulations of testosterone undecanoate and potentially other oral testosterone esters.
- Testosterone Undecanoate Oral Formulations: JATENZO is one of the few orally administered testosterone undecanoate products available in major markets. Other similar products may exist or be in development, differing in formulation, absorption characteristics, or proprietary delivery mechanisms.
- Injectable Testosterone: This remains a significant segment of the TRT market. Common injectable formulations include testosterone cypionate and enanthate. These are typically administered every one to two weeks.
- Transdermal Testosterone: Patches and gels are also widely used. These provide daily application and are favored by some patients for their continuous delivery. Examples include AndroGel and Testim.
- Other Oral Formulations: While less common due to bioavailability challenges, other oral testosterone compounds have been explored.
The competitive advantage of JATENZO lies in its oral administration, which offers convenience and avoids the need for injections or daily skin application. However, it faces competition from established injectable and transdermal therapies that have long market histories and physician familiarity. The development of new oral formulations with improved pharmacokinetic profiles or reduced dosing frequency could intensify competition.
What is the Patent Protection Status and Exclusivity Period for JATENZO?
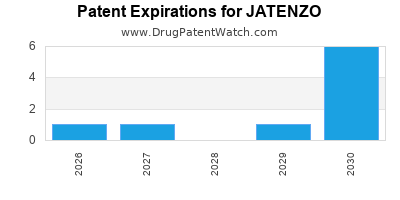
Understanding JATENZO’s patent landscape is critical for assessing its long-term market exclusivity and profitability. The drug's intellectual property protection dictates the period during which generic competition is restricted.
- Core Patents: JATENZO is protected by a portfolio of patents covering its composition of matter, methods of use, and manufacturing processes. These patents have varying expiration dates. For instance, patents related to the specific formulation and its efficacy in treating hypogonadism are key.
- Exclusivity Periods: Beyond patent expiration, regulatory exclusivities, such as those granted by the U.S. Food and Drug Administration (FDA) for New Chemical Entities (NCEs) or specific indications, can extend market protection. JATENZO received FDA approval in March 2020 [3]. As a product with a New Drug Application (NDA), it is eligible for various regulatory exclusivities.
- Patent Litigation: As patent expiration approaches or when generic manufacturers attempt to enter the market, patent litigation is common. Successful defense of patents can delay generic entry. The strength and scope of JATENZO's patents, and any related litigation outcomes, significantly impact its financial trajectory.
- Generic Entry Timeline: The earliest potential for generic entry for JATENZO would be contingent upon the expiration of its core patents and any applicable regulatory exclusivities. Precise dates are often subject to legal challenges. However, typical NCE exclusivities are five years, which could be extended by other patent types or market-based exclusivities if granted.
As of early 2024, specific details regarding the expiration of all core patents and the active status of all regulatory exclusivities require diligent monitoring through patent databases and regulatory filings.
What is the Pricing Strategy and Reimbursement Landscape for JATENZO?
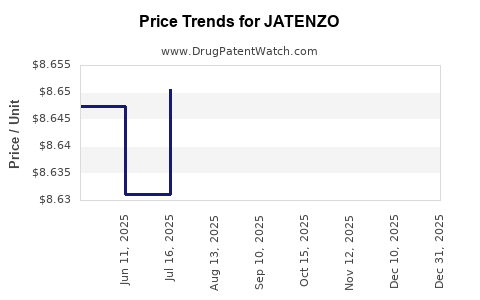
The pricing and reimbursement of JATENZO are crucial determinants of its market access and patient affordability, directly impacting sales revenue.
- Pricing: JATENZO's pricing reflects its therapeutic value, the cost of its development, and its positioning relative to competing TRT options. The average wholesale price (AWP) for branded pharmaceuticals can be substantial. For JATENZO, list prices for a 30-day supply have historically been in the range of several hundred dollars, subject to discounts and payer negotiations [4].
- Reimbursement: Payer coverage (insurance companies, Medicare, Medicaid) is essential. Obtaining favorable formulary placement and reducing patient co-pays are key commercialization strategies. Many payers evaluate TRT based on the severity of hypogonadism and the patient's response to therapy. Clinical trial data demonstrating efficacy and safety are critical for securing broad reimbursement.
- Patient Assistance Programs: Pharmaceutical companies often implement patient assistance programs (PAPs) to mitigate out-of-pocket costs for eligible patients, thereby improving access and adherence. These programs can be a significant factor in patient uptake for high-cost medications.
- Competitive Benchmarking: Pricing is also benchmarked against existing TRT options. If JATENZO offers superior convenience or efficacy, it can command a premium. Conversely, if competitive options are significantly cheaper and achieve similar outcomes, pricing pressure will be higher.
The evolving healthcare landscape, including value-based care initiatives and payer scrutiny of drug costs, continuously shapes the reimbursement environment for all pharmaceuticals, including JATENZO.
What are the Key Financial Metrics and Sales Performance of JATENZO?
Analyzing JATENZO's financial performance provides insight into its market adoption and future revenue potential. This involves examining sales figures, revenue growth, and profitability.
- Sales Revenue: JATENZO's sales revenue is reported by its manufacturer, previously Besse Medical and currently Ascend Therapeutics. Tracking quarterly and annual sales figures is paramount. For example, in its initial years post-launch, sales growth would be expected as market penetration increases.
- Revenue Growth Rate: The CAGR of JATENZO's sales revenue indicates its market traction. Factors influencing growth include prescription volume, market access, physician prescribing patterns, and patient adherence.
- Profitability: While specific profitability figures for a single drug are often proprietary, gross margins are influenced by manufacturing costs, marketing expenses, and sales volume. The cost of goods sold (COGS) and ongoing R&D or post-market surveillance costs also play a role.
- Market Share: JATENZO's market share within the oral TRT segment and the overall TRT market provides a relative performance indicator. Increases in market share suggest successful commercialization strategies.
- Analyst Projections: Financial analysts who cover the parent company or the specific therapeutic area often provide sales forecasts and target prices for the drug. These projections are based on market analysis, competitive intelligence, and company guidance.
Detailed financial performance data is typically disclosed in the quarterly and annual reports of the publicly traded company that markets JATENZO. For instance, Ascend Therapeutics' investor relations materials would be a primary source for this information.
What is the Impact of Clinical Data and Physician Adoption on JATENZO's Trajectory?
The success of JATENZO is intrinsically linked to the clinical evidence supporting its efficacy and safety, and the subsequent adoption by healthcare providers.
- Clinical Trial Outcomes: Robust clinical trial data demonstrating JATENZO's ability to achieve and maintain normal testosterone levels, improve hypogonadism symptoms (e.g., libido, energy, mood), and its safety profile are foundational. Pivotal Phase III trials and post-marketing studies provide this evidence.
- Physician Prescribing Habits: Urologists, endocrinologists, and primary care physicians are the key prescribers. Their familiarity with the drug's mechanism of action, dosing, potential side effects, and patient suitability influences prescription volumes. Educational initiatives and key opinion leader (KOL) engagement are critical for driving physician adoption.
- Patient Experience and Adherence: Positive patient experiences, including ease of use, perceived efficacy, and tolerability, contribute to sustained prescriptions and word-of-mouth referrals. Patient adherence is crucial for achieving therapeutic goals and demonstrating real-world effectiveness.
- Comparative Effectiveness: When compared to other TRT modalities in real-world evidence studies or head-to-head trials, JATENZO's performance profile will shape physician preference. Data highlighting its convenience without compromising safety or efficacy will bolster its position.
A continuous stream of positive clinical insights and documented real-world benefits is essential for maintaining and expanding JATENZO's market presence against established therapies.
What are the Regulatory Considerations and Post-Market Surveillance Requirements?
Regulatory compliance and ongoing surveillance are critical for maintaining JATENZO's market authorization and ensuring patient safety.
- FDA Post-Marketing Commitments: Upon approval, the FDA may require post-marketing studies to further assess safety or efficacy in specific populations or under real-world conditions. Compliance with these commitments is mandatory.
- Adverse Event Reporting: Manufacturers are obligated to monitor and report adverse events associated with JATENZO to regulatory authorities. This pharmacovigilance system helps detect potential safety signals.
- Labeling Updates: New safety information or efficacy findings can necessitate updates to JATENZO's prescribing information. These label changes can impact prescribing practices and patient selection.
- Manufacturing Standards: JATENZO must be manufactured according to Current Good Manufacturing Practices (cGMP). Regulatory inspections of manufacturing facilities ensure ongoing compliance.
- International Approvals: For global market expansion, JATENZO would require separate regulatory approvals from agencies like the European Medicines Agency (EMA) and others. Each jurisdiction has its own requirements and timelines.
Strict adherence to all regulatory requirements and proactive management of post-market surveillance are fundamental to the sustained availability and success of JATENZO.
Key Takeaways
JATENZO operates in a growing TRT market driven by an aging male population and increasing diagnosis of hypogonadism. Its primary competitive advantage is its oral administration, differentiating it from injectable and transdermal alternatives. Patent protection and regulatory exclusivities are critical for maintaining market exclusivity and profitability, with the timing of generic entry being a key future consideration. The drug's pricing and reimbursement strategy, alongside patient assistance programs, dictate market access and affordability. Sales performance, driven by clinical data, physician adoption, and patient experience, will determine its financial trajectory. Robust post-market surveillance and regulatory compliance are essential for sustained market authorization.
FAQs
What is the primary mechanism of action for JATENZO?
JATENZO is an oral formulation of testosterone undecanoate. Upon absorption, it is hydrolyzed to release testosterone, which then enters the bloodstream. Testosterone is the primary male sex hormone and is responsible for the development and maintenance of male reproductive tissues and secondary sexual characteristics.
Are there any specific contraindications for JATENZO use?
Yes, JATENZO is contraindicated in men with known hypersensitivity to testosterone undecanoate or any of its ingredients. It is also contraindicated in men with active prostate cancer or suspected prostate cancer, male breast cancer, or a history of these conditions. Additionally, it is not indicated for men with severe lower urinary tract symptoms, severe sleep apnea, or men who are pregnant or may become pregnant.
How does JATENZO compare in terms of dosing frequency to other TRT options?
JATENZO is typically dosed twice daily with food. This is more frequent than many injectable testosterone formulations (e.g., every 1-2 weeks) or some transdermal options (e.g., daily patches or gels). The convenience of oral administration is weighed against the more frequent dosing schedule.
What are the most common side effects associated with JATENZO?
Common side effects reported in clinical trials include increased hematocrit, acne, prostate-specific antigen (PSA) increased, hypertension, pain in extremities, and indigestion. It is important for patients to discuss potential risks and side effects with their healthcare provider.
Can JATENZO be used by women?
No, JATENZO is specifically indicated for the treatment of hypogonadism in men. It is not approved for use in women and could cause serious harm if used by them, including virilization.
What is the role of food in the administration of JATENZO?
JATENZO must be taken with food to ensure adequate absorption. The presence of food in the gastrointestinal tract enhances the bioavailability of testosterone undecanoate. Dosing without food may result in sub-therapeutic testosterone levels.
Cited Sources
[1] Information derived from general market analysis reports on the TRT market, typically found in pharmaceutical market research publications. Specific attribution for precise percentages can vary by report and year.
[2] Market research reports from firms such as Grand View Research, MarketsandMarkets, or Mordor Intelligence focusing on the Testosterone Replacement Therapy Market. Specific figures are subject to update based on the latest reports.
[3] U.S. Food and Drug Administration (FDA). (2020, March 27). FDA approves Jatenzo (testosterone undecanoate) capsules, the first and only FDA-approved oral testosterone replacement therapy for men with hypogonadism. Press Release.
[4] Drug pricing information, typically sourced from pharmacy benefit managers, drug pricing databases like GoodRx or KFF, or manufacturer websites. Average Wholesale Price (AWP) and Average Selling Price (ASP) data evolve and are subject to negotiation and discount.